Q: Calculate the percent dissociation of crotonic acid (C;H,CO,H) in a 1.7 mM aqueous solution of the…
A:
Q: O Use the following radial probability function to justify i. why the B than has a first lower…
A: B has first ionisation energy greather than Be because in the valence cell of boron has 2p1 ,it…
Q: commas separate options Reagent 1list: ch3mgbr h2o, lialh4 h2o, ch3Br, CH3OH reagent 2: PCC,…
A:
Q: Consider the titration of 100.0 mL of 0.200 M CH3NH2 by 0.100 M HCl. Calculate the pH for through…
A:
Q: Which of the following best describes the Ksp expression for Pb(OH)2? OAP62 (2 x [OH]) OB [Pp2*j (2…
A:
Q: Draw the 2D network structure of crystalline AND amorphous B2O3
A: Given that, the 2D network structure of crystalline AND amorphous B2O3
Q: Jsing the thermodynamic information in the ALEKS Data tab, calculate the standard reaction entropy…
A: CH4(g) + 2O2(g) --> CO2(g) + 2H2O(g)
Q: What volume of water must be added to a 26.33 mL solution of a pH = 1.17 solution of HClO4 to raise…
A: Initial Volume = 26.33 Initial pH = 1.17 Final pH = 3.53
Q: If the solubility of a drug is 3.8 g/L and in chloroform 224.9 g/L calculate its partition…
A: Solubility of drug in water = 3.8 g/L Solubility of drug in chloroform = 224.9 g/L
Q: A researcher employed at Phenomenal Phenols, Inc. has been asked by her supervisor, O. H. Gruppa, to…
A:
Q: Calculate the temperature at which the wavelength of a neutron is 1.36 Å. 4.0 K
A: The answer is given below
Q: The acid dissociation constant K, of carbonic acid (H,CO,) is 4.5 x 10 '. а Calculate the pH of a…
A:
Q: Enzymes do not require activation energy do not change requirment of activation energu increase…
A: Enzyme generally helps a reaction to proceed spontaneously by minimizing/loweing the activation…
Q: Calculate the moles, to the nearest .01 mol, of O2 that will be prepared through the decomposition…
A: Recall the decomposition reaction of potassium chlorate : 2 KClO3 -------> 2 KCl + 3 O2 8.3…
Q: H.
A: -CHO functional group is known for aldehde.
Q: Question 11 Calculate the pH at the equivalence point for the titration of 60.7 mL of 0.574 M NH3…
A:
Q: Predict the sign of AS°rxn (positive or negative) for the following reactions: - v isopropanol…
A:
Q: What is the empirical formula for a compound that is comprised of 54.0% Sodium, 8.50% boron and…
A: The simplest formula or the empirical formula provides the lowest whole number ratio of atoms…
Q: How many milliliters of 15.7% (v/v) acetic acid would you need to use to get 39 milliliters of…
A: Given-> (V/V)% = 15.7 % Volume of acetic acid solution = 39 millilitres
Q: Ammonia, NH,, reacts with oxygen to form nitrogen gas and water. 4 NH, (aq) + 30,(g) → 2 N,(g) + 6…
A: Given-> Weight of NH3 = 3.25 gm Weight of O2 = 4.88 gm Volume of N2 = 0.850 L T = 295 K P = 1.0…
Q: (a) Place the following gases in order of increasing average molecular speed at 25°C: CO, SF, H,S,…
A: In this question, we have to arrange the following gases in order of their increasing average…
Q: How many sites of diversity are there in this combinatorial library? NN NN HN `NH2 он `NH2 6 4 8 3
A: To determine number site of diversity , we would check the positions on which different atoms or…
Q: 3. Determine relationship for electrochemical potential of system, where R insolvable.
A: We have find out relationship for electrochemical potential of system, where R insolvable.
Q: Consider these compounds: A. CUCO3 B. A92CO3 C. MNCO3 D. Ni(OH)2 Complete the following statements…
A: To compare solubility of given salts we would use their Ksp values. The salts whose Ksp is higher…
Q: Calculate the percent dissociation of trimethylacetic acid (C,H,CO,H) in a 0.15 mM aqueous solution…
A:
Q: Draw all possible resonance structures for SO2, SO,, and SO. Use the resonance structures to solve…
A:
Q: Calculate the AS° in J mol K-1 of the following reaction from the values in the table. 3 Fe(s) + 2…
A:
Q: What is the energy of the radiation emitted when an electron in a hydrogen drops from an energy…
A: 13) Given, Electron in a hydrogen drops from n = 6 to n = 4 ninitial = 6 nfinal = 4 Energy of the…
Q: Why does phosphorus have a larger first ionization energy than sulfur? Select all that apply. a)…
A: Phosphorous (Z=15) Configuaration = 1s2 2s2 2p6 3s2 3p3 Sulfur (Z=16) Configuaration = 1s2 2s2 2p6…
Q: Part C The salt ammonium nitrate can follow three modes of decomposition: (a) to HNO3 (9) and NH3…
A:
Q: If the equations for the extreme lines (a and e) using units shown in graph above are: y= 2.6x + 3.0…
A: We have find out Vmax for enzyme.
Q: We want to use Epsom salt (magnesium sulfate; bath salt) instead of common salt (sodium chloride)…
A: Salting out occurs ionic solution of high ionic strength to reduce solubility. Salting out is…
Q: name of the binary molecular compound
A:
Q: Select which of these values is the correct osmotic pressure that a solution of 0.15 mmol/L NaCl…
A: Osmotic pressure: Osmotic pressure is the minimum pressure which needs to be applied to a solution…
Q: Question 23 Which of the following statements is/are true about the equivalence point of a strong…
A: At equivalence point no of moles of acid and base are equal. So, all the strong acid will get…
Q: 48.0 mL of AGNO3 solution are reacted with excess Na,CO3 solution to form Ag2CO3 precipitate. If…
A:
Q: Molarity
A:
Q: What happens to the particles of a precipitate if nucleation is the dominant process by which they…
A: Nucleation is the process in which the crystals grow in a solution by forming a regularized pattern…
Q: Indicate whether EACH statement is true or false A. true A buffer is an aqueous solution composed of…
A: Given statements: A buffer is an aqueous solution composed of two weak acids. A buffer can absorb…
Q: 6) In qualitative terms, how does Bohr's model account for the atomic spectrum of hydrogen? More…
A:
Q: Please make a brief introduction about Glutamic Acid.
A: Glutamic acid is one of 20 amino acids. Amino acids are the chemicals that make up the proteins that…
Q: what volume of
A:
Q: 1. Identify A-E in the scheme below: CH:(CH2),Br B NaH LDA A Isobutyl bromide 1) DIBAL-H 2) H30* E.
A: Initially reactant is given and we have to determine multiple products at various steps of provided…
Q: Calculating reaction entropy using the standard mölår ehtroples. Using the thermodynamic information…
A:
Q: Which of the following is not a conjugate acid-base pair? CH3COOH and CH3COO- HPO42- and…
A: Concept: Conjugate acid and base: According to the Brønsted–Lowry definition of acids and bases, a…
Q: Write the formula of an alkene containing 19 carbon atoms.
A:
Q: Will acidic food coked in a cast iron skillet become FE2+, enriched because of a reaction between…
A:
Q: s as conjugate acid (CA), conjugate base (CB), a
A:
Q: Define the following terms and give an example of each: saturated hydrocarbon, unsaturated…
A: In the given question we need to define some terms. The explanation of saturated hydrocarbon ,…
Q: What is the molar concentration of Na+ ions in 0.0350 M solutions of the following sodium salts in…
A: Given :molar concentration of Na2SO4 solution = 0.0350 M To calculate :- molar concentration of Na+…
What is the product of the following reaction?
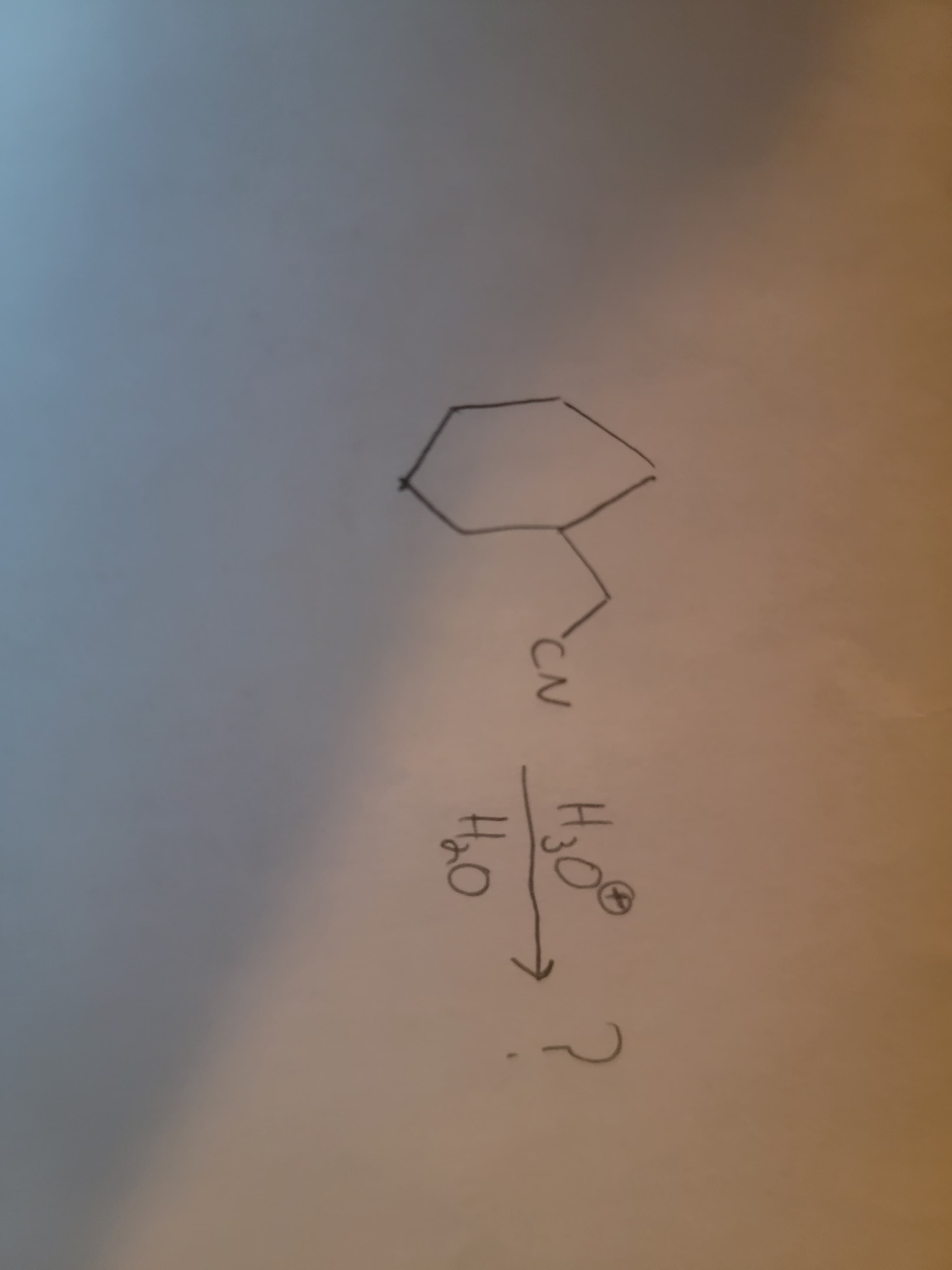
Step by step
Solved in 2 steps with 1 images


