Q: Balance the following redox reactions by the oxidation state change method. K2Cr2O7 + FeSO4+H2SO4 K2...
A: Since you have asked multiple questions, we will solve first one for you. For any particular questio...
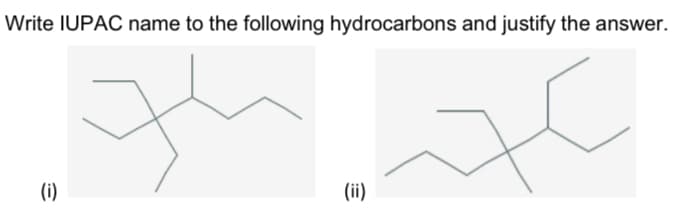
Q: Answerr the following
A: The VSEPR theory explains the structure of the molecule on the basis of valence electrons. The full ...
Q: Indicate the oxidation state of every individual element in the following compounds. g. SO2 j. Na,SO...
A: The total number of electrons that an atom either gains or loses in order to form a chemical bond wi...
Q: calorimetry (show complete solution)
A: Working formula Q = m*S*ΔT where Q = heat liberated by the neutralization reaction, m = mass of the ...
Q: Write the Kc Expression for the Lab’s Reaction Background: The purpose of this lab is to experimenta...
A: At the equilibrium the concentration of reactant and product remain constant over time. It is ex...
Q: calorimetry (show complete solution)
A: Given: Volume of HCl = 5 ml Molarity of HCl = 0.900M Volume of NaOH = 10 ml Molarity of NaOH = 0.750...
Q: Differentiate between H3PO4 and H2C204.
A: H3PO4 is a triprotic acid. Its name is Phosphoric acid. It is an inorganic weak acid. It has Phosph...
Q: Write the product that will be formed with Ethyl Alcohol in the acidic environment of Benzoic Acid.
A:
Q: If a tank contains 453 mol of propane, what volume of propane gas could be supplied at 22°C and 14.2...
A: Given the pressure of propane gas is --- 14.22psi Temperature T=22oC The number of moles of propane...
Q: result of hydroxy benzene and heptane in a bromination test.
A: Bromination test is used to check whether the molecule is saturated or not. In bromination test, bro...
Q: Indicate the oxidation state of every individual element in the following compounds. Fractional oxid...
A:
Q: 1. How many moles of Al are necessary to form 76.2 g of AlBr₃ from this reaction: 2 Al(s) + 3 Br₂(l)...
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost ot...
Q: Using the kinetics data below, determine the orders for A,B, and C. Write the rate law. Then determi...
A:
Q: How many grams of aluminum chloride are needed to prepare 553 mL of a 0.61 M solution?
A:
Q: please I want an answer
A: Given Formula = C4H8 IUPAC Name = ?
Q: 12.2) At 460°C, the reaction SO2(g) + NO2(g) ⇌ NO(g) + SO3(g) has K = 84.7. All gases are at an init...
A:
Q: A gaseous mixture containing 1.5 mol Ar and 3.5 mol CO2 has a total pressure of 7.5 atm. What is the...
A:
Q: Please complete table. Silver Nitrate Solution, AGNO3 Solubility Solubility in HCl in NH3 Anion Reac...
A: Solubility depends upon completely solubility product.
Q: ideal gas law
A: The gaseous state is the state of matter with the least intermolecular force of attraction between m...
Q: This reaction was monitored as a function of time: A+B A plot of 1/[A] versus time yields a straight...
A: To determine the rate law equation of the given reaction. A → B
Q: Calculate the pH pf following solutions: (i) 0.1M solution of acetic acid with 3% dissociation (ii) ...
A: The pH can be calculated by using the formula pH= -log10 [ H+]
Q: Gaseous CH4 and H2O were mixed in a 0.064 L flask at 1800 K. At a certain time, the flask contains 0...
A: given data is volume of the flask is 0.064L temperature is 1800K after some time concentration of ...
Q: Q 1: i) Why in parallel reactions one reactant is giving different products, and how we can decide a...
A: The reaction in which a substance or reactant reacts or decomposes two different way to give two di...
Q: Based on the given information. Need help with the following question. Thank you :) Using a simple d...
A: Given: We have to determine the product using D using C and E.
Q: Need help with this question. Thank you :)
A: Organometallic reagents, specially grignard reagents are synthesized in inert solvents like ethers o...
Q: Need help with this question. Thank you :)
A: Organo-Lithium are the compounds which contain lithium and carbon (R-Li) bonds. Organo-magnesium are...
Q: The answer I have is incorrect.
A: Conjugate base of H2CO3 is = ?
Q: Can I have help with this question?
A: From the molecular formula and NMR data we can predict the structure of that compound.
Q: Determine if the compound is aromatic, anti-aromatic, or non-aromatic
A:
Q: BCC vanadium has 1.999 atom per cell , the lattice cell parameter 0.3030 nm , the number 3 3 (of vac...
A: Unit cell is the smallest portion of a crystal lattice that shows three dimensional pattern of of wh...
Q: Calculate the wavelength?
A: Woodward–Fieser rules are several sets of empirically derived rules which attempt to predict the wav...
Q: Provide the systematic name of the major product after 1-butylcyclopent-1-ene reacts with HBr.
A: Given information: Reaction of 1-butyl-cyclopent-1-ene with HBr
Q: If the pH of a solution is increased from pH 5 to pH 7, it means that the: (please select one correc...
A:
Q: 1. You have 150.0 mL of a 0.517 M solution of Ce(NO₃)₄. a. You have 150.0 mL of a 0.517 M solution o...
A: "Since you have asked multiple question, we will solve the first question for you. If youwant any sp...
Q: Nitrogen and hydrogen react to form ammonia, like this: N, (g)+3H,(g) 2NH, (g) Suppose a mixture of ...
A: A reversible chemical reaction can move in either forward or backward direction. The stage of a reve...
Q: fischer projection fomula
A: To represent Fisher projection formula, carbon atoms must be in a straight line and the most oxidizi...
Q: Be sure to answer all parts. Give the overall reaction order that corresponds to a rate constant wit...
A: We know that, general formula for the calculation of order of the reaction from units of the rate co...
Q: Calculate the planar density of (100) and (110) plane and linear density of [111] and [110] directio...
A:
Q: Can I have help with this question?
A: IR spectroscopy is used to determine the structure of the molecule.
Q: A sample of xenon gas at a pressure of 1.05 atm and a temperature of 29.1 °C, occupies a volume of 1...
A: Boyle's law: According to this law, the pressure of a gas of a fixed amount at a constant temperatur...
Q: 10) A pizza has an area of 3.44 m2. Perform a conversion to each of the following units. a) in^2 b) ...
A: A pizza of area 3.44 m2 Conversions 1. m2 into in2 We know that 1m = 39.37 in there fore 1m2 = (39.3...
Q: Compare the carbon and hydrogen %age between Naphthalene and Toluene.
A:
Q: 1. Answers only item d and e.
A: Redox reactions are those in which there is simultaneous oxidation and reduction going on. Decrease ...
Q: If 10.0 moles of O_{2} are reacted with excess NO in the reaction below, and only 4.9 mol of N*O_{2}...
A: Given data: The moles of O2 reacted= 10.0 mole. The moles of NO2 obtained=4.9 mol. Given reaction: 2...
Q: For the following reaction H₂(g) + I₂(g) ⇌ 2HI(g) ∆H° = -10.64 kJ/mol and ∆S° = 21.48 J/mol・K. A clo...
A: This can be solved using the equations of ∆G°. The steps are given as,
Q: The missing of atoms from the lattice sites is called : a) Vacancy defects, B) interstitial defects,...
A: The lacking or missing of atoms or ions in any crystal structure or lattice is called solid defect. ...
Q: Hydrogen chloride and oxygen react to form chlorine and water, like this: 4 HCl(g) + O2(g) 2 Cl,(9) ...
A: The relationship between the ratio of partial pressures of products and reactants of a reaction in e...
Q: Name the following compounds
A: 1) In primary amine root name is based on the longest chain and the chain is numbered so as to give ...
Q: Which of the following are state functions? (i) energy, (ii) work, (iii) enthalpy, (iv) heat, (v) te...
A: In thermodynamics, state function is a property of a system whose value depend only on the state of ...
Q: Q4 (a) Calculate the pOH pf following solutions: (i) 0.01M solution of KOH considering 100% dissocia...
A:
Step by step
Solved in 2 steps with 2 images


