Q: AHjattice ΔΗ, II
A:
Q: Titration of a 0.485 g sample by Mohr method required 36.80 mL of the standard 0.1060 M AgNO3…
A: The complete balanced reaction ; Cl- + Ag+ → AgCl(s) Given in the question, Mass of sample =…
Q: A 0.3311 -g sample of pure iron (55.847 g/mol) wire was dissolved in acid, reduced to the +2 state,…
A: We need to find molar concentration of cerium ion.
Q: A solution of I3- was standardized by titrating freshly dissolved arsenious oxide (As4O6, FM…
A: The titration is a process in which the amount of solution required to neutralize the another can be…
Q: a) Cr.0,2- + Fe* → Fe3* + Cr3+ (in acidic solution) b) Mn* + BiO3 → MnOq¯ + Bi³* (in acidic…
A: a) Cr2O72-+Fe2+→Fe3++Cr3+ The oxidation state of Cr is +6. b) Mn2++BiO3-→MnO4-+Bi3+ The oxidation…
Q: Titration of 0.2342 g of pure Na,C,O, (134.0 g/mol) required 33.45 ml of KMNO, solution according to…
A: We have to calculate the Molarity of KMno4 solution
Q: 3. What color would I expect a solution of chromate ions to be after I add an excess of acid?…
A: The ion which can change its color when the medium is changed from acidic to basic or vice versa,…
Q: A pure oxide of iron weighing 1.000 g is fused with K2S2O7 and the fused material dissolved in acid.…
A:
Q: 1. Dissolving NaNH, in water will give: Answers A - D -2 A. A solution containing solvated Na+ and…
A:
Q: A 1.000-g sample containing KHC204, H2C204, and impurities required 38.22 mL of 0.1000 M NaOH for…
A: 1.582 m moles of KHC2O4 is present
Q: A 1.000-g sample containing KHC204, H2C204, and impurities required 38.22 mL of 0.1000 M NaOH for…
A: The answer is 1.582 m moles of KHC2O4
Q: 5.4 5.5 6.1 Conclusion 6.2 What is observed when the Na₂SO3 solution is added to the CuCl₂ solution?…
A: Disclaimer “Since you have asked multiple question, we will solve the first question for you. If…
Q: Which member of each pair produces the more acidic aqueous solution: CuCl or Cu1NO322,
A: Acidic strength determines the tendency of acid for the dissociation extent.
Q: What is the pH of a solution of 0.400 M CH₃NH₂ containing 0.100 M CH₃NH₃I? (Kb of CH₃NH₂ is 4.4 ×…
A: Concentration of CH3NH2 = 0.400 M Concentration of CH3NH3I = 0.100 M Kb for CH3NH2 = 4.4 × 10-4 pH…
Q: For breakfast, the nihilistic Rick Sanchez ate 100 g of cereal mixed with 20 g of milk. A 1.500 g…
A: Given: The molar mass of N (nitrogen) is 14.07 g/mol. For cereal: The amount of cereal consumed is…
Q: How many grams of PbF2 (molar mass = 245.2) will dissolve in 300 mL of 0.30 M NaF solution? The Ksp…
A: Given information: Molar mass of PbF2 = 245.2 g Volume V = 300 ml 0.30 M NaF solution. KSP for PbF2…
Q: What is the pH of a solution of 0.400 M CH₃NH₂ containing 0.240 M CH₃NH₃I? (Kb of CH₃NH₂ is 4.4 ×…
A:
Q: Chemistry What is the percentage purity of a sample of impure oxalic acid dihydrate if a sample…
A:
Q: 1. In a redox titration, 12.50 mL of 0.800 mol/L K2Cr2O7 (aq) was used in an acidic solution to…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: ou have 10 mL of an 8% aqueous solution of H2Cl6Pt. The density of the solution is 1.05 kg.L-1…
A: Values given in the question are as follow: the volume of the solution is 10ml an 8% aqueous…
Q: Fe or Fe2*
A: Let us draw the Lewis structures for Fe+2 and Fe+3 .
Q: Part A What is the net ionic equation of the reaction of FeCl2 with NaOH? Express you answer as a…
A: In net ionic equation , we do not write spectator ions
Q: ChO3- + Sh → Ch- + Sh2+ (acidic solution)
A: The balance equation is: ChO3- + 3Sh + 6H +→ Ch- + 3 Sh2+ +3H2O Explanation:
Q: D) A sada- Lime Sample is g0% NaoH and 10%% CaO · 1f 3 gm is dissolved in 250 ml, what is the tutal…
A: Solution Given :mass of sample = 3 gram Normality of solution = 0.51 N Volume of solution = 250 ml…
Q: D. NaCl (aq) + acidified MnO2 (s)
A:
Q: A 0.4320 -g sample of pure iron (55.847 g/mol) wire was dissolved in acid, reduced to the +2 state,…
A:
Q: 2. 100.0-mL sample of spring water was treated to convert any iron present to Fe2+. Addition of…
A: In the given question we have to calculate the concentration of iron in 100 mL sample of spring…
Q: * OWLV2 | Online teachinX C The Deep Blue Compound (+…
A: Concentration of the solution (solution 2) obtained on diluting 0.164 M HCl can be determined as…
Q: A pure oxide of iron weighing 1.000 g is fused with K2S2O7 and the fused material dissolved in acid.…
A: Solution -
Q: Calculate the (Ka) value for the reaction of CH3COOH with NHạ that the pH of the solution is (6.46)…
A: The Ka for the reaction of acetic acid (CH3COOH) is calculated as shown below.…
Q: If the alkalinity of the water is 306ppmNa2CO3 what does this mean?
A: The concentration of a compound can be expressed in ppm (parts per million). One ppm means that one…
Q: n alkaline sample of sodium compounds weighing 1.196 g was dissolved in water, cooled to 15°C,…
A: Given: Mass of alkaline sample of sodium compounds= 1.196 g Normality of H2SO4= 1.058 N The volume…
Q: What is the Ksp for iron (II) sulfide if the iron (II) concentration in a saturated solution is…
A: Introduction Solubility of any particular substance is equivalent to the maximum amount of the…
Q: In following these steps . Complete the table and provide th given chemical reactions in the…
A: Since the table has not been given, thus only the reactions of group 3 cations are provided. Group 3…
Q: For breakfast, the nihilistic Rick Sanchez ate 100 g of cereal mixed with 20 g of milk. A 1.500 g…
A:
Q: Determine the CBOD and NBOD of 125 mg/L of CH;COOH solution. Assume the following reaction! CH3COOH…
A: Solution -
Q: What is the pH of a solution of 0.400 M CH₃NH₂ containing 0.220 M CH₃NH₃I? (Kb of CH₃NH₂ is 4.4 ×…
A: Henderson Hasselbalch equation. We can use pOH = pKb + log [salt] / [base]
Q: A 0.1017 g sample of KBrO3 (MM=166.1) was dissolved in dilute HCI and treated with an unmeasured…
A:
Q: A sample of water in contact with solid Fe(OH)3 is found to have a pH of 7 .5. If no other dissolved…
A: To calculate the pOH of the solution, the equation used is: pH + pOH = 14 ........(1) pOH is…
Q: b. O2 + Sb→ H2O2+ SbO2¯ in basic solution
A: Solution - Balanced equations - A balanced equation is an equation for a chemical reaction in which…
Q: A 10.0 g sample of KCgH5O4 (A)(MW = 204.23 g A) was titrated with 50.00 mL of KOH (B) to endpoint.…
A: The balanced reaction taking place is given as, => Given: Mass of KC8H5O4 taken = 10.0 g. The…
Q: In the chemical changes experiment on copper, which of the steps can be omitted if the goal is only…
A: To determine, which of the steps can be omitted if the goal is only to obtain dry copper (II) oxide…
Q: 4. Potassium dichromate is often used to standardize reducing agents like Fe2+: Cr:0, + 6Fe +14 H*…
A: Given data,Volume of Fe2+=34.00mL=0.034LMass of K2Cr2O7=398.5mg=0.3985gMolar mass of…
Q: What volume of a 0.150 N KI solution is required to react in basic solution with 34.1 mL of a 0.216…
A: The given data contains, Normality of KI = 0.150 N. Volume of KMnO4 = 34.1 ml. Normality of…
Q: H.W 1) Calculate the number of meq. of H;C:0.2H;0 (fw= 126.1) in 0.5 gm of the pure compound…
A:
Q: The acid ionization constant for Fe(H 2O) 62+(aq) is 3 ×10-6. Calculate the pH of a 0.10 M solution…
A: Acid ionization constant denoted by Ka is the equilibrium constant measure of tendency of an acid to…
Q: ) What amperage is required to plate out 0.250 mol Cr from a Cr3+ solution in a period of 8.00 h?
A:
Q: Please help me out with making flowchart with thes steps. Write the result. Procedure 1.…
A: The flow chart for the given steps is shown below
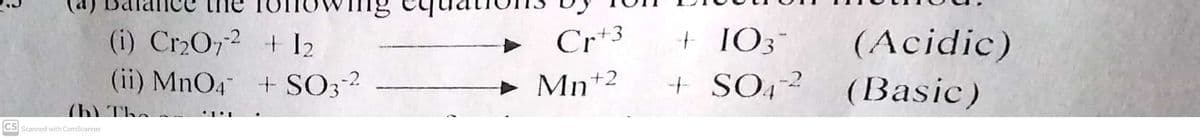
Step by step
Solved in 3 steps with 2 images

- Maria was tasked to determine the concentration of a Fe2(SO4)3 solution. She prepared 5 different standards of Fe2(SO4)3 using the table below as her guide. She ran the standards and the unknown solution through UV-VIS spectroscopy and recorded the absorbances of each solution. HINT: use the dilution equation to determine the concentration of Fe2(SO4)3 in each test tube before preparing the standard curve. NOTE: No need to force the line to pass through zero. Just graph the data as is. What is the slope of the line of the standard curve from the given data? What is the concentration of the unknown solution?A third spectrophotometric method for the quantitative analysis of Pb2+ in blood uses Cu2+ as an internal standard. A standard containing 1.75 ppb Pb2+ and 2.25 ppb Cu2+ yields a ratio of (SA/SIS)std of 2.37. A sample of blood is spiked with the same concentration of Cu2+, giving a signal ratio, (SA/SIS)samp, of 1.80. Determine the concentration of Pb2+ in the sample of blood.please quickly ,within 1hour thanks ! If it takes longer than an hour, i don't need the answer (1) List the detectors ofHPLC. Describe the advantage and disadvantage for two wideused detectors
- An ethanol solution of 3.5 mg/100 ml of compound Y (150.0 g/mol) in a 1.00 cm quartz cell has an absorbance (A) of 0.972 at λmax=235 nm. What is its molar extinction coefficient? Report your answer to the correct number of significant figures. Do not include units in your answer. I need to know what is the correct number of sig figs for this question which will determine what the correct answer is.3. a. Briefly explain why internal standardization method is useful inanalytical chemistry?b. Why does a response factor of an instrument’s detector need to becalculated?Preparing a standard curve. 1. determine the absorbance max of [FeSCN]2+ it is 448.1 2. determine the concentration of FeSCN2+ in the stock solution. M1V1=M2V2 stock solution: ~0.200 M iron (iii) nitrate in 1M nitric acid intial volume: 0.3mL final volume: 10.3mL volume: 10mL ~0.002M potassium thiocyanate initial volume: 0.3mL final volume: 10.3mL volume: 10mL not sure if this is needed stock solutions: ~0.200M iron (iii) nitrate in 1M nitric acid: 0.207M ~0.002M iron (iii) nitrate in 1M nitric acid: 0.00209M ~0.002M potassium thiocyanate: 0.00193M
- Describe the following properties of chromatogram (including formula if applicable): Bandwidth Retention Volume Capacity Factor Selectivity Factor ResolutionA spectrophotometric method for the quantitative analysis of Pb2+ in blood has a normal calibration curve for which Sstd=(0.296 ppb−1)×Cstd+0.003. What is the concentration of Pb2+ in a sample of blood if Ssamp is 0.397?Concentration=2.5 X 10^3 μM OD400nm=0.859 1cm cuvette is used what is the extinction coefficient (μM^-1 cm^-1)
- Compare the resolution for the two components A and B when (tr)b = 5.0 min and (trla = 4.4 min using: 1-Colmn with L = 900 mm, H = 1.0 mm 2- Column with L= 900 mm, H = 3 mmThe nitrate in water was analyzed using a spectrophotometric method. The absorbances of the standards and the unknown sample were obtained at 550 nm.Solution Absorbance 2.00 µM 0.0656.00 µM 0.20510.00 µM 0.33814.00 µM 0.47418.00 µM 0.598Unknown 0.402 Write the equation of the lineWhat is the standard deviation of the calibration curve?Low Concentrations of Ni-EDTA near the detection limit have the following counts in a mass spectral measurement: 175, 104, 164, 193, 131, 189, 155, 133, 151, 176. Ten measurements of a blank has a mean of 45 counts. A sample containing 1.00 μM Ni-EDTA gave 1797 counts. What is the sensitivity in counts/M?



