Q: CO₂Me MeO₂C dimethyl fumarate A 1,3-butadiene A CO₂Me CO₂Me dimethyl maleate
A:
Q: 13) Draw Lewis structures for the following molecules: HI, CH3CH₂CH3, CH3OH, HSSH, O2, CS2 (SCS).
A: A Lewis structure is a representation of a molecule that shows the arrangement of atoms and the…
Q: The specific heat of a certain type of cooking oil is 1.75 J/(g-°C). How much heat energy is needed…
A: The specific heat, ‘c’ of cooking oil is 1.75 J/(goC)Mass, ‘m’ of oil is 2.34 kg=2340 gInitial…
Q: vis dot structure of methanol (CH3OH) is given below. Estimate the enthalpy of combustion of ethanol…
A: Enthalpy of the combustion of methanol is to be calculated.
Q: MISSED THIS? Watch KCV: Cell Potential and Concentration ; Read Section 20.6. You can click on the…
A: [Pb2+] = 0.0500 M[Cu2+] = 1.50 Mn = 2 electron transferEoCu2+/Cu = 0.34 VEoPb2+/Pb = -0.126 VEcell =…
Q: An ideal gas is contained in a cylinder with a volume of 5.1*10^2 mL at a temperature of 30.°C and a…
A: An ideal gas has negligible volume in compare to volume of the container, and the molecules of ideal…
Q: An aqueous solution contains 0.384 M acetic acid. How many mL of 0.203 M potassium hydroxide would…
A: Given => Molarity of acetic acid aqueous solution = 0.384 M Volume of this Solution = 225 ml pKa…
Q: Arrange the solutions in order of increasing acidity. Rank solutions from least acidity to greatest…
A:
Q: 1. What is the common name of CaCO3? Describe how it can be used in a real life situation. It's…
A: Given compound is : CaCO3 To describe: common name of the compound and its uses in real life.…
Q: What would be the solubility product constant if 0.337 g of lead (II) iodide (MW is 461 g/mol)…
A:
Q: Example: Write an equation for the precipitation reaction that occurs (if any) when you mix…
A:
Q: The nonvolatile, nonelectrolyte estrogen (estradiol), C18H2402 (272.40 g/mol), is soluble in benzene…
A: Mass of estrogen = 12.5 gramsVolume of benzene solution = 172 mLMolecular weight of estrogen =…
Q: Identify the class of organic substance for each of the following molecules. b) CH3CCH c)…
A: CH3CCH and CH3CH2CHO are given ,we need to predict the class of organic substance to which they…
Q: Design a buffer that has a pH of 9.71 using one of the weak acid/conjugate base systems shown below.…
A: A buffer solution is to be designed to get a desired value of pH from the given pairs of weak acid…
Q: Which organic starting material is needed to complete the following reaction? H₂C B ? A 1)…
A: oxymercuration–demercuration reaction, analkene is treated with mercury(II) acetate, Hg(OAc)2, and…
Q: The reaction of LDA with acetophenone produces halogenation an ylide an enol an enolate alkylation
A:
Q: A sample of rock was found to contain 8.3 mg potassium-41 and 0.48 mg of cacium-41. Calculate the…
A: GivenHalf Life of potassium = 4.1×10^10 yrsSample of rock contains 8.3mg of potassium+ 0.48mg of…
Q: How many valence electrons are in Ar²+? 2+
A:
Q: Complete the following two syntheses problems for each one giving the reagents and product from each…
A: The type of reactions used to carry out the given synthesis arePhotochemical radical…
Q: 6) Give IUPAC names for the following molecules en a) b) F F
A: Given Molecules -We need to write the IUPAC name of the above molecules.
Q: Calculate the volume, in L, of water that must be added to dilute 15.6 mL of 10.4 MHCI to 0.0900…
A:
Q: The activation energy for the gas phase decomposition of t-butyl alcohol is 274 kJ. (CH3)3 COH…
A: The activation energy of the reaction = 274 kJThe rate constant at 804 K i. e. (k1) = The rate…
Q: b. Balanced formula equation: HCNO (aq) + Complete ionic equation: Net ionic equation: + + + + +…
A:
Q: Compare and contrast the physical properties of elements in group 1,2 7
A: Let's contrast the physical characteristics of the elements in the periodic table's Group 1 (alkali…
Q: Gnereally speaking, which of the following is the most reactive to reactions with a nucleophile? A B…
A: We know priority order - Aromaticity > Mesomeric > Hyperconjugation > Inductive effect +I…
Q: What is the oxidation number of oxygen in K2O2? +2 -2 -1 +1 -1/2
A: Oxidation number is charge of the atom that exist by the gains or loses of electrons to form a…
Q: Which of the following molecules has the lowest pKa? A B с D OH A CF3CH₂OH B OH NO₂ D
A: pKa = - logKa Higher the acidity of the compound, higher the value of Ka and lower the value of pKa…
Q: Name the following compound; CH3 CH₂CHCHCH₂CH₂CH₂CHCH₂CH3 O 3-Methyl-8-propylnon-1-ene O…
A:
Q: This is the chemical formula for talc (the main ingredient in talcum powder): Mg3 (Si₂05), (OH)2 An…
A: Given :- Chemical formula for talc = Mg3(Si2O5)2(OH)2Number of moles of Mg = 0.014 moles
Q: Be sure to answer all parts. Ammonia has a Kp of 1.8 × 105. Find [H3O+], [OH¯ ], pH, and pOH for a…
A: Given,The concentration of Ammonia Solution= 0.550 M
Q: Draw the two possible products formed when this diene reacts with HBr. Do not include any byproducts…
A: When a conjugated diene i.e. alternate double bond molecules is treated with HBr, there is a…
Q: Question : HA is a weak acid. Its ionization constant, K₁, is 5.7 x 10-13. Calculate the pH of an…
A: IntroductionThe given ionization constant for HA is K = 5.7 x 10^(-13). This value tells us that HA…
Q: Why are the OH groups of carboxylic acids more acidic than alcohols? resonance stabilization of the…
A:
Q: CIO4* (use black sphere for CI) PH3 (use black sphere for P) H₂O (use black sphere for O)
A: The given problem is based on the concept involving Lewis theory and VSEPR theory to predict…
Q: Question 14 According to the PHET simulation, the average mass of the Mg atom is closer to Mg-24…
A: The average mass of a magnesium (Mg) atom, as determined by experimental measurements and…
Q: The KM value of lysozyme is 6.0 x 10-6 mol L-1 with hexa-N-acetylglucosamine as the substrate. The…
A: IntroductionAdditionally, the initial rate of the reaction, denoted as vo, is given as 3.2 µmol L^−1…
Q: Determine if the following processes are endothermic ( +∆?° ) or exothermic ( ―∆?° ). a. Combustion…
A: Processes with their corresponding classificationsCombustion of gasoline: Exothermic (-∆H) Water…
Q: COMPOUND LEWIS DOT STRUCTURE HCN (use black sphere for C) SO3 (use black sphere for S) NO₂™ (use…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first 3 sub-parts…
Q: A 27.2 mL sample of 0.383 M triethylamine, (C2H5)3N, is titrated with 0.371 M nitric acid. After…
A:
Q: Uranium hexafluoride is a solid at room temperature, but it boils at 56°C. Determine the density of…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: Freon-12 (CF₂ Cl2) is commonly used as the refrigerant in central home air conditioners. The system…
A: Pressure of gas have several units like mmHg, pascal, atm etc.Here we need to convert the pressure…
Q: 5. In the following reaction, the enthalpy of the forward reaction is energy for the forward…
A: IntroductionThe information provided includes the enthalpy change (ΔH) for the forward reaction,…
Q: The rate of effusion of a particular gas was measured and found to be 31.7 mL/min. Under the same…
A:
Q: Write a balanced equation between t-amyl alcohol(2-methyl-2-butanol) and hydrogen bromide at room…
A: The given reaction of t-amyl alcohol(2-methyl-2-butanol) and hydrogen bromide leads to a…
Q: For each of the following balanced equations, indicate how many moles and grams of all reactants and…
A:
Q: What happens when the codeine molecule (C18H21NO3) is exposed to air? What reactions are obtained
A:
Q: 15.38 Draw a stepwise mechanism for the following reaction. [1] NaH [2] H₂O H-D + NaOH
A:
Q: many MOLECULES of tetraphosphorus decaoxide are present in 4.02 moles of this compound ? molecules.
A: As we know, 1 mole of any substance contains 6.022 × 1023 molecules of that substance.
Q: 2A. What is the theoretical yield and limiting reagent for the Friedel Crafts acylation reaction of…
A: Friedel-Crafts acylation is an acylation of aromatics using an acyl halide and a Lewis acid…
Q: Which of the following compounds is(are) hydrolyzed to butanoic acid upon heating in H₂O, H₂SO4?…
A: -> Ester on hydrolysis give carboxylic acid and alcohol.-> Amide on hydrolysis give amine and…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

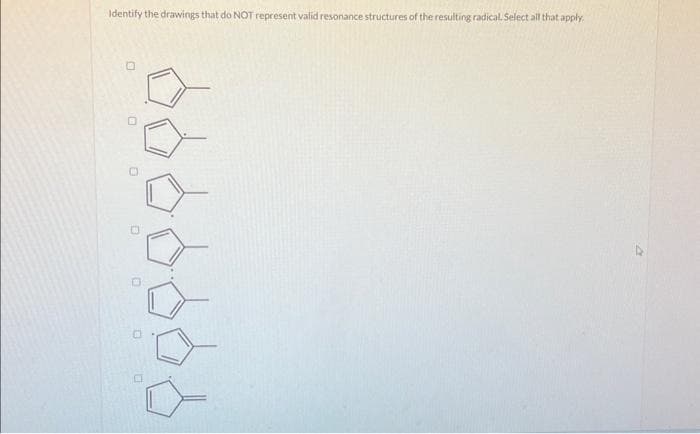
- Please guide me on how to draw the best resonance structure, thank you.How do you assign R or S to a molecule when the lowest-priority group is not oriented toward the back, on a dashed wedge?In a mass spectrometry, hogh energy protons is use to bombard the molecule so that it will break down into fragment. Is this true or false? True False
- Should the final resonance structure be included in the radical resonance? Or is the first and fifth structure the same?For 1 and 2, draw the appropriate arrows to show the bond cleavage and bond formations in these reactions. Show the important nonbonding electrons.That structure have 152 molecular mass, can you help me draw the fragments that represents the following m/z values: 106, 95, 81, 41, and 27
- Why is less energy required to break a C-H bond to form a more highly substituted radical? Can somebody summarize the words on this page to make it more understandable?For each of these molecules, draw the important resonance forms and predict which resonance form is likely to be the major contributor. Can you explain thanksWhich of the following indicated bonds will have the highest frequency IR stretch?

