Q: What reagent(s) need to be added to benzene to synthesize the following product?
A:
Q: [References] EMWORK bidium-87 decays by B production to strontium-87 with a half life of 4.7 x 1010…
A: Half life=4.7×1010year Atomic mass of Rb=86.90919 Atomic mass of strontium (Sr)=86.90888 Final…
Q: ty 4: Critical Thinking: Answer the following items. Why observing patterns is an important skill?…
A: The periodic table, also known as the periodic table of elements, is a tabular display of the…
Q: Show the electron configuration of the following elements and underline the configuration that shows…
A:
Q: ● How can the following compounds be separated? -CH3 -CH3 OH OH Kow = 15 pk = 3.0 -CH3 Kow = 40…
A:
Q: What is the IUPAC name for the following molecule? OH je 4-hydroxyhexan-3-one 3-hydroxyhexen-one…
A: For IUPAC naming following steps are required- First of all find the longest carbon chain…
Q: 5.00 g sodium metal reacts with excess sulfuric acid at 27°C and 110 kPa. 2 Na+ H₂SO4 → H₂+ Na₂SO4…
A:
Q: A student needs exactly 25.0 mL of a 1.5M solution of NaOH (molar mass: 40.00 g/mol). They have…
A: Given: Concentration of NaOH = 1.5 M Volume of solution required = 25.0 mL = 0.025 L…
Q: calculate the for the EO following reaction - 2 cr + 3 MnO₂ (s) + 1₂ H+ (aq) → 3 mm²+ (aq) + 2 cr3+…
A: The explanation is given below-
Q: Which of the following are enantiomers of each other? H H H A Br 114 B Br Br C H H Br D 11/ H
A: Enantiomers : enantiomers having opposite stereochemistry at chiral centers. Assign R,S nomenclature…
Q: Reaction 3: N₂(g) + 20₂(g) Reaction 2 can be described as an [Select] [Select] reaction 2NO₂(g) ΔΗ =…
A:
Q: ( When the following reaction is balanced under acidic conditions, what is the coefficient in front…
A:
Q: What is the molecular geometry of the molecule below? :F: I :F -Ec=N: a) linear Ob) trigonal planar…
A: In this question we have to tell the molecular geometry of the molecule.
Q: The equilibrium constant, Kc, for the following reaction is 1.29x10-2 at 600 K. COCI₂(g) =CO(g) +…
A: Given data,Kc of the reaction=1.29×10-2Initial moles of COCl2=0.353 molesVolume of vessel=1.0L
Q: over Short Answer 2. 3. Rank the following molecules from most acidic (1) to least acidic (4) (ень…
A:
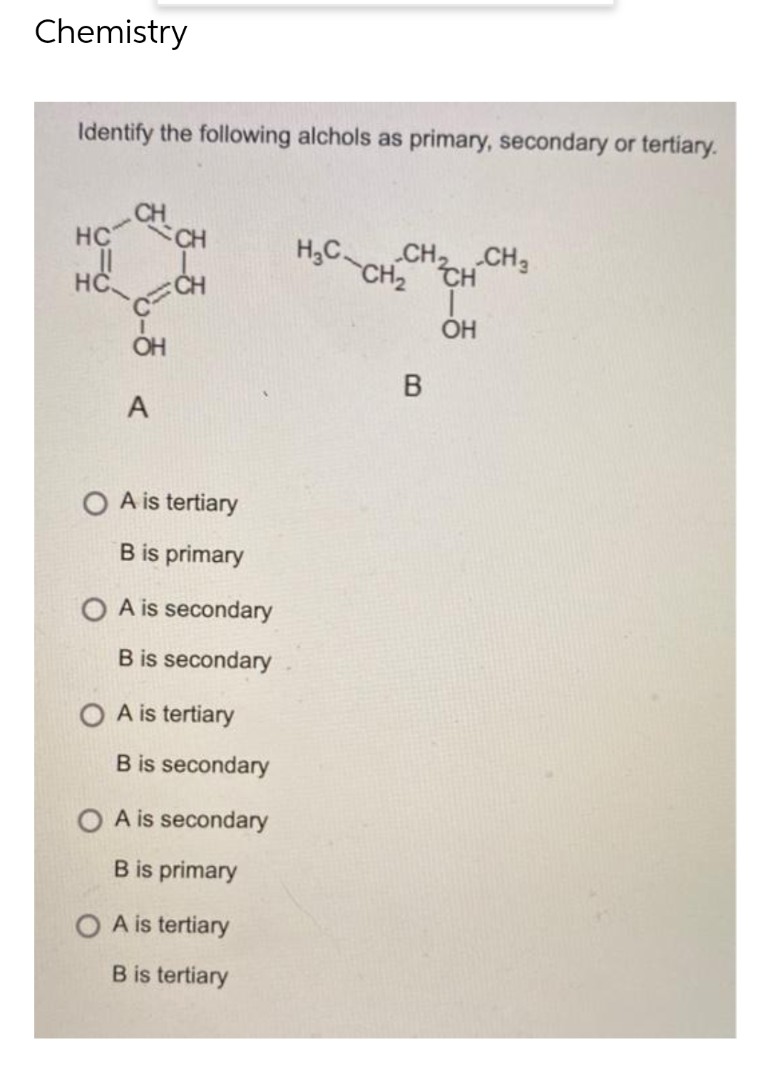
Q: Identify the following as pure substance, homogeneous solution, heterogeneous mixture or colloidal…
A: We have to identify which one is pure substance, homogeneous solution, heterogeneous mixture or…
Q: A buffer solution is prepared by adding 5.00 g of CH3COOH (acetic acid, a weak acid) to 7.50 g of…
A:
Q: [Ti(H₂O)6]4+, is colourless. k There are multiple transitional energies, and all visible wavelengths…
A: A complex compound contains central metal ion and ligand, ligands donates electrons to central metal…
Q: Fill in the blanks: Provide the necessary compounds to form heptane according to the given reagents.…
A: This is an example of preparation of Gilman reagent followed by synthesis of alkane by using Gilman…
Q: Fill in the blanks as instructed below. Enter only one of the provided word options each blank. Note…
A:
Q: 19 Which of the following ketoesters can be formed by Dieckmann cyclization? L: على علم بلنه DE A D…
A: Dieckmann cyclization : Intramolecular claisen condensation is known as Dieckmann cyclization.
Q: Fill in the blanks: The SN2 mechanism is most likely to occur in the substitution reaction of a in…
A:
Q: Fill in the blanks: Br H₂C. CH₂ CH₂ The major product of the elimination of HBr from the above…
A:
Q: What reagent(s) need to be added to benzene to synthesize the following product? CH3 Br CH3OCH 3…
A:
Q: Which of the following reactions would have a positive AS" ? (i) Na₂SO4(s)--> 2 Na*(aq) + SO4² (aq)…
A: Entropy order -> S(gas) > S(liquid) > S(solid) ->Hence , if number of gaseous moles…
Q: How many chiral carbon atoms are there in penicillin? H onex -0
A: A chiral carbon atom is a carbon atom that is attached to four different types of atoms or groups.
Q: CH3CH(OH)COOH has a stereogenic center and is achiral Select the correct response: True False
A: Note : Carbon is attached four different groups is called asymmetic carbon or chiral carbon.…
Q: Consider a gas cylinder containing 0.250 moles of an ideal gas in a volume of 6.00 L. The cylinder…
A: We have to predict the work for the gas
Q: 15. Consider the following information about four different acid solutions: Solution Acid…
A: Given : We have to write which solution is more acidic and why.
Q: A pure sample of Substance S is put into an evacuated flask. The flask is then heated steadily and…
A: In the heating curve , each region specific a particular process. So first we would specify each…
Q: Give the mechanism and Choose SO (A) (c) Ti) A H H III C ju) B comect option, ? H
A: In this question we have to tell the product of the reaction and also its mechanism.
Q: Draw the reaction for the Edman degradation product (PTH-amino acid) of the first amino acid residue…
A: We have to draw the reaction for the product formation.
Q: The table gives the concentrations of C₂H₂O(g) as a function of time at a certain temperature for…
A: The data given is,
Q: What are the E/Z configurations of the following compounds? CI CH₂Br H3CO CH=CHNH2 HO -OH CC13 HO…
A:
Q: A 10.00 g charcoal brick from a barbeque measures a C-14 decay rate of 5050 disintegrations per gram…
A: 1 gram of C-14 sample has a disintegration rate of 921 disintegrations per hour initially. Initial…
Q: Using the table of thermodynamic data provided, for the reaction below: CS2(1) + 302(g) → CO2(g) +…
A:
Q: Under which conditions is a reaction spontaneous at all temperatures, according to AG = ΔΗ – ΤΔS ?…
A: We have to predict the correct conditions.
Q: Using the table of thermodynamic data provided, for the reaction below: SiH4(g) + 8HNO3(g) → SiO2(s)…
A:
Q: What is the solubility of Cr(OH)₃ at a pH of 9.80? (Ksp Cr(OH)₃ is 6.70 × 10⁻³
A: Given - - > pH = 9.80 Ksp of Cr(OH)₃ = 6.70 × 10-31 We know that, 14 - pH = - log[OH-] 14 - 9.8…
Q: Pls help ASAP. Pls help in all asked questions.
A: a) Given: Lithium oxide decomposes into lithium metal and oxygen gas.
Q: Provide the necessary compounds to form heptane according to the given Ether A 2 Li B Pentane Cul -…
A: Given: We have to make heptane for the given reaction.
Q: In the submission boxes below, write out each of the reactions found when forming an aqueous…
A: Selenious acid is a weak polyprotic acid , so it would donate proton and form its conjugate base. As…
Q: 4) Circle the benzene analog that undergoes Friedel-Crafts alkylation/acylation most rapidly? -COOH…
A: 4. EDGroups increase the reactivity of benzen ring towards electrophile. Therefore anisole reacts…
Q: Suppose 12.0 mL of an unknown weak base (B) is titrated with 0.100 M HCI. The unknown base reacts…
A: Given, Reaction : B+HCl ⇌ BHCl Volume of unknown weak base = 12.0 mL Molarity of HCl =0.100 M
Q: What is the pH (to two decimal places) of a saturated aqueous solution of copper(II) hydroxide (at…
A:
Q: The vapor pressure of pure liquid A at 300K is 575 Torr and that of pure liquid B is 390 Torr. These…
A: Here we are required to find the total vapour pressure and the composition of liquid at 300k
Q: 3 27 Overall Reaction: CH4(g) + NH3(g) → HCN(g) + 3H2(g) Reaction ΔΗ° (kJ) -91.8 N2(g) + 3H2(g) →…
A:
Q: Balance formation reaction for the following compund Formation reaction of CH3CHCO2H (I) (propanoic…
A: The reaction given is the formation reaction of CH3CH2COOH (NOTE: The formula of propanoic acid…
Q: Reaction Method: Reaction Mechanism Sublimation, Elimination, Reaction with Metal, Ester Formation,…
A: Type of reactions: 1). Physical transformation reactions: Sublimation reaction : it's a phase…
Q: 14-7. Butadiene can be formed by dehydrogenation of n-butane: C¸H₁ ‹› C₂H₂ + 2H₂ This reaction is…
A: We have find out the answer.
Step by step
Solved in 2 steps with 2 images

- pl.s ans.werName of Cr3(AsO4)4Calcium ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) Ca (s) 0 0 41.4 Ca (g) 178.2 144.3 158.9 Ca2+ (g) 1925.9 CaC2 (s) -59.8 -64.9 70.0 CaCO3 (s, calcite) -1206.9 -1128.8 92.9 CaCl2 (s) -795.8 -748.1 104.6 CaF2 (s) -1219.6 -1167.3 68.9 CaH2 (s) -186.2 -147.2 42.0 CaO (s) -635.1 -604.0 39.8 CaS (s) -482.4 -477.4 56.5 Ca(OH)2 (s) -986.1 -898.5 83.4 Ca(OH)2 (aq) -1002.8 -868.1 -74.5 Ca3(PO4)2 (s) -4126.0 -3890.0 241.0 CaSO4 (s) -1434.1 -1321.8 106.7 CaSiO3 (s) -1630.0 -1550.0 84.0 Carbon ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) C (s, graphite) 0 0 5.7 C (s, diamond) 1.9 2.9 2.4 C (g) 716.7 671.3 158.1 CCl4 (l) -135.4 -65.2 216.4 CCl4 (g) -102.9 -60.6 309.9 CHCl3 (l) -134.5 -73.7 201.7 CHCl3 (g) -103.1 -70.3 295.7 CH4 (g) -74.8 -50.7 186.3 CH3OH (g)…

