Q: The C-H bond in acetone, (CH3)C=O, has a pką of 19.2. Draw two resonance structures for its…
A: Solution- The resonance structure with negative charge on carbon and oxygen are drawn below-…
Q: NH он d) O,N- -NH2 -NH2 O,N- -NH2 -NH2 CH3
A: Bases are defined as the molecules which can donate it's electron pair. The more easily a molecule…
Q: Select the stronger acid from each pair of compounds
A: Stronger acid strength is based on the ability of proton donation from that compound, which in turn…
Q: Which molecule is more acidic? H F F F F 2,2-difluoro isomer 3,3-difluoro isomer エ-Z エ-Z
A: This is multiple questions type So only first question is solved. F is electronegative is group and…
Q: NH2 NH2 NH2 NO2
A: The given molecules are: All of these compounds are basic in nature and basicity is inversely…
Q: 2.72 Answer the following questions about the four species A-D. :0: :0: :ÖH :ö: N: A в D a. Which…
A: We have to find: The two species that represent a conjugate acid-base pair, The two species that…
Q: Which of the following compounds is most acidic? A в D B
A:
Q: Predict which compound in each of the following pairs of compounds is more acidic and explain…
A: Acids are those compounds which leave hydrogen ion on addition of water. Bases are those compounds…
Q: .C. H2C CH2 H2C. CH2 S
A:
Q: 1. Rank the following compounds from most to least acidic at the indicated hydrogen, and explain…
A:
Q: bel the three bold hydrogens in compounds A, B, and C in order of acidity, with 1 being the most c…
A: Acidity can be defined as tendency of a compound to lose hydrogen ion.
Q: Rank the compounds in each group in order of increasing acidity.
A: The more the electronegative substituent, the stronger the acid. This is because it weakens the bond…
Q: What indicated hydrogen atom is the most acidic? B HAA H₂ of C CH3 D
A: We have to select the most acidic H
Q: Explain why the N—H proton in X is more acidic than the O—H proton. X was a key intermediate in the…
A: Since the acidity molecule depends on the stability of the anion formed after removal of the H. And…
Q: Draw the structure of a constitutional isomer of compound B that fits each description. a. an isomer…
A: The given compound is,
Q: 4. The explicitly indicated proton in the following compound is much more acidic than a typical…
A: According to Bronsted-Lowry concept, acid is a substance which can lose proton and forms its…
Q: F F но LOH HO
A: The Compound which can release H+ easily will be more acidic.
Q: 1. The correct number of a acidic protons in the following compounds: (a) (b) (c) (d) (e) (f) а. (a)…
A: The answer of the above question is given in step 2
Q: Which C–H bonds in the following molecules are acidic because the resulting conjugate base is…
A: Given compounds,
Q: Rank the compounds in each group in order of increasing basicity.
A:
Q: 2. Which structure is the stronger acid, A or B? Explain your answer. F Br A B
A:
Q: Rank the compounds in each group in order of increasing acidity.
A: The ease of losing H+ and thereby forming a stable anion would make the acid amongst the choices to…
Q: 15. For each of the pairs of compounds below, identify the more acidic compound. List the factor…
A: NOTE : according to the policy we can only Answer first three sub parts. The question is based on…
Q: n each of the following compounds, identify the most acidic hydrogen. Draw the conjugate base of the…
A: Most acidic hydrogen is which gives most stable conjugate base after it removed.
Q: 3.38 Draw the conjugate acid for each of the following bases: a. b. c. NANH2 d. H20 е. f. NH, g. h.…
A:
Q: The C – H bond in acetone, (CH3)2C = O, has a pKa of 19.2. Draw two resonance structures for its…
A: The pair of acid and base differing from each other by just one proton is called conjugate acid-base…
Q: I. Using an energy diagram, compare the pKa's of the ionizable protons in the following molecule. H…
A:
Q: Which proton is the MOST acidic in the following compound? CI CI A B A D в) в
A:
Q: Rank the compounds in each group in order of increasing basicity.
A: Basicity- The ability of compound to donate lone pairs of electrons or accept protons is called…
Q: 8. Indicate which compound is more acidic. Explain your answer. он он NO2 ČH3
A: Phenols are acidic in nature. The acidity of phenols is higher than the alcohol and lower than the…
Q: Rank the indicated hydrogen atoms in the following molecule in order of increasing acidity, i.e. 1…
A: The most acidic hydrogen is the one where the negative charge is much stabilized.
Q: Which of the following would be the best base for performing the following elimination reaction? *…
A:
Q: 3. 1
A: To identify the most acidic hydrogen atom in a compound, consider the conjugate base formed after…
Q: OEt EtO OEt
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: Which compound in each pair is the stronger acid?
A: Since the strength of acid depends on the stability of its conjugate base formed after removing…
Q: Considering the protons highlighted in red in the two compounds shown below, which compound is more…
A:
Q: Rank the labeled protons in each compound in order of increasing acidity.
A: 1) the order of acidity for the mentioned H will be b > a > c This is because when we remove…
Q: Which anion (A or B) is the stronger base?
A: Given both the compounds are bases and it known that bases haev the ability to abstract the proton…
Q: с. d. .CI HO
A:
Q: What indicated hydrogen atom is the most acidic? B A H₂ C. C H₂ C CH3 -D
A: Acidity of Hydrogen atom of a molecule depends upon stability of it's conjugate base. More is the…
Q: Rank the nitrogens below in order of decreasing basicity (most basic to least basic) N. (a) 'N' (c)…
A: More the s-charter of Nitrogen (N) has less basic character as more strongly bonded and has less…
Q: Rank the following ions in order of increasing basicity.
A: Since we answer upto 3 sub-parts,so we'll answer first three. Please resubmit the question…
Q: Rank the following in order of increasing basicity. The first compound in your list should be the…
A: The basicity of the compound depends on the availability of the lone pair of electrons. More is the…
Q: 14 HC it ○ニ エ 土
A: Acidity is defined as the ease with which a molecule can donate protons. if it can donate PROTONS…
Q: What will help you determine whether a compound or a Lewis base? (a) Color (b) "P NMR spectrum (c)…
A: The question is based on the concept of acidity & basicity. A Lewis acid is a substance which…
Q: Which anion (A or B) is the stronger base?
A: INTRODUCTION: Bases is defined as the substance which releases OH- ion in aqueous solution and also…
Q: a) Draw the conjugate base formed when the following compound is deprotonated. b) Rank the following…
A: • This molecule is symmetrical to both the conjugate base identical.(b) When conjugate base is…
Q: 2.3. Determine whether the hydrogen atom for H2O is protic or hydridic. Protic Hydridic 2 points…
A: 2.3 Protic Due to hydrogen bonding. 2.4 Aprotic Due to tendency to release H- and forming stable…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

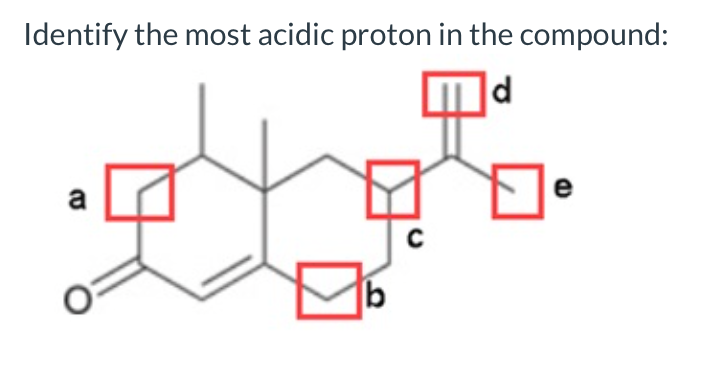
- Summarize the relationship between pKa and acid strength by completing the following sentences: a. The higher the pKa of an acid, the stronger or weaker the acid. b. The lower the pKa of an acid, the stronger or weaker the acid.which compound has the most acidic proton?Identify and explain the most acidic proton on the following molecule.
- Predict which compound in each of the following pairs of compounds is more acidic and explain yourreasoning for each.(a) HSO4− or HSeO4−(b) NH3 or H2O(c) PH3 or HI(d) NH3 or PH3(e) H2S or HBrrank the indicated protons in decreasing order of acidityWhich of the following is the strongest acid? Group of answer choices CH3OH CH3OH2+ CH3NH2 CH3NH3+
- Rank the given compounds in order of decreasing basicity. 1=most basic, 4=least basicThe C – H bond in acetone, (CH3)2C = O, has a pKa of 19.2. Draw two resonance structures for its conjugate base. Then, explain why acetone is much more acidic than propane, CH3CH2CH3 (pKa = 50).Identify the more acidic compound in each of the following pairs:

