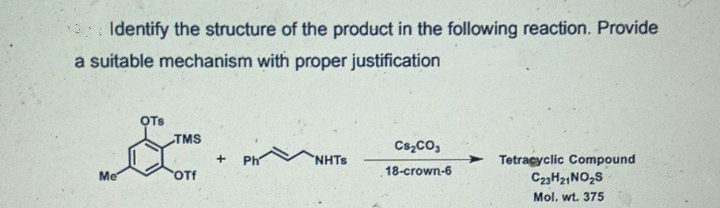
Identify the structure of the product in the following reaction. Provide a suitable mechanism with proper justification OTs TMS Cs,co, > Tetragyclic Compound C23H21NO,S Ph 'NHTS Me OTf 18-crown-6 Mel ud 375
Q: An inexperienced graduate student treated dec-5-ene with borane in THF, placed the flask in a…
A:
Q: ) Purpose the efficient synthesis for the following transformations: B) Give the product(s) for each…
A: The synthesis of the given compounds are given below-
Q: The reactivity of the below compounds towards nucleophiles is: R CI NH2 OR a O a. b>cod a Ob. acd» b…
A: The derivatives of carboxylic acid are derived by the replacement of OH group of carboxylic acids,…
Q: 3. The utilization of diols as intermdiates towards the ultimate oxidative cleavage of alkenes is an…
A: Aldehyde on reaction with Grignard reagent followed by THF to form secondary alcohol.
Q: OH RCOCI „NH2 EtO OEt A Base H,0*
A:
Q: (c) Write the product(s) for the following reaction with supportive mechanism : ŞiMez KH →? Bu, Нех…
A: Here the given reaction is an example of Peterson Olefination. So, in this reaction alkene is…
Q: Me N. Meo OMe MeN- MeN- H...) -N- MeN- Br MeCN, 100 °C NaH, THF; Br H then 6M HCI MeO2C, -Me Me…
A:
Q: Provide the products that complete the following reaction equations. Note: unless otherwise…
A: Since you have posted a question with multiple sub- parts, we will solve first three sub- parts for…
Q: 18. Suggest a plausible mechanism with structures of the following reaction: Br, H,0 CH3 CH2B.…
A: In the reaction mechanism of the above reaction, first the alkene will attack on the Br2 molecule as…
Q: 10. Nitril group, -CN, is a meta director. Predict the product for the following reaction, by…
A:
Q: MeO MeO. OH 5.0 eq. Li, ETOH HO. liq. NH3 MeO OMe OMe
A: In presence of Li/ liq NH3 or Na/ liq NH3 benzene undergoes birch reduction. Due to birch reduction…
Q: divls depends oin the moisture content of the reaction mixture. Propose a detailed mechanism for…
A:
Q: b. Provide mechanisms and/or products for the following transformations. Po Ln, Cul, PhBr 1. HBpin,…
A: Note : 1-hexyne reation with HBpin followed by PdLn is called Suzuki Miyura coupling. 1-hexyne…
Q: Boc- `N H CO₂Me i) 9-BBN ii) Ph-Br, 5 mol% PdCl₂(dppf), K3PO4, DMF Вос. `N H CO₂Me
A:
Q: H3C CHз (DMF) POC13 Na,CO3, H20 H thiophene P
A: Concept - In the formylation reaction, formyl group (-CHO) will attached to the compound. The…
Q: Give the major product of each of the following reactions: Bra d. CH,C=CCH, CH,I, excess HBr e.…
A: According to Markonikov's addition, the more electronegative part goes to the more substituted C in…
Q: HCIO4 Br2 MECO2H Br
A:
Q: Provide a complete, detailed and stepwise mechanism for the following acid 3. catalyzed hydration…
A: SN1 reaction is known as Unimolecular Nucleophilic substitution Reaction, where there is no need of…
Q: dict the organic products of the following reaction. Show stereochemistry clearly. The (R) or (S)…
A: The reagent OsO4 introduces two hydroxyl groups on the same side to yield enantiomers as shown…
Q: Draw the major organic product(s) generated in the reaction below. Pay particular attention to…
A: Welcome to bartleby ! As per company rules we can't answer more than one question per session . So,I…
Q: Predict the Product of the reaction and indicate regiochemistry and stereochemistry when relevant.…
A: An alkene gets converted to viccinal diols(1,2-diols) in presence of OsO4. The stereochemistry of…
Q: An inexperienced graduate student treated dec-5-ene with borane in THF, placed the flask in a…
A:
Q: 1. LIAIM(OCICH.J, CH, 2. но он он H;SO4
A: The question is based on the concept of organic reactions. we have to identify the Product formed…
Q: (c) Predict the product(s) for the following reaction with supporting mechanism. i) SeO, Aq. dioxane…
A: This ragents are oxidizing agents.Oxidation takes place with DDQ and SeO2.
Q: 4. Write the product and give mechanism. (i) *CO (ii) RCN/.. -CH3 PMe Rh Me,P
A:
Q: Provide reasonable arrow pushing mechanisms for a through d explain your reasoning for your chosen…
A: As per our company guidelines we are supposed to answer only one question. kindly repost other…
Q: please show the detailed mechanism for each step for the reaction below. OTBDPS Me TMANO (anhydrous)…
A: Step 1 : alklyne addition to two coblat atoms. Step 2 : olefin addition to one of the coblat atom.…
Q: SA. Identify the product and write a detailed mechanism to account for its formation CH3 H,o PRODUCT…
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: NaBH, H+ agua CH,OH H,0 H+ Ag,0 1) H+ 2) NH, OH-
A: In this question, we will draw the products for all reaction by using mechanism and intermediate…
Q: b) Write the product C and D. Suggest possible mechanism : 1) LIAIH4 C,H5-CH CH–CHO 2) H„O, RT 1}…
A: LiAlH4 is a good reducing reagent.It reduces double bond when they are polarized(in resonance) as in…
Q: otherwise indicatec provide the major organic products of the reactions. Please inciude…
A: Note: According to our guidelines we are supposed to answer only first three subparts. Kindly repost…
Q: 5) Draw the product structure of the compound generated when ethylbenzene is treated with CO, HCI,…
A: Gattermann-koch and grignard reactions
Q: Which of the following reaction scheme is expected to afford the following product? HO ОН O The…
A:
Q: Deduce the structure of compounds E -Lin the road map bbelow You must be able to show the detailed…
A:
Q: (c) Predict the product(s) for the following reaction with supporting mechanism. i) SeO2 Ag diovone
A: Selenium dioxide, SeO2 is an oxidizing agent generally employed in the allylic oxidation of alkenes…
Q: 2. Predict the major product(s) of the following reaction sequences. 1. I, NaOH (excess reagents),…
A:
Q: Br (CH3)COK + CHB13 Br (CH3);COH
A: Singlet- Carbene : This Carbene has a lone pair of electron for donation again , it has a vacant…
Q: b) Ozonolysis of an unknown alkene yields the diketone shown in the scheme below. Suggest the…
A: This is the ozonolysis reaction of Alkene.In this reaction ozonide is an intermediate form and PPh3…
Q: b. Provide mechanisms and/or products for the following transformations. Pd°Ln, Cul, PhBr 1. HBpin,…
A: 1. 1-Hexyne reacts with HBpin produces borate. This is used in Suzuki coupling ( see below). 2.…
Q: Predict the product and provide a detailed mechanism. Bu,SnCl (0.1 eq) NaBH4 (1.2 eq) hv…
A: Introduction: For a good radical coupling reaction, a mixture of tertiary butyl tin chloride and…
Q: H,Cro, CH, NH, H+ NABH,CN -он -H NABH, но H- -он HO, CH,OH H H,C.
A:
Q: 3) Provide mechanisms for the following synthetic sequence: heat OH () но OH /H* (ii) 0-C %3D OMe H.…
A:
Q: b. Provide mechanisms and/or products for the following transformations. Pd°Ln, Cul, PhBr 1. HBpin,…
A:
Q: Give complete mechanism of following reactions: CH, 0-H HBr CH; --C-CH–CH, → (CH,), C–CE–CH; a. H;O…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: но осн 1. b. CI CI H DMSO HO, 2. NET3 C. of PPH3
A: Hemiacetal deprotection Swern oxidation Wittig reaction
Q: fd} Provide the products of the following reactions with mechanism : 1) DIBAL-H →? Hexane, -70 °C fi…
A: DiBAL- H is a reducing agent which reduces ester to aldehyde.
Q: Give the major organic product(s) of each of the following reactions or sequences of reactions. Show…
A: 7. Alkyl halide reacts with the sodium azide to form alkyl azide and the alkyl azide is reduced into…
Q: 3. Complete the following reactions by giving products in each case. KMnO4 1) LIAIH, 2) H3O* NH2OH…
A: Answer:- This question is answered by using the simple concept of chemical reactions of organic…
Q: Meo OMe 3. The Mannich reaction can take many forms, including reactions where aromatic heterocycles…
A: Step 1 : sn2 reaction between alkyl bromide and pyridine nitrogen. Step 2 : Michael addition…
Step by step
Solved in 3 steps with 1 images

- give reactions and intima mechanism of following reactions-Acetoxybenzene (PhOC(=O)OCH3) is much less reactive than ethoxybenzene (PhOCH2CH3) in electrophilic aromatic substitution reactions. Suggest an explanation for this result, based on an analysis of the inductive and resonance electronic effects of the two substituents on the stability of theWheland intermediate for para substitution by an electrophile E+( plz give detail step by step mechanism )
- Identify the pericyclic reactions in the followingreaction schemes. Give the complete reactionname and indicate the course of the reaction withthe aid of the arrow notation.write the mechanism and predict the product, and includ the stereochemistryExplain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.
- Identify the pericyclic reactions in the followingreaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.plz provide DETAILED written explanation and AVOID USING MECHANISMS.w how enols, enolate ions, andenamines act as nucleophiles. Predictthe products of their reactions withhalogens, alkyl halides, and otherelectrophiles. Show how they areuseful in synthesis.
- Propose a suitable mechanism based on the observation .Suggest a detailed mechanism for the reaction below. Represent the product in the most stable conformation and also its stereochemistry.Provide a mechanism for the following reaction and rationalise the reactivity in terms of the 3 dimensional structure of the starting material and a consideration of the appropriate orbital interactions.

