Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter21: Hydrocarbons
Section: Chapter Questions
Problem 2STP
Related questions
Question
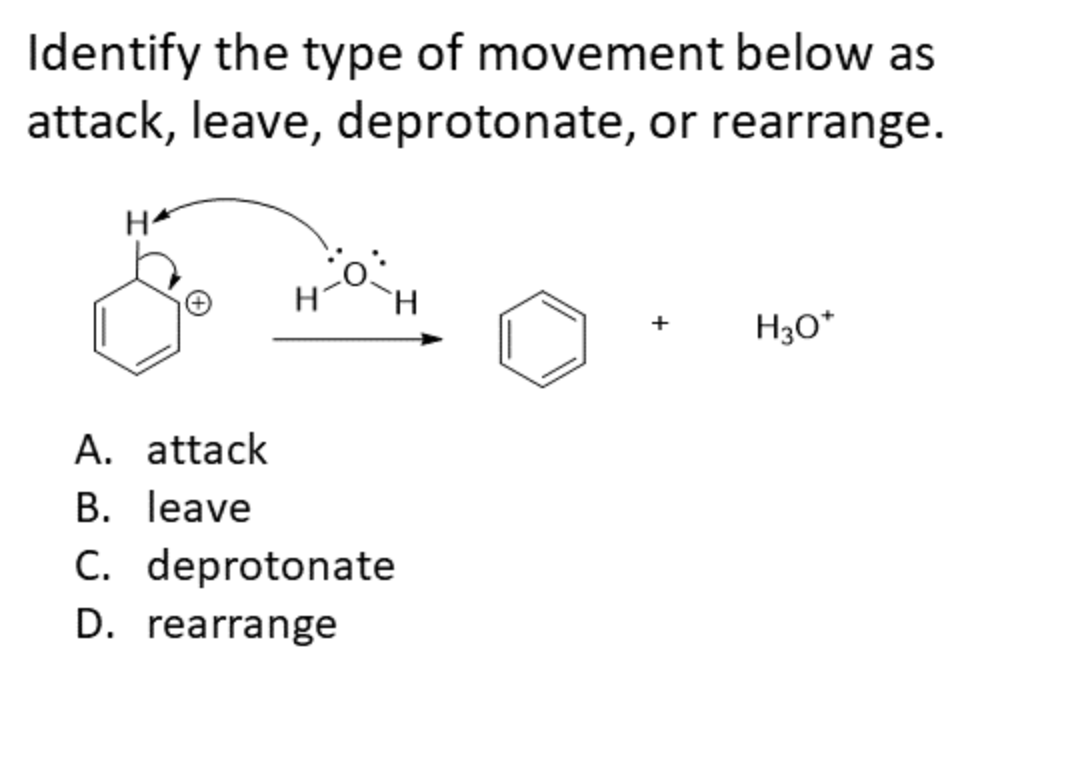
Transcribed Image Text:Identify the type of movement below as
attack, leave, deprotonate, or rearrange.
H30*
A. attack
B. leave
C. deprotonate
D. rearrange
Expert Solution
Step 1
Solution
Benzene is very vulnerable to electrophilic substitution reactions compared to chemical reactions because it loses its aromaticity throughout addition reaction. As benzine contains delocalized electrons spanning over carbon atoms within the ring, it's extremely engaging to electrophiles and is additionally extremely stable to electrophilic substitutions. Generally, the electrophilic substitution reaction of benzine may be a three-step method involving:
Generation of the electrophile.
Intermediate carbocation formation.
Removal of a nucleon from carbocation intermediate.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co
