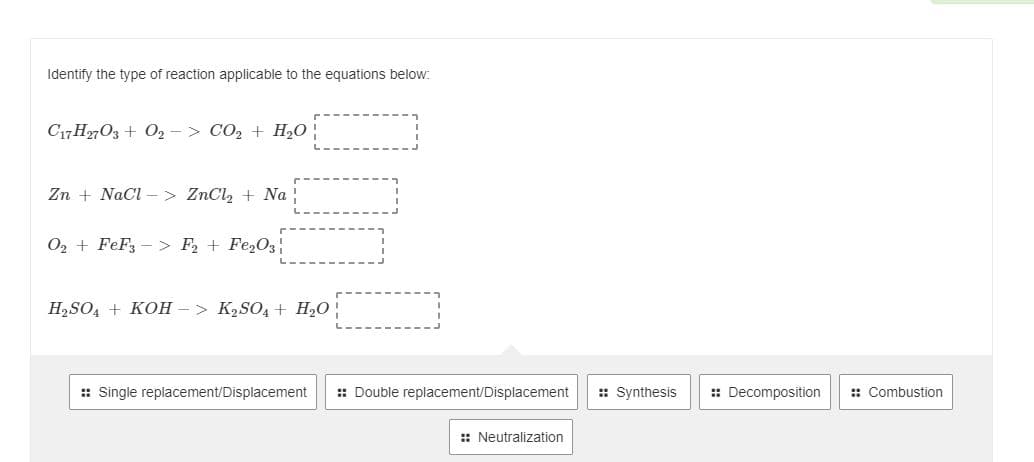
Identify the type of reaction applicable to the equations below: C17 H2703 + 02 - > CO2 + H,0 Zn + NaCl –> ZnCl, + Na O2 + FeF3 – > F, + Fe,O3 H,SO, + KOH – > K2SO4 + H2O | : Single replacement/Displacement : Double replacement/Displacement :: Synthesis : Decomposition : Combustion :: Neutralization
Q: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW:…
A: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution…
Q: reactant: 0.400g of p-amionphenol (MW=109.1) 0.450 ml of acetic anhyride (mw= 102.1 d= 1.08 ml)…
A: Hello. Since the question contains more than three subparts, the first three subparts shall be…
Q: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCI & 2 g C6H1206 are present in 100 mL ORS solution (MW:…
A: Given that - Mass of NaCl = 0.35 g Mass of KCl = 0.15 g Mass of NaHCO3 = 0.25 g Mass of C6H12O6…
Q: Given: 0.35g NaCI, 0.25 g NaHCO3, 0.15 g KCI & 2 g C6H1206 are present in 100 mL ORS solution (MW:…
A: Mass of NaHCO3 = 0.25 g Volume of solution = 100 mL Concentration of bicarbonate = ?
Q: 5) compare your calculated AH value with a published value.
A: Given, Experimental △H for MgO = – 598 kJ/mol (as calculated in question 4) The standard…
Q: A sample of processed meat scrap weighing 4.000 g is digested with concentrated H;SO, and…
A: Given: Weight of meat scrap sample = 4.000 g To calculate the percentage protein in the meat scrap.
Q: A solution of I3- was standardized by titrating freshly dissolved arsenious oxide (As4O6, FM…
A: The titration is a process in which the amount of solution required to neutralize the another can be…
Q: The ascorbic acid, C6H806 (176.124 g/mmol) content of a bottle of peach juice containing 2.50 L was…
A: I have attached a detailed solution for the question in the following steps:
Q: Calculate AG° for the below chemical reaction using the provided AG° values. 2 PCI3 (g) + O2 (g) → 2…
A: Given: ∆Go POCl3(g) = -502.5 kJ/mol ∆Go PCl3(g) = -269.6 kJ/mol ∆Go O2(g) = 0 kJ/mol
Q: Q2. a) What is Atom Economy? Calculate Atom Economy for the following reactions, i) Synthesis of…
A:
Q: Oa is required for complete Combustion
A: Number of moles = mass/molar mass Mass = moles × molar mass
Q: A mixture of 2.00 g BaCl2.2H2O (molar mass = 244.2g/mol) and 1.00 g of NazPO4.12H20 (molar mass =…
A: Given that: Mass of BaCl2.2H2O = 2.00g Molar mass of BaCl2.2H2O = 244.2g/mol Moles =…
Q: A www-awn.aleks.com Significant Figures Counter Chapter 5. The Nervous System-Neurology and…
A: Given, the mass of ammonium perchlorate = 9.30 g The balanced chemical equation is given - The…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: Here we are required to balance the redox reaction
Q: rption of 333 Jo antanenus at -
A: When ∆G of the Reaction is negative then Reaction is spontaneous. and if ∆G of the reaction is…
Q: Write balanced formation equations for the following, indicate proper states:…
A: The compounds given are N2O (g), Na2S2O7 (s), C2H6O (l) and B3N3H6 (s). Formation reactions are…
Q: Mass of unknown metal Carbonate.0058g Volume of uniknown solution CmL) Volume of Hcl added CmL)…
A: Since you have posted a question with multiple questions , we will solve first question for you. To…
Q: 3 Bal, (aq) + 2 Nag POq cag) 7 BalPO42 + l6 Nal (aq Cónsider the reaction of 30.0mL of Oi270m Balz…
A:
Q: Vitamin C (ascorbic acid, C&H&O6, 176.12 g/mol) can be measured by redox titration with iodine…
A: Given: mass of ascorbic acid = 0.315 g volume of I3- solution = 29.2 mL molar mass of ascorbic acid…
Q: Using the data table and balanced chemical equation below, calculate the AS (to one decimal place)…
A: We are having the values as given in the table: We need to calculate change is entropy in the whole…
Q: Complete the combustion products of sucrose. Balance the equation. C12H22011 O2 Name of products:…
A: Combustion is the process of burning of a compound; in which the compound reacts with excess of…
Q: Calculate the standard enthalpy of formation of Caco3 at 298K in kļ/mol from the foll CaO (s) + CO…
A: Given reaction is : CaO (s) + CO2(g) →CaCO3 (s) ; ∆H298o= - 42.85 Kcal/molCa (s) +…
Q: Questions are attached
A: The molarity, molality, formality and normality are expressions for concentrations of solutions. The…
Q: Given: 0.35g NaCI, 0.25 g NaHCO3, 0.15 g KCI & 2 9 C6H1206 are present in 100 mL ORS solution (MW:…
A: In 100 mL of ORS solution : The mass of Na = 0.35 g The mass of NaHCO3 = 0.25 g The mass of KCl =…
Q: Consider the series of reactions to synthesize the alum (KAl(SO4 )2 · xH2O(s)) from the…
A: To calculate the moles of alum formed from the given number of moles of each reagent, we would use…
Q: 63.) The compounnd sodium thidsu Hate pentahycloate, NagS,O3 - H20, s importont commercially to the…
A: 63. Given unbalanced equation, S8(s) + Na2SO3(aq) + H2O(l) → Na2S2O3 · 5 H2O(s)
Q: Calculale we paIts per m MON 117 A 0.8512-g sample containing KCIO; (122.55 g/mole) and impurities…
A: Given that - Mass of sample containing KClO3 = 0.8512 g Molar mass of KClO3 = 122.55 g/mole…
Q: A sample of processed meat scrap weighing 4.000 g is digested with concentrated HSO. and…
A: The solution of the given question is
Q: 1. A 1.2040 g sample containing sodium carbonate and inert material was dissolved in water and…
A: 1.) First we would write a 4 chemical equation between sodium carbonate and HCl . Then Calculate…
Q: 56g of the coal was kjeldahlized and NH3 gas evolved was absorbed in 50 ml of 0.1 N sulphuric acid.…
A:
Q: Which of the tollowing practicos will not come under groen chomistry? O A Using HyO, for bleaching…
A: Green chemistry is also called sustainable chemistry. Green chemistry is the design of chemical…
Q: What is the solubility product, Ksp of NiCO3 if its solubility is 3.61e-4 M? 7.21e-4 1.80e-4…
A: Ksp of salt can be calculated using solubility.
Q: Suppose you standardize a Na2S203 solution by dissolving 25.0 mg of KIO3 in acid and allow it to…
A:
Q: What is the enthalpy of reaction (in kJ/mol rxn) for CaCO3(s) → CaO(s) + CO2(e) if Ca(s) + Cgraphite…
A:
Q: Predict the product/s of the following reactions AND identify the type of reaction taking place.…
A: The various types of reactions are:- Combination Reaction Displacement Reaction Decomposition…
Q: Consider the series of reactions to synthesize the alum (KAl(SO4 )2 · xH2O(s)). If you start the…
A: The limiting reagent in a chemical reaction is a reactant that is present in smaller quantity and…
Q: NaOHbom 0.001 het place in waste viase bon ringe ef and refill with de noitoloa bion oilaxo lo…
A: Given, Molarity of H3PO4 = 0.481M. Volume of Ba(OH)2 = 24.7mL. Molarity of Ba(OH)2 = 0.695M. The…
Q: Predict the products and balance the following eqn. and identify the type of reaction 7) Bacl +…
A: The given reaction are as follows, a) BaCl2 (aq) + H2SO4 (aq) → b) C6H12 (g) + O2 (g)→ c) Zn(s)+…
Q: A sample known to consist of NaOH orNaHCO3, or Na2CO3 or possible compatible mixtures of these,…
A: 2Na2CO3+2HCL⇌2NaHCO3+2NaCL b) 2NaHCO3+2HCL⇌2NaCL+2H2CO3
Q: Write the oxidation state of the carbon in the following compounds. CH3-CH-C. Ci O =C=0 H. H--C-Ci
A: Oxidation state of carbon: Number of bonds with which it is attached more electronegative atom
Q: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW:…
A: molarity is defined as the number of moles of solute upon the volume of solution
Q: Complete the following equations to show the products that form in each of the reactions we observed…
A: Solution: The balanced chemical equation are as follows: (a) Ca(s) + 2 H2O(l) → Ca(OH)2 (aq) + H2…
Q: i. NaH, DMF ii. CH3CH₂Br iii. NaOH, H₂O iv. H₂O, heat ?
A:
Q: 6- In the reaction below by complete reaction of 2 mol KCIO3, 120.6 g KCl is produced. Calculate the…
A: In the given reaction KClO3 decomposes into KCl and O2. The equation for the balanced chemical…
Q: A 10.0 g sample of KCgH5O4 (A)(MW = 204.23 g A) was titrated with 50.00 mL of KOH (B) to endpoint.…
A: The balanced reaction taking place is given as, => Given: Mass of KC8H5O4 taken = 10.0 g. The…
Q: Provide the organic product(s) of the following reaction. .CI 1. Mg, ether 2. CO2 3. HС, Н20
A:
Q: Mail-Francesca A Tantillo-Out X Homepage-CHM150-251 Chen X Question 19 of 20 Vitamin C (ascorbic…
A:
Q: Calculate the standard enthalpy of formation of CaCO3 at 298K in kJ/mol from the following data: CaO…
A: The standard enthalpy of formation of a compound means the enthalpy of the compound for its…
Q: Use the reactions and AG° values below to determine AG° for: 3 CH4(g) + 4 O2(g) → C3O2(g) + 6 H20(g)…
A: Hess's law states that the standard reaction enthalpy is the sum of the standard enthalpies of the…
Q: A sample containing a mixture of SrCl, · 6 H,0 (MW = 266.62 g/mol) and CsCl (MW = 168.36 g/mol)…
A:
Step by step
Solved in 4 steps

- Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)One litre of a saturated aqueous solution of Ag2SO4 (MW = 311.79 g mol- 1) at 25 °C is evaporated to dryness. 4.844 g of Ag2SO4 residue was produced. What is the solubility product (Ksp)?Really hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. When the compounds analyzed by Robrob were passed on to the next test, anotherscientist, Kikoko, needed to determine the molecular weight of the active compound. They created a solution with a concentration of 20.0%w/w from the solid active compound and the solution had a density of 1.40 g/mL. From this solution, 10μL was taken and mixed with 190μL of reagent and water. It was analyzed and showed a concentration of 5x10-4 M compound.a. What is the concentration (in molarity) of the 20%w/w solution? b. Why was the concentration of the solution expressed as %w/w initially? c. What is the molecular weight of the active compound?
- SOLUBILITY – reactions with conc. Sulfuric acid Based from the video link --- https://www.youtube.com/watch?v=JmlDkC6IdKA&t=21s 1 mL of conc. H2SO4 was placed in a separate test tube. The test samples, cyclohexane and cyclohexene were added separately. Observe what happened after the reaction. Question: Based from your observation, write the results of the following samples: (a) Cyclohexane, (b) Cyclohexene, and (c) Toluene. Thank you very much!iven the data in the table below, ΔH°rxn for the reaction C2H5OH (l) + O2 (g) → CH3CO2H (l) + H2O (l) is ________ kJ. -79.0 -1048.0 -476.4 -492.6 The value of ΔH°f of O2 (g) is required for the calculation.An impure sample of Na3PO3 weighing 0.1 g is dissolved in 35 mL of water. A solution containing 45 mL of 3% w/v HgCl2, 30 mL of 10% w/v sodium acetate, and 10 mL of glacial acetic acid is then prepared. After digesting, filtering, and rinsing the precipitate, 0.2857 g of Hg2Cl2 is obtained. Report the purity of the original sample as % w/w Na3PO3.
- Aleks data for PbCO3 is 7.40 x 10^-14.Stoichiometric calculations. (show computations)K, a pharmaceutical scientist aims to synthesize paracetamol by reacting 3.075 mg of p-aminophenol and 2.25 milliliters of acetic anhydride to produce paracetamol and acetic acid. C6H7NO + C4H6O3 à C8H9NO2 + C2H4O2 Is the chemical reaction balanced? If yes, write YES. If not, what should be the balanced equation? What is the molecular weight of p-aminophenol? What is the molecular weight of acetic anhydride? What is the molecular weight of paracetamol? What is the limiting reactant? How many grams of paracetamol was formed? K was able to produce 1.88 grams of paracetamol. What is the percentage yield?Solution for 0.105g of the K3[Fe(C2O4)3] (MW= 437.2 g/ mol) were dissolved in sulfuric acid and got rated with 9 mL of 0.028M KMn04. What is the experimental weight percent of oxalate in the sample?
- A 0.9134-g of KIO3 is dissolved in 500-mL distilled water. A 50.0-mL aliquot portion was run down into an Erlenmeyer flask and 2-g of KI and 2-mL of 6M HCl were added to it. Directly after, the solution was titrated with a 24.47-mL Na2S2O3 solution to a faint yellow solution. Starch TS was added to the resulting solution and the titration with the Na2S2O3 solution continued wherein an additional 1.33-mL was required to bring the blue color of the solution to disappear completely. MW: KIO3 = 214.0 g/mol ; Na2S2O3 = 158.1 g/mol Compute the M of the Na2S2O3 solution. 0.1047 M None of the choices 0.1107 M 0.09926 MA 0.9000-g of KIO3 is dissolved in 500-mL distilled water. A 50.0-mL aliquot portion was run down into an Erlenmeyer flask and 2-g of KI and 2-mL of 6M HCl were added to it. Directly after, the solution was titrated with a 24.80-mL Na2S2O3 solution to a faint yellow solution. Starch TS was added to the resulting solution and the titration with the Na2S2O3 solution continued wherein an additional 1.33-mL was required to bring the blue color of the solution to disappear completely. MW: KIO3 = 214.0 g/mol ; Na2S2O3 = 158.1 g/mol Compute the N of the Na2S2O3 solution. None of the choices 0.09657 N 0.1075 N 0.1017 NThe mercury in a 0.8142-g sample was precipitated with an excess of paraperiodic acid, H5IO6: 5 Hg 2+ + 2 H5IO6 ---> Hg5(IO6)2 (s) + 10 H + The precipitate (MW = 1448.8 g/mol) was filtered, washed free of precipitating agent, dried, and weighed, and 0.4114 g was recovered. Calculate the a) % Hg (200.6 g/mol) b) % Hg2Cl2 (472.1 g/mol)



