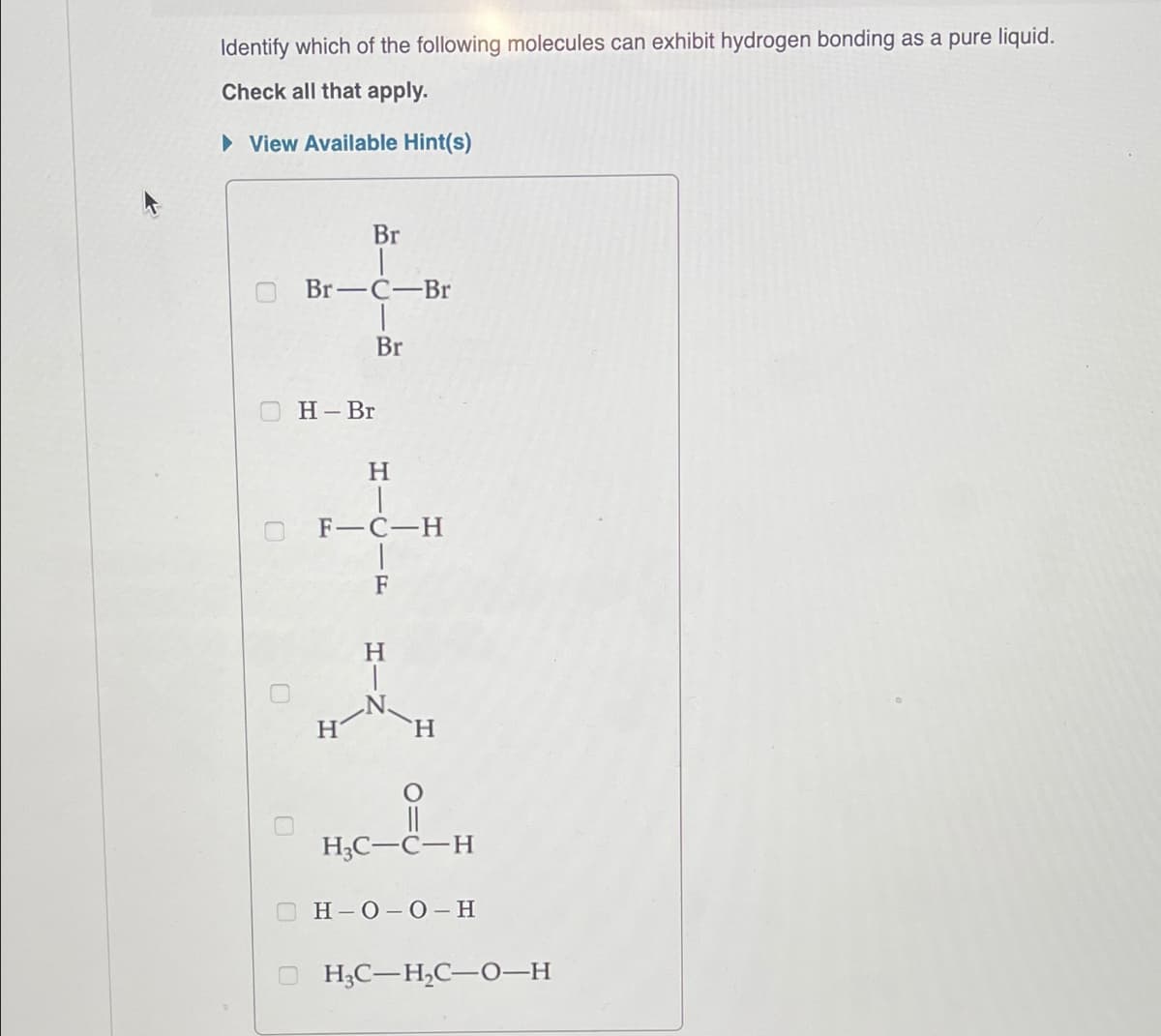
Identify which of the following molecules can exhibit hydrogen bonding as a pure liquid. Check all that apply. ▸ View Available Hint(s) Br Br-C-Br Br H-Br H F-C-H F H H H3C-C-H H-O-O-H H3C-H₂C-O-H
Q: Ceaction B. 1. HNO3, H2SO4 2. CH3CI, AICI 3 Draw the product of reaction B. NO₂
A: The objective of this question is to draw the product obtained from the given reaction.
Q: In making buffer 4, dissolve 5.04g disodium hydrogen phosphate (base) and 3.01g of potassium…
A: A buffer of disodium hydrogen phosphate (base) 3.01g of potassium dihydrogen phosphate (acid)V =…
Q: In making buffer 4, dissolve 5.04g disodium hydrogen phosphate (base) and 3.01g of potassium…
A: A buffer of disodium hydrogen phosphate (base) 3.01g of potassium dihydrogen phosphate (acid)V =…
Q: What are the products of the following hydrolysis? a primary amine and an aldehyde O a primary amine…
A: The objective of the question is to identify the products of hydrolysis when different types of…
Q: Solve question 11 and explain the mechanism if possible. Do the other questions if you can please.
A: The product of the reaction-11 is d.Explanation:
Q: An equilibrium is established for the reaction 2 CO(g) + MoO2(s) = 2 CO2(g) + Mo(s). Use the…
A: Answer: The value of Kp = 1.89 x 10-2Explanation:Step 1:Step 2:
Q: The magnesium and calcium ions present in seawater have the following concentrations: [Mg2+] = 059 M…
A: The objective of the question is to find the pH at which the selective precipitation of Mg2+ ion…
Q: Propose a chemical structure for the name below. Make sure to clearly define the stereochemistry…
A: A chemical structure is the spatial arrangement of atoms present in a molecule. It determines the…
Q: 7. Reaction Scheme. о А N 1. xs Mel, xs K2CO3 2. Ag2O, H₂O 3. heat NH2 NH2 or two differnet methods…
A: Step 12: Combine the outcomes of the reactions involving both ketone fragments to describe the final…
Q: Current Attempt in Progress × Your answer is incorrect. What is the major product for the following…
A: The reaction is completed in three steps- acid- base reaction, nucleophilic addition reaction then…
Q: A 0.170 mol quantity of NiCl, is added to a liter of a 1.20 M NH, solution. What is the…
A: The objective of this question is to find the concentration of Ni2+ ions at equilibrium when NiCl2…
Q: i need the answer quickly
A: Answer:-This question is answered by using the simple concept of naming the organic compounds using…
Q: Assignment Score: 96.7% Question 3 of 10 > 0 Atkins Jones • Laverman 22,133 C Resources Cx Give Up?…
A: The solution and answer to the given question are explained in the explanation…
Q: 4 Part B If you dilute 17.0 mL of the stock solution to a final volume of 0.300 L, what will be the…
A: Given ,
Q: 2. Draw the products formed when terpinolene, a fragrant molecule found in many cannabis strains, is…
A: The objective of the question is to identify the products.
Q: Please explain question 16 and draw out the mechanism using the correct reagent. Explain the other…
A: The correct option of question 16 is (a)Explanation are here in the picture with mechanism The…
Q: Determine how many liters 8.69 g of carbon dioxide gas would occupy at the following conditions. (a)…
A: Amount of carbon dioxide = 8.69 gUsing Ideal gas equation: PV=nRTP: Pressure in atmV: Volume in Ln =…
Q: Predicting the qualitative acid-base properties of salts Consider the following data on some weak…
A: To answer this question, you need to understand the concept of conjugate acid-base pairs.A conjugate…
Q: A particular reactant decomposes with a half-life of 117 s when its initial concentration is 0.251…
A: The objective of this question is to determine the value and units of the rate constant for a…
Q: For the reaction 2 A(g)⇋B(g)+2 C(g), a reaction vessel initially contains only A at a pressure of…
A: 760 torr ≡ 760 mm of HgSo, 1 torr ≡ 1 mm of HgKp = 3.724Explanation:Approach to solving the…
Q: Order: ABC 1.5 g. Stock: ABC 2500 mg / 2 mL. How many mL will you give? O 1 mL 0.1 mL 0.6 mL 1.2 mL
A:
Q: 8.31d KMnO4, NaOH Cold ? Modify the given carbon skeleton to draw th enantiomers. Note: you can…
A: Structure of product of the given reaction is to be determined.
Q: (a) A 25.0 mL sample of 0.175 M methylamine CH3NH2 (Kb = 1.3 x 10-5) is titrated with 0.150 M HBr.…
A:
Q: Draw the starting structure that would yield this product under these conditions. 1. Br2 (1 equiv),…
A: This is an example of Hell Volhard Zelinsky (HVZ) Reaction
Q: 8. Propose a mechanism that gives the product below. SH KO-t-Bu HO-t-Bu HO. S
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: what is the product of this reaction? + HBr
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: raw the product of this reaction. Ignore inorganic byproducts. Br2 (2 equiv) Q
A: The objective of the question is to find the product of the given reaction in which substituted…
Q: A first-order reaction has a rate constant of 0.00539 s half-life of the reactant? -1. If the…
A: Given:The reaction is of first-orderRate constant (K) = 0.00539 s-1Initial concentration = 2.50 MWe…
Q: Please explain question 15 and draw out the mechanism using the correct reagent. Explain the other…
A: The correct answer of Question 15 is :- [ Option - C ] Explanation:Option a: This option is…
Q: What is the configuration of the chiral center of the following molecule? OR ○ S Meso OH
A: According to Cahn-Ingold-Prelog rule-1) More atomic number having more priority.2) If first atom is…
Q: Identify the configuration of each chiral center in the following compound: NH₂ In the boxes below,…
A: The objective of this question is to identify the configurations in the given compound.
Q: Provide a detalied arrow pushing mechanism for the following deils alder reactions
A: The objective of this question is to provide a detailed arrow-pushing mechanism for a Diels-Alder…
Q: 3.a. Explain the following observations: (i). The bond dissociation energy of the hydrogen molecule…
A: The objective of the question is to explain various observations related to chemical bonding, states…
Q: Many Choice: How does resonance stabilize the NO3 ion? :Ö: :Ö: N [] [] O: :0 N a. Electron…
A: The objective of the question is to understand how resonance stabilizes the NO3 ion.
Q: 1. In the synthesis of isopentyl ethanoate, whose reaction is shown below, Дон OH + HO H2SO4 вл +…
A: The objective of the question is to determine the following What is the limiting reagent?What is the…
Q: Macmillan Learning Attem A sample of 69.8 g of tetraphosphorous decoxide (P4O10) reacts with 64.6 g…
A: Consider the given balanced equation is as follows;Given information:Mass of = Mass of =
Q: How many electrons, protons, and neutrons does an Iron-55 atom have? 026 electrons, 26 protons, and…
A: Given that,Atom = Atomic number of Number of elctrons =?Number of protons=?Number of neutrons=?
Q: References Use the References to access important values if needed for this question. The Solubility…
A: The objective of this question is to find the concentration of chromate ion required to just…
Q: What is the predicted major product for the reaction shown? OH CI OH Cl2/AICI 3
A: The substituents present on the benzene are opposing groups i.e., one group (OH) is ortho and para…
Q: 4 Part A Ammonia is an important chemical used in the production of fertilizer. Industrial…
A: The enthalpy change of a chemical reaction can be calculated by using the bond energies. The…
Q: 5) Predict the product (only one observed) and provide a step by step mechanism of the following…
A: Since alpha hydrogen is present at ortho of the halogen group, therefore, the reaction will proceed…
Q: What is the solubility of Ca3(PO4)2 Ksp = 1.3 x 10-32 in a 0.048 M Ca(NO3)2 solution? 5.4 x 10-15M ,…
A: The objective of this question is to find the solubility of Ca3(PO4)2 in a 0.048 M Ca(NO3)2 solution…
Q: The energies, E, for the first few states of an unknown element are shown here in arbitrary units.…
A: Electrons in a given energy state will absorb energy and transfer to a higher energy level. The…
Q: x When 25.0 mL of a 8.46 × 10-4 M aluminum iodide solution is combined with 15.0 mL of a 4.67 × 10-4…
A: The objective of the question is to determine whether a precipitate forms when a solution of…
Q: What is the pOH of a 0.50 M solution of polyprotic acid H3X with K1 = 2.5 x 10-4, K2 = 5.6 x 10-8,…
A: Given,Molarity of triprotic acid ( H3A ) = 0.50 MK1 = 2.5 x 10-4, K2 = 5.6 x 10-8 and K3 = 3.0 x…
Q: How many stereogenic centers are present in compound? хво CO₂H
A: ->Stereogenic centers, also known as stereocenters or chiral centers, are atoms in a molecule…
Q: The reduction is..... ............ of loss of electrons O gain, electrons loss, protons Ogain,…
A: Given that,The reduction is _ _ _ __ of _ _ _ __ _ _.Choose the correct option.
Q: What is the pH of a 1.6 x 10-7 M solution of Ca(OH)2? X Selected Answer: C. 7.51 Answers: A. 6.80 B.…
A: The objective of this question is to calculate the pH of a 1.6 x 10^-7 M solution of Ca(OH)2. The pH…
Q: How many steps are in this reaction? Energy 1 2 3 4 + HC H Reaction coordinate H CI 7
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: What is the product of the following sequence of reactions? NaOH NaNH2 H3O+ NH2 IV ||
A: An E2 elimination takes place where a chloroalkane is transformed into alkene. At first the Acidic…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- The following information is given for n-pentane at 1atm: boilingpoint=36.20CHvap(36.20C)=357.6J/g meltingpoint=129.7CHfus(129.7C)=116.7J/g specificheatgas=1.650J/gC specificheatliquid=2.280J/gC A 26.10-g sample of liquid n-pentane is initially at 51.40C. If the sample is heated at constant pressure (P=1atm), how many kJ of energy are needed to raise the temperature of the sample to 65.30C?Which intermolecular force(s) would you predict are present in a pure liquid sample of the molecule shown below? Select all that apply. -London Dispersion Interaction -Ion-dipole Interaction -H-bonding -Dipole-Dipole InteractionCooing Curve of Water i) What phase(s) at each of the numbered sections?ii) At what temperature is this substance a) condensing and b) freezing?iii) At which number section(s) is/are kinetic energy of the molecule is the greatest?iv) Relate the answer at (iii) to the associated intermolecular force of the molecules.
- ANSWER THE QUESTIONs IN NOT LESS THAN THREE (3) SENTENCES AND NOT MORE THAN FIVE (5) SENTENCES NO PLAGIARISM PLEASE :) 3. WHICH ASSUMPTIONS OF THE KINETIC MOLECULAR THEORY FOR GASES ARE MODIFIED TO EXPLAIN THE SHAPE AND VOLUME OF LIQUIDS AND SOLIDS? 4. HOW DO INTERMOLECULAR FORCES RELATE WITH THE ASSUMPTIONS OF THE KINETIC MOLECULAR THEORY?Answer the following situation about intermolecular forces ( The situation in general is all about Kinetic Molecular Theory ) Situation: A girl brush a strip of water on the clean sheet of bond paper and she do it again the same procedure using shampoo and alcohol. She have observed that alcohol evaporate first , second is the shampoo and last is the water. Question:Why do you think alcohol evaporate first, shampoo evaporate second and water evaporates last? Explain your answer (state your observations).In which of the following substances would you expect to find hydrogen bonding? Explain your reasons.
- A. DIRECTION: Define the following and give an example of each:1. Dispersion force2. Dipole-dipole attraction3. Hydrogen bond B. DIRECTION: Open the PhET States of Matter Simulation to answerthe following questions: Link: https://phet.colorado.edu/en/simulation/states-of-matter 1. Select the Solid, Liquid, Gas tab. Explore by selecting differentsubstances, heating and cooling the systems, and changing thestate. What similarities do you notice between the foursubstances for each phase (solid, liquid, gas)? What differencesdo you notice? 2. For each substance, select each of the states and record the giventemperatures. How do the given temperatures for each statecorrelate with the strengths of their intermolecular attractions?Explain.ANSWER THE QUESTIONs IN NOT LESS THAN THREE (3) SENTENCES AND NOT MORE THAN FIVE (5) SENTENCES NO PLAGIARISM PLEASE :) 1. HOW DO LIQUIDS AND SOLIDS COMPARE WITH GASES IN TERMS OF ATTRACTIVE FORCES? 2. WHICH ASSUMPTIONS OF THE KINETIC MOLECULAR THEORY FOR GASES ARE MODIFIED TO EXPLAIN THE SHAPE AND VOLUME OF LIQUIDS AND SOLIDS? 3. HOW DO INTERMOLECULAR FORCES RELATE WITH THE ASSUMPTIONS OF THE KINETIC MOLECULAR THEORY?The critical point coordinates of ethanol (46.068 g mol-1, d = 0.79 g mL-1) are 514 K and 62.18 atm, the normal boiling point is known as 78.4 °C. 2,442 mL of ethanol is placed in an 8.6 L beaker at 24 °C and allowed to equilibrate. What is the mass percent of liquid ethanol remaining relative to the initial total amount in the container?
- Rank for the following liquids in order of increasing viscosity: lowest first. I. CH3CH3NH2 II. CH3CH2CH2CH3. III. CH3CH2CNIn the previous two questions, the actual pressure is less than the ideal pressure. Which false assumption in the kinetic molecular theory of gases has more effect on this error? (Note - the actual pressure isn't always less than the ideal pressure - it depends on the conditions. In this set of examples, however, the conditions give Preal<Pideal.) a. Gas molecules are in constant random motion b. nothing can ever be truly ideal c. Intermolecular forces d. molecular size -------------------------------------------------------------------------- Previous Questions: 1) Assuming ideal behaviour, calculate the pressure in atmospheres of 2.19 kilograms of hydrogen chloride gas (HCl(g) - yes, hydrochloric acid is also called hydrogen chloride, and it's a gas at room temperature. It's "liquid" when you use it in the lab because you use it dissolved in water!) in a 25.0 L container at 18°C. 2) Given the Van der Waals constants for hydrogen chloride, below, calculate the pressure in…The temperature inside a pressure cooker is 115 C. Calculate the vapor pressure of water inside the pressure coker. What would be the temperature inside the pressure cooker if the vapor pressure of water was 3.50 atm? Can you plz explain using a simple manner, Im horrible at math also use the clausius i provided below.

