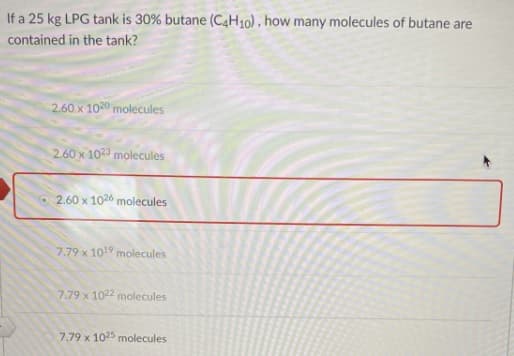
If a 25 kg LPG tank is 30% butane (C4H10) , how many molecules of butane are contained in the tank? 2.60 x 1020 molecules 2.60 x 102 molecules 2.60 x 1026 molecules 7.79 x 1019 molecules 7.79 x 1022 molecules 7.79 x 1025 molecules
Q: the mass of 1 gallon of gasoline is about 2.7 kg. Assuming the gasoline is entirely octane, C8H18,…
A: Given The mass of 1 gallon of gasoline is about 2.7 kg or 2700 grams and the gasoline is entirely…
Q: Equivalent weight of Al(OH)3 is а. 39 g O b. 27 g O c. 78 g O d. 26 g
A: Option d) 26 g
Q: How many grams of rhenium are in a 88.24-gram sample of ReCl 3? The molar mass of ReCl 3 is 292.57…
A:
Q: A 2.00 g sample of an organic compound contains 0.675 g of carbon. How much carbon would a 4.00 g…
A: ◼️ Given that: 2.00 g of organic sample has 0.675 g Carbon Carbon in 4.00 g of same organic…
Q: What is the product of each of the following reaction sequences?
A: Active methylene are those compounds that have a -CH2 group sandwiched between two carbonyl…
Q: How many MOLECULES of water are present in 9.27 grams of this compound? 2.80 x 1032 molecules H2O…
A:
Q: What is the number of molecules in 319g of Li2SO4? (Take NA = 6
A: Mass of Li2SO4 = 319 g NA = 6 x 1023 . Number of Molecules = ?
Q: Calculate the mass (in kilograms) of chlorine in 25 kg of each chlorofluorocarbon (CFC).a. CF2Cl2 b.…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Use the molar mass of Ca3(PO3)2 to calculate the moles of oxygen in 6.66 x 106 g of CP. Give your…
A: given mass of Ca3(PO3)2 = 6.66 x 106 g Molar mass of Ca = 40.078 g/mol Molar mass of P = 30.974…
Q: What is the number of molecules in 256g of Li3PO4? OA 1.46 x 10-24 O B. 1.46 x 1023 OC. 1.46 x 10-23…
A: 1 mole of compound contain 6.022×1023 molecules . 1 mole = 6.022×1023 molecules To find the number…
Q: What is the mass of 2.11x1024 molecules of C2H6?
A: 1 mole of C2H6 contains 6.023*1023 molecules. Number of molecules present in C2H6 2.11*1024…
Q: Calculate the mass of C in 5.46 g of C2H4? 4.68 g C 13.1 g C O 65.6 g C O 0.455 g C
A:
Q: What is the molar mass of the following compounds? N2H4 b.…
A: The molar mass of a substance can be defined as the mass of that substance present in one mole. It…
Q: What is the number of molecules in 319g of Li2SO4? (Take NA = 6 x1023)
A: The question is based on mole concept. We have to determine number of molecules present in the…
Q: How many molecules of ascorbic acid (vitamin C: C6H8O6)are in a 50 mg tablet? (need molar mass)
A: Given data contains, Mass of tablet is 50 mg. The molar mass of ascorbic acid (C6H8O6) is calculated…
Q: How many individual formaldehyde (CH20 = 30.03 g/mol) are found in a 32.0 mL sample %3D of gaseous…
A: Data given, Volume of formaldehyde = 32.0 mL The density of formaldehyde = 0.815 g/mL Molar mass of…
Q: 17) How many molecules of water are present in 54.7g of water? 5.94 x 10 26 molecules 1.98 x 10 23…
A: Since number of molecules = number of moles X Avogadro's number And number of moles = mass / molar…
Q: Use the following chemical equation C6H12 + 9 O2 ----> 6 CO2 + 6 H2O (OR C6H12 + 9 O2 right arrow…
A: Given reaction: C6H12 + 9 O2 → 6 CO2 + 6 H2O We have to calculate the mass of CO2 that can be…
Q: 45. What volume of 3.0 M NaOH can be prepared using exactly 96.0 g of NaOH? А. 0.14 L В. 0.70 L 0.80…
A: Given data : 1. Concentration of NaOH = 3.0 M 2. Mass of NaOH = 96.0 g NaOH To calculate : Volume…
Q: How many molecules are present in 300.0 g of benzaldehyde (C6H5CHO)? 2.836 x 1023 molecules 6.023 x…
A: Molar mass of Benzaldehyde = 106.12 g/mol
Q: How many moles is each of the following? 15.5 g SiO2 0.0688 g AgCl 5.96 g KOH 937 g Ca(C2H3O2)2
A: Number of moles = mass molar mass
Q: What is the mass of 2.2 x 10 ° CO 2 molecules? O a. 1.0 x 10-12 g O b.1.6 x 10-13 g O. 12x 106 g O…
A:
Q: The density of water is 1.00 g/mL at 4°C.How many water molecules are present in 2.36 mL of water at…
A: given density of water = 1 g/ml
Q: Calculate the mass in grams of 1.31×1024 molecules of propylamine. The chemical formula for…
A: Mole : It is the unit of measurement of the amount of substance. It's SI unit is mol. One mole of…
Q: The number of moles of 3.06 X 10–3 g of C2H5NO2 is 0.0408 moles.
A: In international system of units, Mole is considered as a unit which helps to calculate the amount…
Q: Equivalent weight of Al(OH)3 is а. 26 g O b. 27 g С. 78 д O d. 39 g
A:
Q: The density of octane, C8H18C8H18, is 0.703 g/mL0.703 g/mL. Calculate the number of moles of octane…
A: Given Data : 1.density of octane = 0.703 g/mL 2. Volume = 49.1 mL Molar mass of octane = 114.23…
Q: What number should be placed in the blank in order for this equation to follow the Law of…
A:
Q: The number of grams of 1.38 x 1022 molecules of C12H22011 is: 0.0229 g 7.84 g 342 g 2.29 g
A: Avogadro's number gives the number of atoms, molecules or ions in one mole of the substance and it…
Q: An empty propane tank weighs 7.22 kg. When it is filled it weighs 25.20 kg. How many propane…
A: To find the number of molecules of propane.
Q: How many carbon atoms are in one molecule of Li2CO3? 6 atoms 5 atoms 1 atom 3 atoms 6.02…
A: The molecular formula of the lithium carbonate is = Li2CO3 The number of carbon atoms in one…
Q: How many molecules of H2O would be found in 54.05 grams of water? choices: 3.252 x 1025 molecules…
A:
Q: How many grams of NO2 is required to produce 61.5 grams of NO? 1. 13.4 g O 2. 283 g 3. 120 g 3NO2(g)…
A:
Q: How many MOLECULES of water are present in 9.27 grams of this compound? 3.10 x 1023 molecules H2O…
A: Note : 1 mole compound contains 6.023 x1023 molecules. 1 mole = 18 grams water. 18 gr water contains…
Q: C6H12 + 9 O2 ----> 6 CO2 + 6 H2O (OR C6H12 + 9 O2 right arrow 6 CO2 + 6 H2O) How many grams of CO2…
A: Given reaction is, C6H12 + 9 O2 ----> 6 CO2 + 6 H2O Other data, The molecular weight of water is…
Q: The relative mass of propylene (C₃H₆) is 42.1 g/mol. How many carbon atoms does a polypropylene…
A: Given data is: Molar mass of polypropylene = 42.1 kg/mol 42.1kg1 mol×1000g1kg=42100g/mol formula of…
Q: How many molecules of carbon dioxide are in 12 1 g? (Avogadro's number = 6.022 x 1023) %3D O 8.84 x…
A: Molar mass of CO2 is 44g/mol In molar mass of CO2, there are avogadro no. of CO2 molecules ,i.e.,…
Q: 3. H30 + Y 国 2. HO O
A: The given reaction is an example of a directed aldol condensation reaction.
Q: Considering the chemical equation :below C2H3B13 + 11 O2 → 8 CO2 + 6 H20 + 4 6 Br2 What is the…
A: The balanced reaction taking place is given as, => 4 C2H3Br3 + 11 O2 -----> 8 CO2 + 6 H2O + 6…
Q: Gibberellic acid, C19H22O6, is used to promote plant growth, particularly in seedlings. How many…
A: Avogadro's number gives the number of atoms, molecules or ions in one mole of the substance and it…
Q: A bottle of Ibuprofen (C 13 H 18 O 2 ) contains 1.204 * 10 ^ 24 molecules. How many grams of…
A: Interpretation - To determine the grams of ibuprofen are contained in the bottle when a bottle of…
Q: 1 ед. NaBH4 methanol 1) excess LIAIH4 OH 2) H20 HO 1) еxcess MgBr 2) H3O* Ph3P=
A:
Q: An empty propane tank weighs 7.22 kg. When it is filled, the tank weighs 25.20 kg How many propane…
A:
Q: How many atoms are contained in 12 g of water? O 2.4 x 1024 O 1.2 x 1024 4.0 x 1023 O 1.3 x 1023
A: To calculate the number of atoms in 12 g of H2O Molar mass of H2O = 18 g/mole Now we calculate moles…
Q: 13) If a 25 kg LPG tank is 30% butane (C4H10) , how many molecules of butane are contained in the…
A:
Q: Calculate the number of molecules in 17.8 grams of Carbon Dioxide (CO2)? O 2.11 x 1023 molecules O…
A: Given that, Mass of CO2 = 17.8 To find, the number of molecules in 17.8 g CO2?
Q: 1. Hg(OAc), "2O 2. NaBH4 3. PB13 4. Mg/ether
A: alcohol is the product.
Q: How many MOLECULES of water are present in 9.27 grams of this compound? A 7.10 x 1023 molecules H20…
A:
Q: What is the mass of a drop of methanol containing 2.90x1022 CH3OH molecules
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Soda ash (sodium carbonate) is widely used in the manufacture of glass. Prior to the environmental movement much of it was produced by the following reaction. CaCO3 + 2 NaCl → Na2CO3 + CaCl2 Unfortunately, the byproduct calcium chloride is of little use and was dumped into rivers, creating a pollution problem. As a result of the environmental movement, all of these plants closed. Assume that 125g of calcium carbonate (100.09 g/mol) and 125 g of sodium chloride (58.44 g/mol) are allowed to react. Determine how many grams of useful sodium carbonate (105.99 g/mol) will be produced. How many grams of useless calcium chloride (110.98 g/mol) will be produced? You should also determine how many grams of excess reactants are left (indicate which one is the limiting reactant)The combustion of gasoline produces carbon dioxide and water. Assume gasoline to be pure octane (C8H18) and calculate how many kilograms of carbon dioxide are added to the atmosphere per 7.8kg of octane burned.A chemist adds 70.0mL of a 2.40 x 10^−4 mol/L copper(II) fluoride (CuF2) solution to a reaction flask. Calculate the micromoles of copper(II) fluoride the chemist has added to the flask. Be sure your answer has the correct number of significant digits.
- What is the molar volume of solid quartz, SiO2?(density =2.65g/cm3) (molar volume is the volume occupied by one mole)On January 28, 2014, in Germany, a barn containing 90 cows exploded when a stray spark caught the methane trapped in the building on fire. The building was damaged and one cow was slightly injured with minor burns. Assume that the 90 cows were producing 250L of methane per day and that there was a 15 hour collection in the barn when it ignited. __CH4 + __O2 → __ CO2 + __H2O a) How many liters of methane was in the barn when it exploded? b) What mass of water was generated in the explosion? (SET WORK INTO TRAIN TRACKS)20 cm3 of a gaseous hydrocarbon required 90 cm3 of oxygen for complete combustion. Both volumes were measured under the same conditions.The hydrocarbon must be?
- Solid ammonium chloride, NH4Cl,NH4Cl, is formed by the reaction of gaseous ammonia, NH3,NH3, and hydrogen chloride, HCl.HCl. NH3(g)+HCl(g)⟶NH4Cl(s) NH3(g)+HCl(g)⟶NH4Cl(s) A 4.18 g4.18 g sample of NH3NH3 gas and a 4.18 g4.18 g sample of HClHCl gas are mixed in a 0.50 L0.50 L flask at 25 ∘C.25 ∘C. Identify the limiting reagent. How many grams of NH4Cl will be formed by this reaction? What is the pressure in atmospheres of the gas remaining in the flask? Ignore the volume of solid NH4Cl produced by the reaction.How many grams of water would be formed from 96.0 g NH3 in a reaction represented by the balanced equation below. Show how to set up the calculation using dimensional analysis by selecting the correct factors from the menu. Then select the correct answer from the answer menu. 4 NH3(g) + 3 O2(g) →→ 2 N2(g) + 6 H2O(l)Draw 6 water molecules in each beaker below. On the left should be 6 liquid water molecules. On the right draw the gas particles that are present after evaporation, but before any substances escape the beaker.
- As part of the aspirin synthesis lab the orgo students also had to perform the following calculation to demonstrate their knowledge. Are you able to help them work this out? Saponification is a process in which soap is produced from the chemical reaction between animal fat (triglycerides) and a strong base such as KOH. An example of such balanced chemical reaction is shown here: C51H98O6 + 3KOH → C3H5(OH)3 + 3C16H31O2K (Triglyceride) (Soap) if during the saponification reaction 231.5 g of C51H98O6 is mixed with 231.5 g of KOH and 160 g of soap is produced. Calculate the theoretical yield of soap C16H31O2K and indicate who is the limiting reactant. (Provide your answer to 2 decimal places) Calculate the percent yield for this reaction (Provide your answer to 1 decimal place) *Show ALL steps and mathematical equations involved in your calculations to help the orgo students. Remember to label all…The U.S. proven natural gas reserves in 2013 were 323 trillion ft3 . How long will these reserves last if there are no imports or exports and if the U.S. annual rate of use of 24.5 trillion ft3 continues?A website promoting the use of alternative energy vehicles and hybrid technologies claims that, "A typical automobile in the USA uses about 4040 gallons of gasoline every month, producing about 750 lbs of carbon dioxide." To determine the truth of this statement, calculate how many pounds of carbon dioxide are produced when 40.0040.00 gallons of gasoline are subjected to complete combustion. Assume that the primary ingredient in gasoline is octane, C8H18(l),C8H18(l), which has a density of 0.703 g/mL.

