If the initial sample is 1.25 g, what mass of the original iridium‑192 remains after 65 days? mass remaining: 0.675 g (this is correct, I just need answers to the last two questions) Estimate the half‑life of the radioisotope. half-life: days How many days would it take for one‑third of the sample to decay? time:
If the initial sample is 1.25 g, what mass of the original iridium‑192 remains after 65 days? mass remaining: 0.675 g (this is correct, I just need answers to the last two questions) Estimate the half‑life of the radioisotope. half-life: days How many days would it take for one‑third of the sample to decay? time:
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter14: Nuclear Chemistry
Section: Chapter Questions
Problem 14.104PAE
Related questions
Question
If the initial sample is 1.25 g, what mass of the original iridium‑192 remains after 65 days?
mass remaining:
0.675 g (this is correct, I just need answers to the last two questions)
Estimate the half‑life of the radioisotope.
half-life: days
How many days would it take for one‑third of the sample to decay?
time:
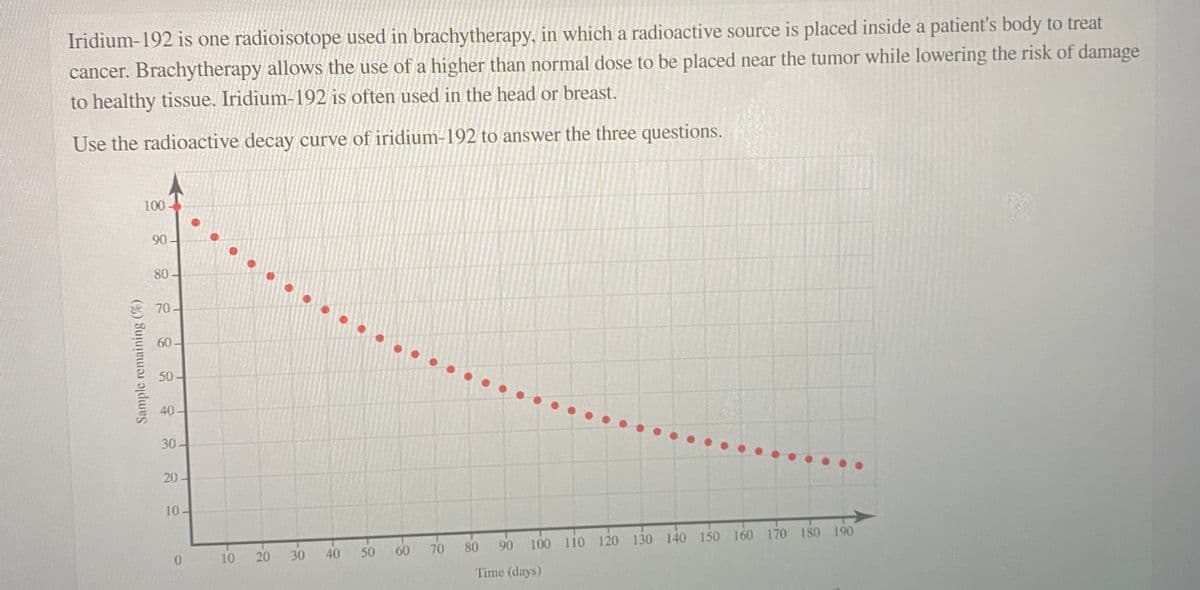
Transcribed Image Text:Iridium-192 is one radioisotope used in brachytherapy, in which a radioactive source is placed inside a patient's body to treat
cancer. Brachytherapy allows the use of a higher than normal dose to be placed near the tumor while lowering the risk of damage
to healthy tissue. Iridium-192 is often used in the head or breast.
Use the radioactive decay curve of iridium-192 to answer the three questions.
Sample remaining (%)
100
90
80
70
60
50
40
30-
20
10.
10
20 30
40 50
60
70
90 100 110 120 130 140 150 160 170 180 190
Time (days)
80
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning