If the reaction to form cyanohydrins from substituted benzaldehydes exhibits a Hammett P value of +2.33. Which is the correct ranking (1 is the slowest. 4 is the fastest) for the rates of cyanohydrin formation of the following aldehydes (Substituent constants, 6, for each aldehyde are given in parentheses). ? (+0.34) (4) F (+0.10) Me O О Correct ranking i is # H Correct ranking is # F3C (+0.53) H Correct ranking is # О MeO. (-0.12) H Correct ranking is #
If the reaction to form cyanohydrins from substituted benzaldehydes exhibits a Hammett P value of +2.33. Which is the correct ranking (1 is the slowest. 4 is the fastest) for the rates of cyanohydrin formation of the following aldehydes (Substituent constants, 6, for each aldehyde are given in parentheses). ? (+0.34) (4) F (+0.10) Me O О Correct ranking i is # H Correct ranking is # F3C (+0.53) H Correct ranking is # О MeO. (-0.12) H Correct ranking is #
Chapter24: Amines And Heterocycles
Section24.9: Heterocyclic Amines
Problem 24P
Related questions
Question
Please help with this
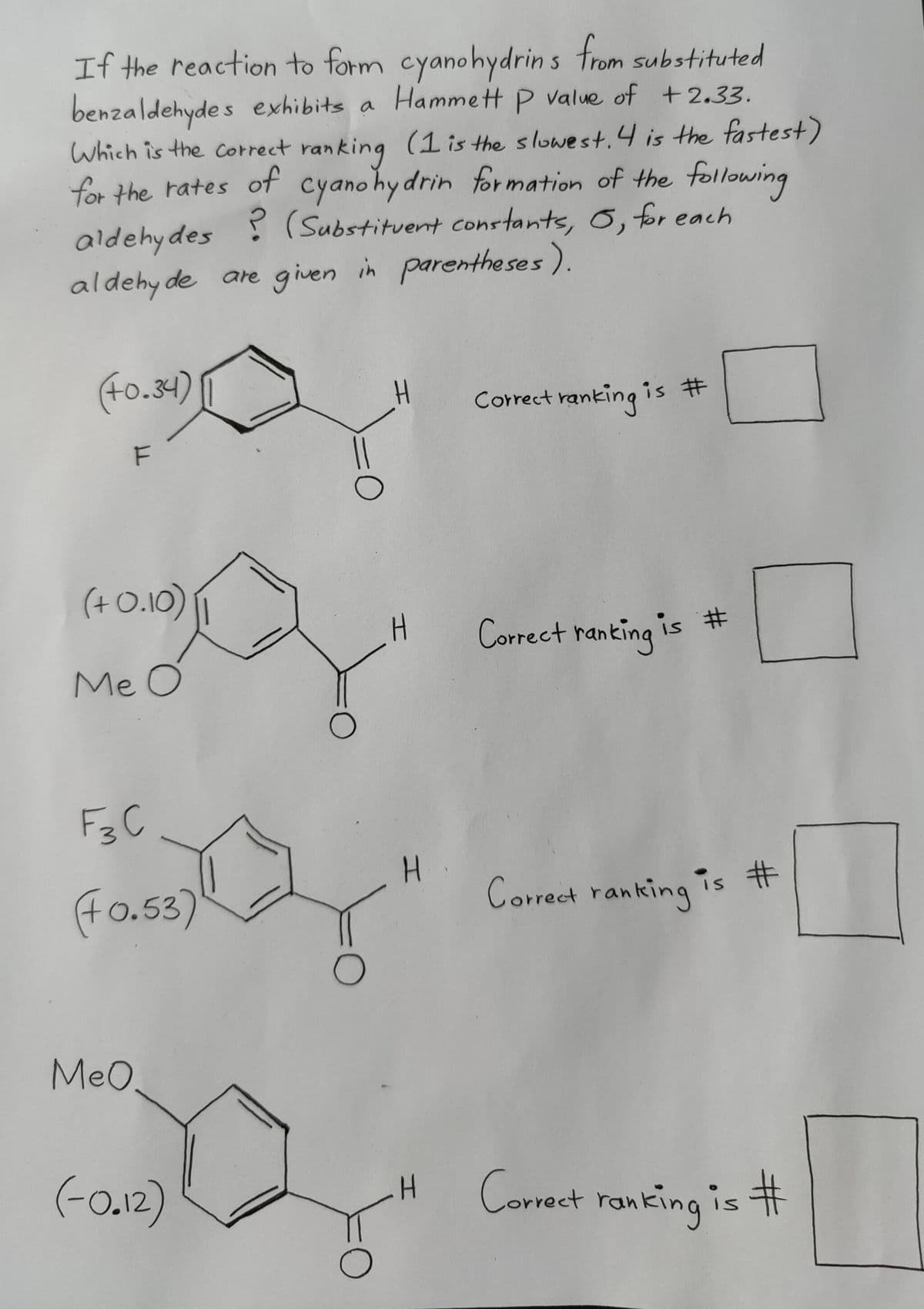
Transcribed Image Text:If the reaction to form cyanohydrins from substituted
benzaldehydes exhibits a Hammett P value of +2.33.
Which is the correct ranking (1 is the slowest. 4 is the fastest)
for the rates of cyanohydrin formation of the following
aldehydes (Substituent constants, 6, for each
aldehyde are given in parentheses).
?
(+0.34)
(4)
F
(+0.10)
Me O
О
Correct ranking i
is #
H
Correct ranking is #
F3C
(+0.53)
H
Correct ranking is #
О
MeO.
(-0.12)
H
Correct ranking is #
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps

Recommended textbooks for you



