If the standard reference half-cell used to construct a table of electrode potentials was Zn²+ (aq) + 2e - Zn(s), the standard electrode potential for Fe2(aq) + 2e - Fe(s) would be Select one: O +0.31V O-0.45V O +0.45V O-0.31V If the standard reference half-cell used to construct a table of electrode potentials was Zn² (aq) + 2e- Zn(s), the cell potential for a Fe(s) | Fe²(aq) || Cu²(aq) | Cu(s) cell would be Select one: O-0.79V O-1.55V O +1.55V O +0.79V
If the standard reference half-cell used to construct a table of electrode potentials was Zn²+ (aq) + 2e - Zn(s), the standard electrode potential for Fe2(aq) + 2e - Fe(s) would be Select one: O +0.31V O-0.45V O +0.45V O-0.31V If the standard reference half-cell used to construct a table of electrode potentials was Zn² (aq) + 2e- Zn(s), the cell potential for a Fe(s) | Fe²(aq) || Cu²(aq) | Cu(s) cell would be Select one: O-0.79V O-1.55V O +1.55V O +0.79V
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter32: Voltaic Cell Measurements
Section: Chapter Questions
Problem 2ASA
Related questions
Question
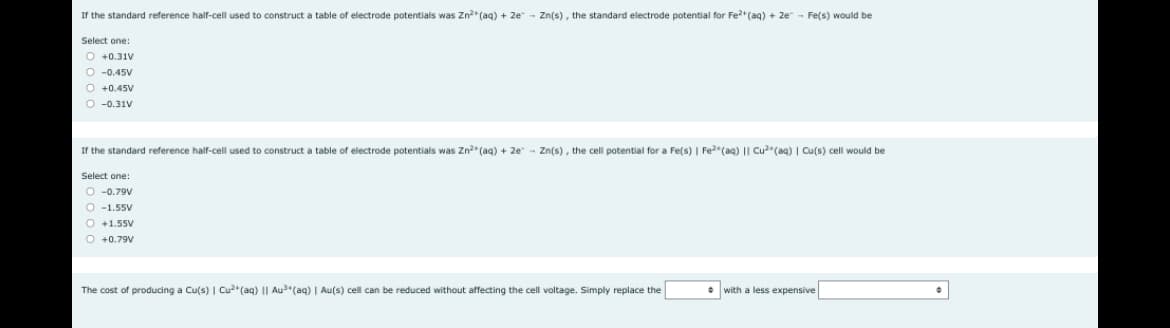
Transcribed Image Text:If the standard reference half-cell used construct a table of electrode potentials was Zn²+ (aq) + 2e - Zn(s), the standard electrode potential for Fe²+ (aq) + 2e - Fe(s) would be
Select one:
O +0.31V
O -0.45V
O +0.45V
O -0.31V
If the standard reference half-cell used construct a table - electrode potentials was Zn²+ (aq) + 2e Zn(s), the cell potential for a Fe(s) | Fe²(aq) || Cu²(aq) | Cu(s) cell would be
Select one:
O-0.79V
O-1.55V
O +1.55V
O +0.79V
The cost of producing a Cu(s) | Cu2+ (aq) Au³+ (aq) | Au(s) cell can be reduced without affecting the cell voltage. Simply replace the
with a less expensive
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning