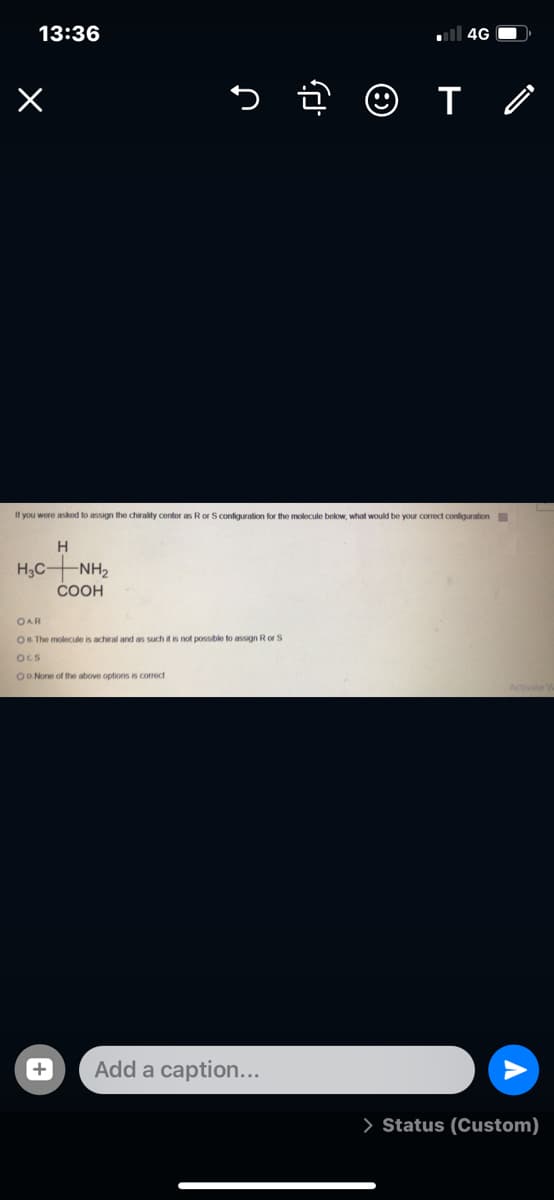
If you wore asked to assign the chirality centor as Ror S configuralion for the molecule below, what would be your correct conliguration H3C NH2 COOH
Q: Determine the number of chiral carbon atoms in the follow- ing compound, and then calculate the…
A:
Q: How many chirality centers are there in the following molecule? H;C CH3 CH3 O1 2 3 4 O 5
A:
Q: Indicate whether the following molecule is chiral or achiral. но- CH3
A: Achiral
Q: How many asymmetric centers does 2,4,6 tnmethy iheptane have
A: Asymmetric centres
Q: 3. Identify all stereocenters in the following molecules as R or S. Q **|| NH₂ H buted
A: According to guidelines I am supposed to answer one question only.
Q: Which of the following is considered as chiral molecule? Br CH3 H3C CH3 it А. C. Br- H3C CI Only B…
A: Chirality: A carbon that is attached to four different groups
Q: 11) Label all chiral centers in the following molecule as R or S F Br
A:
Q: .Which of the following molecules would be chiral (has a mirror image) c) a) H. b) CH3 он d) OH CI…
A:
Q: Q3.ldentify all the chiral centers in each molecule and determine the absolute configuration as R or…
A: Since you have asked multiparts we will solve the first threesubparts for you.
Q: Draw the optical isomers of CH3-CHOH-CHOH-CH3 . INDICATE the absolute configuration (R/S) of each…
A:
Q: 4. Identify the chiral centers in the following molecule and label each as R or S. HO Il .
A: The chiral centers are the carbon atoms having four distinct groups attached to it. The priority of…
Q: In the Fischer projection below, what are the configurations of the two asymmetric centers? H2CH HO…
A: Rules for assigning R and S notation to fischer projection. • Assign priorities to the atoms/groups…
Q: I. Given the following compounds: CI а. НО-СН2 — CH— СH b. CH3- CH2 – CH OH į. i. Determine and give…
A: Chiral Carbon do not have any symmetry.
Q: SH OH H3C CH3 ČH2OH -CECH ČH3 E HOOC D
A: R/S nomenclature is based on CIP rule which states that higher is the atomic number of directly…
Q: Q3.ldentify all the chiral centers in each molecule and determine the absolute configuration as R or…
A: Atoms in circle are phantom atoms.
Q: Identify each of the following compounds as either chiral or achiral. If the molecule is chiral,…
A: A molecule is chiral if it cannot be superposed on its mirror image by any rotations. A chiral…
Q: OH HN. Br
A: Here we have to say the molecules are chiral or achiral-
Q: Q5/ Assign Ror S configuration to the chirality centers in the following molecules: (a) H OH Ib) H…
A: The configuration of the chiral molecule is given by projecting the wedge projection into Fischer…
Q: Wat are the four alcohols with the molecular formula C4H9OH. How many of these are chiral?
A: Four possible alcohols with the molecular formula C4H9OH are: Butan-1-ol Butan-2-ol Butan-2-ol…
Q: Br C -CH3 CH3
A:
Q: 5. Draw the optical isomers of CH3-CHOH-CHOH-CHO. Indicate the absolute configuration of each chiral…
A: Draw optical isomers of the given compound and identify which is enantiomer and which is…
Q: CH3 a) H3CBr H F CH3 b) н c) H- CI Br CI CH3 CHI5
A: Enantiomers of a compound are the stereoisomers which are non-super imposable mirror images. A…
Q: Which of the following molecules are ACHIRAL (i.e. NOT chiral)? (can choose more than 1) OH F OH B…
A:
Q: Two structures are shown. Identify the asymmetric carbons in each structure, classify as chiral or…
A: 2nd is the image that showsasymmetri carbon in compound A. COMPOUND A IS ASSYMETRIC .THERE ARE ONE…
Q: 4. Label the chiral centers in the following molecules with an asterisk (*
A:
Q: Two structures are shown. Identify the asymmetric carbons in each structure, classify as chiral or…
A: Chiral compounds are those which doesn't possess the symmetry elements.
Q: Specify the configurations (R or S) of chiral centers a and b in the chem3D structure below. F a…
A: Rules for R and S configuration are as follows: The priority of each group to the stereo center…
Q: 5.48 Draw all possible configurational isomers of the molecule shown here. of the following species…
A: The IUPAC NAME FOR THE GIVEN COMPOUND IS., 2,3-dibromo-4,5-dichloroheptane
Q: 1. Locate/Label the chirality center of the given molecules. a. OH H2N C H CH3 CH3 SH
A: Isomers are the ones that have the same molecular formula but the different structures of atoms in a…
Q: Which is the correct assignment of chirality at C2 and C4 of the following molecule? HO a. 2S,4S b.…
A:
Q: Q.3 For the following molecule, draw all stereoisomers and identify all pairs of enantiomers and…
A: Enantiomers Enantiomers are stereo isomers that are non- super imposable on its mirror images. They…
Q: Answer questions for the following molecule: Circle any/all chiral centers. b. Is this molecule…
A: A chiral centre is the atom bonded to four different groups in such a way that it has a…
Q: 5. Each molecule below has CH3 но, a chiral carbon. Determine the absolute configuration (R/S) for…
A: Chirality: The geometric property of molecules where the structure of the molecule and its mirror…
Q: (a) ÇI (b) (c) (d) NO `NH2 (e) (f) (g) CH3 Br H CH3 (h) LCH3 H;C H,C `H. 5.39 For each molecule in…
A: The chiral centres in the molecules and the meso compounds has to be identified
Q: Determine whether each molecule shown below is chiral or achiral (not chiral). Br CI CH3 H,C-CH H,C…
A:
Q: 2. For the following pairs of molecules, determine if their relationship is enantiomers,…
A:
Q: 1. Identify as chiral or achiral. но. н. он он он Он
A: Chirality is determined on the basis of the fact that all four functional groups or atoms present…
Q: How many chirality centers are there in the following molecule? CH3 H;C O 2
A: Chirality center is carbon atom having four different groups attached. It is denoted by (*)
Q: Which is the correct assignment of chirality at C2 and C4 of the following molecule? OH 4 Но Н a.…
A:
Q: Duestion 3 For the lompound Name the compound Identify each chiral center Are goometric isomors…
A: The chiral center is the carbon which is attached to four different groups.
Q: Q3. (A) Identify the following compounds as chiral or achiral.
A: Identify the compounds as chiral or achiral.
Q: How many chirality centers are in the molecule shown below? CH;CHCNH NH2 CH; ce ph ale xin CO,H 6.…
A: We have to identify different chiral centre present in the given molecule.
Q: Draw a structural formula of the SS configuration of the compound shown below. CH2F HO2C • Use the…
A: Given- There must be S configuration at chiral center.
Q: '. How many chirality center/s are in the molecule shown below? Show them Br H;C-CH,CH(CH3)CHCH3
A:
Q: Determine whether three molecules—H2O, CH2BrCl, and CHBrClF—aresuperimposable on their mirror…
A: Chirality can be defined as the property where the compound forms the non-superimposable mirror…
Q: a) Determine the absolute configuration of the chiral carbons in the following molecule: CHO H- HO-…
A: Assignment of R/S configuration is based on the CIP rule of Priority. In fischer projection given…
Q: Q1. Draw the enantiomers for the following molecules and assign all chiral centres as R or S:…
A: Hello. Since multiple questions have been posted, the first question shall only be solved in this…
Q: Q3.ldentify all the chiral centers in each molecule and determine the absolute configuration as R or…
A:
Q: How many asymmetric/chiral centers are found Butaclamol? This drug is a potent antipsychotic and has…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- with a model of methane (CH4) can the mirror image be superimposed on the original (in regard to Chirality)Most naturally occurring amino acids have chiral centers (the asymmetric a carbon atoms)that are named (S) by the Cahn–Ingold–Prelog convention (Section 5-3). The commonnaturally occurring form of cysteine has a chiral center that is named (R), however.(a) What is the relationship between (R)-cysteine and (S)-alanine? Do they have the oppositethree-dimensional configuration (as the names might suggest) or the same configuration?(b) (S)-Alanine is an l-amino acid (Figure 24-2). Is (R)-cysteine a d-amino acid or anl-amino acid?the stereocenter labelled in blue has an a. R absolute configuration b. S absolute configuration
- The potential energy of a CH3 group in ethane as it is rotated around the C-C bond can be written V= 1/2V0(1 +cos φ), where φ is the azimuthal angle as shown and V0 = 11.6 kJ mol-1. (a) What is the change in potential energy between the trans and fully eclipsed conformations? (b) Show that for smal lvariations in angle, the torsional (twisting) motion around the C-C bond can be expected to be that of a harmonic oscillator. (d) Estimate the vibrational frequency of this torsional oscil lation.Specify the configurations (R or S) at ALL chiral centers in the molecules below. Please note that some molecules below may have more than 1 chiral centers, and you are responsible to find all the chiral centers.How many stereocentres are present in butan‑2‑ol and 2‑bromobutane?
- For the following molecule a)Identify the chiral centers b)assign the order of priority to the substituents c) the corresponding stereochemistry.4. For each of the following structures:a. Identify Chiral Centersb. Identify the chiral carbon configuration as R or Sc. Identify internal symmetry centers if they existd. Identify whether the structure is chiral or achirale. Identify mesostructures from existingWhat are the stereochemical configurations of the two diastereomers of (2S,4R)-2,4-hexanediol? Use the wedge/hash bond tools to indicate stereochemistry where it exists. Include H atoms at chiral centers only. If a group is achiral, do not use wedged or hashed bonds on it.
- How many asymmetric centers does 2,4,6 tnmethy iheptane haveCompounds with several chiral centres may have configurations referred to as meso. Meso configurations arise when the molecule can adopt a configuration with an internal mirror plane. When this occurs, the mirror image of a molecule with an internal mirror plane is an identical configuration. Thus, even though the molecule may possess several chiral centres, they are reflection of each other (within the molecule) thus resulting in an achiral meso form. This occurs for example for tartaric acid (Figure 3.1). Because of this, tartaric acid only has three stereoisomers instead of four. It has the two chiral (R,R) and (S,S) enantiomers, and the meso form. Figure 3.1. Stereoisomers of tartaric acid. Given the information above on meso forms, how many different stereoisomers do compounds A to D (Figure 3.2) have? Figure 3.2. Structure of compounds A, B, C and D.Define F–centre. Mention its one consequence.

