Q: 218) Isume 318) Delng uilibrium? he rate of the forward reaction is equal to the rate of the reverse…
A: As we know, Rate of reaction is the change in concentration of products/reactants in unit time…
Q: How many hydrogen atoms are in a 12.2 g sample of ammonia?
A:
Q: The equilibrium constant, K. for the following reaction is 55.6 at 698 K. H2 (g) + 12 (g)=2 HI (g)…
A:
Q: Calculate the value of the equilibrium constant (K) at 25 °C for the following cell reaction: Sn(s)…
A:
Q: 4. The following materials are available to you: AgNO, to make any molarity of solution needed,…
A: Voltaic cell
Q: For the following cell reaction, the standard cell potential is 1.34 V. To determine the cell…
A: The answer is n = 10. See solution below: Half cell reactions: Oxidation: 5 H2SO3 + 5 H20 => 5…
Q: The reaction of an acid and a base to form water and a salt is called: a. combination b.…
A: We have to predict the correct term for given sentence.
Q: 1- 160.0 mL of 0.25 M HF with 230.0 mL of 0.31 M NaF Ka(HF)=6.8×1O^-4 Express your answer using two…
A:
Q: How many unpaired electrons are in the ground state electron configuration of selenium?
A: Selenium has atomic number 34 So, Se has total 34 electrons
Q: Which of the following is true of pH? It is a measure of the overall acidity. It is a measure of…
A: Acids - - > Acids are any substance that tastes sour in aqueous solution, makes blue litmus paper…
Q: How many moles of LiBr are present in 375 mL of a 0.175 M LiBr solution?
A: Given :- molarity of LiBr solution = 0.175 M volume of solution = 375 mL To calculate :- no.of…
Q: Give a clear handwritten answer and explanation
A:
Q: What is the binding energy in kJ/mol Cl for chlorine-37? kJ/mol 17'H+ 20,n C1 The required masses…
A:
Q: Fill in the equilibrium table for the reaction of H2(g) and N2(g) to form NH3(9). Initial…
A:
Q: If you collected the first drop from the distillation, what would be the mole percent methanol in…
A: Distillation is commercial process to purify the materials.
Q: CH CHOO Br NO, CHCI/AICI3 CI CI CH3 CH3 HBr at 40°C
A: 1. Aromatic Sn2. 2. Aromatic Sn2. 3. Fridel craft alkylation. 4. 1,4-addition at high temperature.
Q: Which of the following describes the reaction: 86 222 Rn → He + 218 Po 84 O alpha decay reaction O…
A: There are different kind of radioactive decays like alpha, beta , positron etc. In radioactive…
Q: Which of the following give(s) the correct order of electron affinities? O Li > Na > K > Rb Na > Mg…
A: Electron affinity :As one electron is added to neutral gaseous atom, it produced a negatively…
Q: 4) Pick up each group that is attached to benzene, and place it in the right family of groups. Ph-X,…
A:
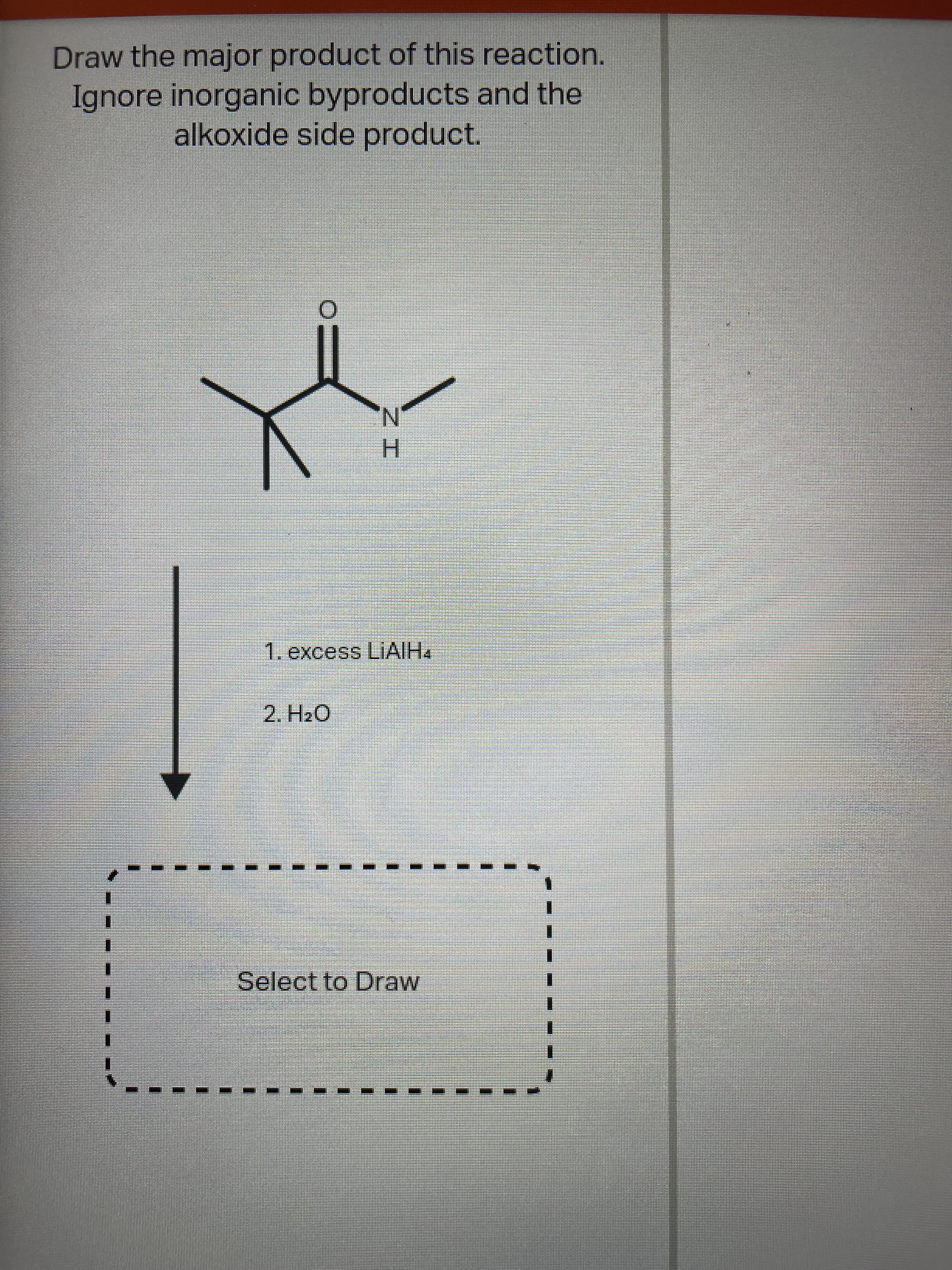
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. lithium…
A:
Q: 10) For bromine, AH°vap = 30.91 kJ/mol and AS°vap 93.23 JK-1mol-1 at 25°C. What is the normal…
A: Given, Delta H = 30.91 kJ/mol Delta S = 93.23 J/(mol K)
Q: Which of the following describe(s) the following reaction? Choose all that apply. 141- ABa 56 Kr +…
A: ->When neutron is bombards on heavier nuclei then it splits into smaller nuclei .It is called…
Q: Give the IUPAC name of the following compound:
A:
Q: Chy-C - CH, + H* CHg - C- oH + HO-CHy → CHy-Ö-O-CHg + HOH CHy-Ċ = CHg +HqO %3D 84
A: In reaction 1- Ethanoic acid + methanol --> Methyl acetate In reaction 2- 2-Methyl propene…
Q: Solve for the molar solubility of solid Fe2(SO4)3 in a solution of 0.75M BaSO4. 1.098 x 105. Compare…
A:
Q: rall reaction and standard cell potential of an electrochemical cell constructed from half-cells…
A:
Q: An unknown volume of gas is held at a temperature of 115 K in a container with a pressure of 64…
A: given :- initial pressure = 64 atm initial temperature = 115 K final pressure = 32 atm final…
Q: How many grams of silver can be plated onto an object in 9.87 minutes at 4.74 A of current ?…
A:
Q: How many molecules of carbon monoxide (CO) needed to react with-excoss trOn () oxide-(E03) to…
A:
Q: gallium: [Ar]4s²3d1º4p' 3d10 4s2 ,4p' inner electrons: outer electrons: Incorrect Incorrect valence…
A:
Q: A chemist must prepare 650. mL of 1.00 M aqueous sodium chloride (NaCl) working solution. She'll do…
A: Given :- concentration of stock solution = 1.24 M concentration of dilute solution = 1.00 M…
Q: The % concentration by mass of a solution prepared by dissolving 14.5 g NaCl in 235. g solution is:…
A:
Q: At a certain temperature, the reaction 2HF(g) H2(g) + F2(g) has Kc = 1.2 × 10-13. Does this reaction…
A:
Q: NH3g) + HC(G) the equilibrium concentration of HCI when 0.237 moles of NH,Cl(s) are introduced into…
A: As we can see, it is heterogeneous equilibrium. And pure solid and pure liquids are not included in…
Q: What are the [H,O*| and [OH] of Diet Coke that has a pH of 3.17? ()
A:
Q: 4. CHg CH2-NH- CH2 CHz 5. CH, CH2 CH2 CH2-Ċ-0H
A: Organic compounds can be defined as the compounds containing carbon and hydrogen atoms. For example,…
Q: Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are…
A:
Q: Consider the reaction 4HCI(g) + 02(g)2H20(g) + 2C12(g) Using the standard thermodynamic data in the…
A:
Q: A Haber process reactor contains 25.0% nitrogen and 75.0% hydrogen for the synthesis of ammonia.…
A:
Q: Calculate to five significant figures the binding energy in J/nucleon of the nucleus of an atom of…
A:
Q: 1. 20.0 ml of 0.500 M Acetic Acid (CH3COOH) is titrated with 0.500 M KOH. Find the pH a. Before…
A:
Q: As the concentration of reactants decrease, the rate of the reaction generally O remains constant. O…
A: As the concentration of the reactants decrease, the rate of 5ge reaction generally Remains…
Q: In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used…
A: Specific heat capacity of water: 4.182 J/(g oC) Specific heat capacity of copper: 0.39 J/(g oC)…
Q: Calculate the volume in milliliters occupied by one molecule of ethyl alcohol, C2H5OH , in a beaker…
A: We have to calculate the volume in mL.
Q: Which of the following is the cell notation for a voltaic cell based on the following reaction?…
A: Cu2+(aq) + Pb(s) + SO42–(aq) → Cu(s) + Pb2+(aq) + SO42-(aq) Oxidation half reaction :-…
Q: Calculate the pH of each of the following solution? a) 0.10 M HCl b) 0.10 M NaOH
A:
Q: Give the mechanism that leads to the product. If multiple products are possible, you must list all…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which enol will undergo tautomerization most slowly? A B OH Ме. Me Но- Ме Ме Me O A O B ос
A: 1) The enol that will undergo tautomerization most slowly is
Q: 7. Air is composed of approximately 78% N2, 21% O2 and 1% percent other gases. Calculate the density…
A:
Q: Which of the following describes the reaction: positron emission O beta decay reaction bombardment…
A: Given that, a decay reaction is We have to tell the type of the decay reaction.
Step by step
Solved in 2 steps with 3 images

- What is the major product of the follwoing reaction. The correct answer is B, please show the full mechansim and compare it to the other options.Organic Chemistry II: How do we know which direct the reaction? Why Methoxy Functional Group direct the reaction instead of fluorine? Is it something to do with electronegativity ? Or is it something to do with withdrawing group?? How do we know if a gourp is more withdrawing ??????Please show all arrowing pushing steps to find the product of this reaction.
- 1. what reaction and meachanism is this ? 2. Explain mechanism what happenning in each step on this picture 3. why is it says "stop" when NaOEt added ? does reaction stop there ? why does it stop ? ? what principle ?Draw the major product of this reaction. Ignore inorganic byproducts and the phthalic acid side-product.Draw the final product of the third step after treatment with KMnO4, KOH, and heat followed by aqueous acidic workup.
- Please clearly draw the overall reaction taking place between methyl salicylate and sodium hydroxide. Be sure to include all reagents and by-products. Show the mechansim as well.For the reaction scheme below: (a) Suggest a reagent X that could be used to accomplish the first step. (b) Draw the curly arrow mechanism for the second step in the scheme.Do I use a Wittig reaction?

