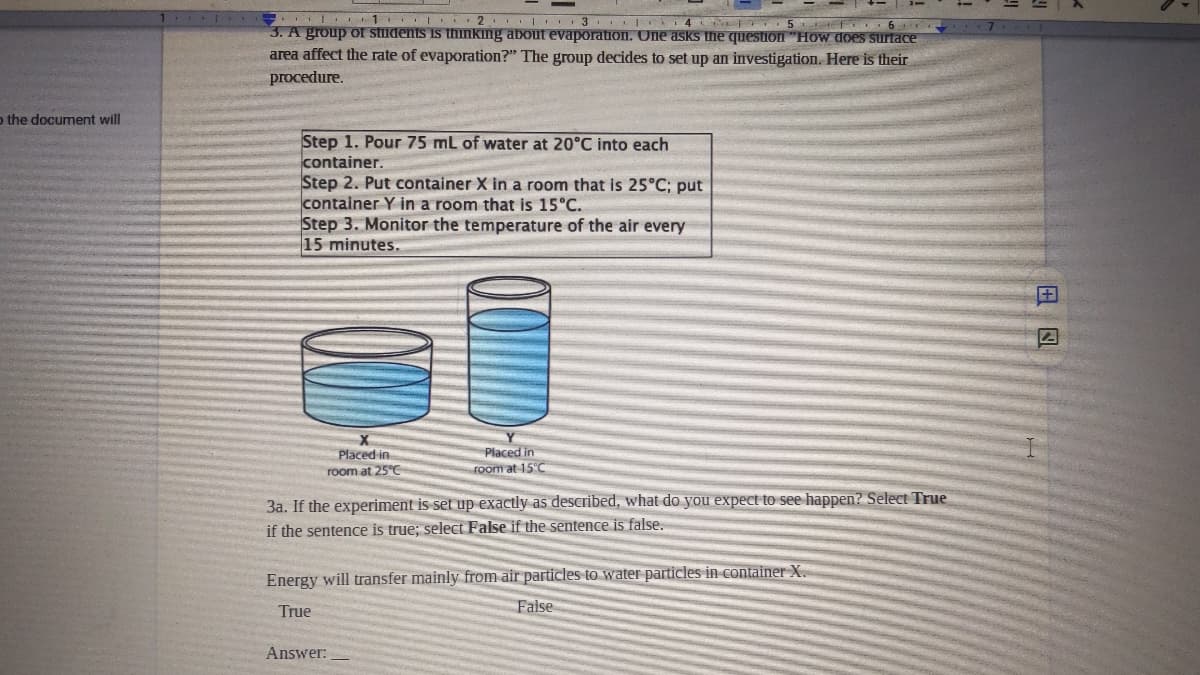
II 1 I .2. I | II3 6. 3. A group of students iS thinking about evaporatiIon. One asks the question "How does surtace area affect the rate of evaporation?" The group decides to set up an investigation. Here is their prоcedure. Step 1. Pour 75 mL of water at 20°C into each container. Step 2. Put container X in a room that is 25°C; put container Y in a room that is 15°C. Step 3. Monitor the temperature of the air every 15 minutes. Placed in Foom at 15°C Placed in room at 25°C 3a. If the experiment is set up exactly as described, what do you expect to see happen? Select True if the sentence is true; select False if the sentence is false. Energy will transfer mainly from air particles to water particles in container X. True False Answer:
II 1 I .2. I | II3 6. 3. A group of students iS thinking about evaporatiIon. One asks the question "How does surtace area affect the rate of evaporation?" The group decides to set up an investigation. Here is their prоcedure. Step 1. Pour 75 mL of water at 20°C into each container. Step 2. Put container X in a room that is 25°C; put container Y in a room that is 15°C. Step 3. Monitor the temperature of the air every 15 minutes. Placed in Foom at 15°C Placed in room at 25°C 3a. If the experiment is set up exactly as described, what do you expect to see happen? Select True if the sentence is true; select False if the sentence is false. Energy will transfer mainly from air particles to water particles in container X. True False Answer:
Chapter5: Gases
Section: Chapter Questions
Problem 108E
Related questions
Question
Transcribed Image Text:. 1 I 2 3 4 5 6
3. A group of students iS thinking about evaporatiIon. One asks the question "How does surtace
area affect the rate of evaporation?" The group decides to set up an investigation. Here is their
procedure.
o the document will
Step 1. Pour 75 mL of water at 20°C into each
container.
Step 2. Put container X in a room that is 25°C; put
container Y in a room that is 15°C.
Step 3. Monitor the temperature of the air every
15 minutes.
Placed in
room at 25°C
Placed in
Foom at 15°C
3a. If the experiment is set up exactly as described, what do you expect to see happen? Select True
if the sentence is true; select False if the sentence is false.
Energy will transfer mainly from air particles to water particles in container X.
True
False
Answer:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning