(ii) Rank the carbonyl compounds given below in terms of their relative reactivity with sodium borohydride. CH3 (iv) Suggest two factors which combine to make the aldehyde react with sodium borohydride at a different rate from the ketone. (v) Based on electronegativity one might assume that the ester and acid chloride possess similar reactivity towards nucleophiles. Suggest why this turns out not to be the case (your answer should mention resonance and inductive effects).
(ii) Rank the carbonyl compounds given below in terms of their relative reactivity with sodium borohydride. CH3 (iv) Suggest two factors which combine to make the aldehyde react with sodium borohydride at a different rate from the ketone. (v) Based on electronegativity one might assume that the ester and acid chloride possess similar reactivity towards nucleophiles. Suggest why this turns out not to be the case (your answer should mention resonance and inductive effects).
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter23: Amines
Section: Chapter Questions
Problem 23.37P
Related questions
Question
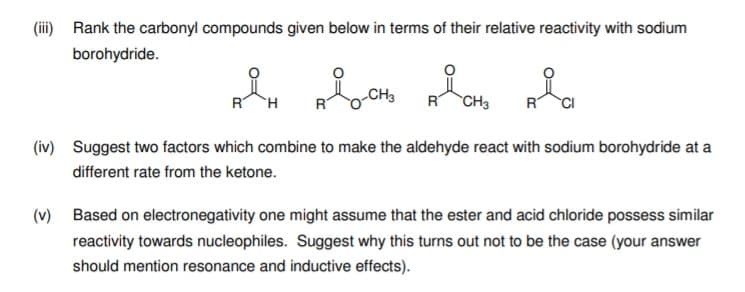
Transcribed Image Text:(iii) Rank the carbonyl compounds given below in terms of their relative reactivity with sodium
borohydride.
RT
R CH3
CI
(iv) Suggest two factors which combine to make the aldehyde react with sodium borohydride at a
different rate from the ketone.
(v) Based on electronegativity one might assume that the ester and acid chloride possess similar
reactivity towards nucleophiles. Suggest why this turns out not to be the case (your answer
should mention resonance and inductive effects).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning