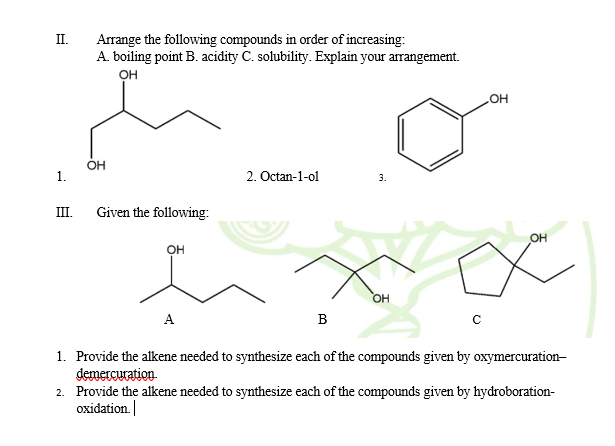
II. Arrange the following compounds in order of increasing: A. boiling point B. acidity C. solubility. Explain your arrangement. OH HO OH 2. Octan-1-ol 3.
Q: Pyridine (right) is an essential portion of many biologically active compounds, such as nicotine and…
A: Pyridine is an organic heterocyclic, chemical C5H5N compound. It is similar to that of benzene…
Q: Isomers that are mirror images of each others are called Select one: O a. Enantiomers O b.…
A: Enantiomers, otherwise called optical isomers, are two stereoisomers that are identified with one…
Q: 1. Complete the given table below. What properties do each of the following R groups have?…
A: Basic-Positively charged, alcohol group Acidic - Negatively charged Neutral - No net charge Polar -…
Q: 5) Decide on the most likely mode of denaturation for each of the following compounds... a) hexane…
A: Denaturation is the process of the breaking the linkages or bonds that are formed in the compounds…
Q: The bond Indicated with a green arrow below Is referred to as a... bond. Check the proper term that…
A: Monosaccharides- Simple sugar molecule- having One oxygen atom and two Hydrogen atoms for a single…
Q: It is possible to make a buffer that functions well near pH7 using citric acid, which contains only…
A: Citric Acid is a type of weak acid with a pH of 3 to 6 and is commonly used as a buffering agent,…
Q: The pk, of the conjugate acid of guanidine is 13.6, making it one of the strongest neutral organic…
A: Guanidine(CH5N3) and conjugate acid is called the guanidinium cation, (C(NH2)+3) pka - Acid…
Q: ◆ Arginine Phenylalanine ◆ Cysteine ◆ Serine Glutamic acid ◆ Glutamine ◆ Isoleucine Leucine A.…
A: There are 20 naturally occurring amino acids. In a polypeptide chain, the alpha amino and the alpha…
Q: 4) The result above occurred because the potassium permanganate crystals have a weight than the…
A: As per our company's guidelines we are supposed to answer a single at a time, please repost the rest…
Q: 1. How many molecules of NaCl are there in a 50.0 g sample of NaCl? The molecular weight of NaCI is…
A: Note: We are authorized to answer one question at a time since you have not mentioned which question…
Q: 9. What bonds are affected by: а. Нeat b. Organic solvents c. Metallic salts d. Alkaloidal reagents…
A: Proteins are composed of amino acids linked by peptide or amide bonds (covalent), which form the…
Q: 5. What is the reaction of phenolphthalein and methyl orange in acid-base solution? Acid Base…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Polymers of macromolecules are disassembled to monomers by: O a. Dehydration reaction O b. Disulfide…
A: Polymers are the large molecules that are formed by the attachment of small subunits of molecules…
Q: _7. Which of the following FA has the lowest melting point? A. Stearic acid B. Palmitic acid C.…
A: A fatty acid is a molecule that is composed of a long hydrocarbon chain attached to the carboxylic…
Q: 7. The only carbohydrate which is not having any chiral carbon atom is (in 1 to 3 sentences, explain…
A: Carbohydrates are essential biomolecules which are also sugar molecules. Carbohydrates, along with…
Q: Soft drinks usually have a pH of approximately 3.1. What is the [H3O+] concentration in a soft…
A: Consumption of soft drinks is a very controversial issue for public health. Soft drinks contains…
Q: 1. Label the "C" monosaccharide 2. Draw the open chain Fischer projection formula of the…
A: In the given molecule the different sugars are attached with the help of glycosidic bonds.
Q: 1. What class of biomolecules does this compound belong to? Explain why. 2. Break this compound down…
A: A biomolecule is any of the numerous substances that are produced by the cells and living organisms.…
Q: The configuration of the given structure below is: * CH,OH он ÓH O alpha-L O alpha-D O beta -L O…
A: The carbohydrates can be represented by two types of configurations: L-isomer and D-isomer. When the…
Q: 1. Choose the formula for threonine: a) CH₂ -CH-COOH OH NH₂ b) CH3 -CH-CH-COOH OHNH,
A: Amino acids are essential for the growth and development of the body. Amino acids are considered to…
Q: Your friend need to purify a sample left behind by an older graduate student to isolate the…
A: The answer given below in the image
Q: How many H2O particle were produced by creating the following How many peptide bonds were produced…
A: Introduction: Amino acids are joined together by a condensation reaction between carboxylic and…
Q: 7. The chemical you are testing has a concentration of H* as 10³ M (must watch the video). A) What…
A: pH is a measure of the concentration of hydrogen ions in a given solution. Higher the concentration…
Q: 2. Of the following amino acids, which contains an alcohol? a b HN. HN. HN. HN. HO, HO, HO, SH NH.…
A: Introduction: Amino acids are biomolecules comprise of two functional groups; an amino group (-NH2)…
Q: 1) If the pH of the rain drops from 6 to 5 due to air pollution, the rain has become: a) two time…
A: Acid rain caused when the rain is mixed with the volatile toxic compounds like sulfur dioxide and…
Q: 3. Identify the monosaccharide represented by each of the following structures. Name each compound H…
A: Monosaccharide or simple sugars are the most basic unit of carbohydrates that can not be hydrolyzed…
Q: 5, describe the unique properties of water.
A: Water's unique properties: Water can dissolve a wide range of chemicals, which is why it is such an…
Q: 13. ich of the following bonds share electrons? A. Ionic bonds B Covalent bonds C. All of the above…
A: Different biomolecules are formed due to the formation of chemical bonds. A chemical bond is an…
Q: 4. Which of the following would be considered a basic solution? A. Solution with [OH-] = 3.8 x 10 M…
A: We can find out this using formula pH + pOH = 14 pOH = -log[OH-] and pH = -log [H+]
Q: 2. Write the formula for the conjugate acid for each of the following bases: a) CO3- b) H:O c) H:POr
A: A conjugate acid–base pair, according to the Bronsted–Lowry definition of acids and bases, consists…
Q: 9. Which of the following is a compound lipid?|
A: Compound lipids are esters of fatty acids which consist of fatty acid, alcohol, and additional group…
Q: III. Convert the ff. cyclic sugar structures in Fischer formula into their corresponding Haworth…
A: Carbohydrates: Is a biomolecule having ketone and aldehyde groups also known as aldoses and ketoses…
Q: 4. Determine if the following compound is chiral or not. OH Но. HO Me ZOH ZoH HO HO, OH Ме CI CI OH…
A: The parameters based on which chirality can be determined are: 1. Visualise the mirror image of the…
Q: 3. What are the effects of the following solvents when mixed with lipids? What type of hydrolysis is…
A: The lipids are the chemical substances or macromolecules that are usually insoluble in water but…
Q: Explain why lipids are insoluble in polar solvents. 2. Aside from their sources and physical states…
A: Lipids: Lipids are a heterogeneous group of naturally occurring organic compounds that include…
Q: 1. Polar Covalent Bonds a. Draw one water molecule. b. Indicate which bonds are polar covalent bonds…
A: Water is a polar molecule. It made up of 2 hydrogen atoms and 1 oxygen atom. Molecular formula…
Q: H3C- -CH3 CH3 H3C CH3 Но ČH3 -CH3 A B D H2N NH2 OH CH3 -CH3 -CH3 H3C- CH3 H3C- H3C CH3 H;C ČH3 F G H
A: A functional group is a substituent in a molecule which governs it's characteristics of chemical…
Q: 7. Which of the following is a valid buffer? A. 0.30 M HNO, and 0.30M NANO, B. 0.20 M HF and 0.10 M…
A: Buffers are solutions that do not alter pH when diluted or when tiny volumes of acids or alkalis are…
Q: 1. The K, of acid HX is 2.0 x 10 NaX is a salt that produces its conjugate base X What is the pH of…
A: Given Values: Ka value of [HA] = 2.0 × 10-6 pKa = - log Ka = - log 2 × 10-6 = 6 - log 2 = 6 -0.30 =…
Q: 1. Write the formula for the conjugate base for each of the following acids: а) НСО- b) HРО c) HBRO
A: The absolute concentration of acid is required to keep the buffer's pH stable when new ions are…
Q: Which one of the following statements about a molecule of acetonitrile (CH,CN) is TRUE? Select one:…
A: Chemical bonds are simply electrostatic attractive forces between atoms. There are mainly 3 types of…
Q: 1. Which of the following is a weak base? A. CH,COOH B. CH;CH;NHCH, C. HOCN D. NAOH E. None of…
A: Base and acid both interact with each other and form salt and water. They both are used in everyday…
Q: Below is the structure of butyl alcohol. Based on the results of the experiment you just performed,…
A: Butyl alcohol is known to effect cell permiability.
Q: 9. Añ ester is treated with aqueous acid. What is this reaction called? OA Esterification O B. Acid…
A: Esters are neutral, class of organic compounds that react with water and produces alcohols and…
Q: 6. What functional groups are present in the following molecul HạN. OH NH. A) amine and carboxylic…
A: There are several functional groups that can be found in a molecule. Some examples of functional…
Q: 1. Draw a circle around and give the name of each functional group in the following twomolecules.…
A: A functional group of a compound is an atom or a group of atoms placed in a certain way that tends…
Q: How is it possible for soap to have a pH greater than 10? What substance used in soap making that…
A: The majority of the soaps have a pH within the range of 9-10. The majority of the shampoos have a pH…
Q: Polymers of macromolecules are disassembled to monomers by: O a. Van der Waals interaction O b.…
A: A polymer is a large molecule that is made from many small repeating chemicals called monomers.
Q: 1) A specific fat/oil has on over 50% of its triglyceride sites oleic acid, 28% palmitic acid, and…
A: Since you have posted multiple questions, we will answer the first one for you. If you want a…
Q: 9. An ester is treated with aqueous acid. What is this reaction called? A. Esterification B. Acid…
A: Esters are neutral, class of organic compounds that react with water and produces alcohols and…
Step by step
Solved in 2 steps with 2 images

- 1. provide three reasons why most of the research on carotenoids concentrates on b-carotene. 2. draw the retinol ester that has stearic acid as the carboxylic acid portion of its structure.1. Why is β-carotene less polar than chlorophyll b? Mention any TWO functional groups which make chlorophyll b more polar than carotene. 2. What is the degree of unsaturation of a β-carotene molecule?1.The specific rotation of a sample of mandelic acid [PhCH(OH)CO2H] is -9.48° and of pure S-(-)- mandelic acid is -158°. Calculate the (1) % optical purity, (2) ee, (3) % composition and (4) draw a clear stereo-structure for S-(-)-mandelic acid. 2. Explain each of the following concepts: a)positional isomerism b) Newman projection formula c) Chemoselectivity 3. use the molecular formula C4H8O2 to illustrate the following isomers: a) (+)-gauche and antiperiplanar b) chain isomer c) Enantiomer d) Cis-and Trans cycloalkanes
- In observing a Haworth or cyclohexane-chair representation of alpha-d-glucopyranose, the anomeric carbon can de best identified by: a) the carbon atom which is bonded to the most H atoms b) the carbon atom which is bonded to the least H atoms c) The carbon atom which has its OH group pointing down d) The carbon atom which is bonded to two oxygen atoms Cellulose differs from amylose in that: a) cellulose has 1-6 branches while amylose does not b) amylose has 1-6 branches while cellulose does not c) amylose has alpha glycosidic bonds while cellulose has beta glycosidic bonds d) cellulose contains sulfate while amylose does not. The glycosaminoglycan (mucopolysaccharide) which is not normally associated with joint tissue is: a) heparin b) keratan sulfate c) chondroitin sulfate d) hyaluronic acid In glycoproteins, which amino acid does not bond sugar molecules? a) Asn b) Ser c) Thr d) Gly Which feature do all lipid molecules share in common? a) fatty acid molecules…The following chemical reactants produce the ester ethyl ethanoate (C4H8O2): C2H6O + CH3COOH What type of reaction occurs to make ethyl ethanoate? condensation hydrolysis combustion acid-base reactionIV. Draw the Fischer projection formula for the enantiomer of each of the following monosaccharides.
- 1The sugar alcohol often used in “sugarless” gums and candies is L-sorbitol. Much of this alcohol is prepared by reduction of D-glucose. Compare these two structures and explain how this can be.59. Arrange the following five hydrocarbons in order of increasing of boiling pointA. 2-methylhexane B. heptane C. 3,3-dimethylpentandeD. Hexane E. 2-methylpentanceGive at least 10 examples of biological compounds having an alkyne functional group and identify the biochemical importance of each compound.
- 1. Halogenation depends on which concept? a. presence of double bond in the glycerol portion b. presence of double bond in the fatty acid portion c. presence of carboxylic acid groups d. reactivity of potassium ions 2. Why do lipids with long chain fatty acids have lower saponification value? a. they are linear in structure, thus it is difficult to saponify b. they have lower number of carboxyl group per 1 gram of sample c. they require less amount of KOH during titration d. they require less HCL during back titration 3. Melting point: what happens when the carbon chain length of fatty acids become shorter? a. increased melting point b. decrease melting point c. no changeAcetone, CH3–(C=0) –CH3, and urea, NH2–(C=0)–NH2, have similar chemical structures. (a) Compare the possible intermolecular forces for acetone with those for urea. (b) Compare the intermolecular forces between phycocyanin and acetone with those between phycocyanin and water. Briefly explain how acetone disrupts the folding in phycocyanin. (c) Compare the intermolecular forces between phycocyanin and urea with those between phycocyanin and water. Briefly explain how urea disrupts the folding in phycocyanin. (d) Compare your experimental observation on the disruption of phycocyanin folding in the presence of acetone with that in the presence of urea. Briefly explain whether your comparison is consistent with the properties of acetone and urea.The linear tripeptides are formed from the following three amino acids as the starting materials in the condensation reaction. Select the correct statement: (a) There are 27 possible tripeptides and among them, 13 tripeptides are optically active. (b) There are 26 possible tripeptides and all are optically active. (c) There are 27 possible tripeptides and among them, 26 tripeptides are optically active. (d) There are 26 possible tripeptides and among them, 15 tripeptides are optically active.

