Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter3: Matter
Section: Chapter Questions
Problem 12CR
Related questions
Question
100%
I’m not sure how to do the density of the materials
Transcribed Image Text:Post Attendee - Zoom
Launch Meeting - Zoom
O Post Attendee - Zoom
(5) Maher Zain
nstructure.com/courses/6926/assignments/150320
..
M Gmail
YouTube
A Maps
i Assignments | Micr.
Mail - Aligab, Isaak.
Dashboard
* VHL Central | Home
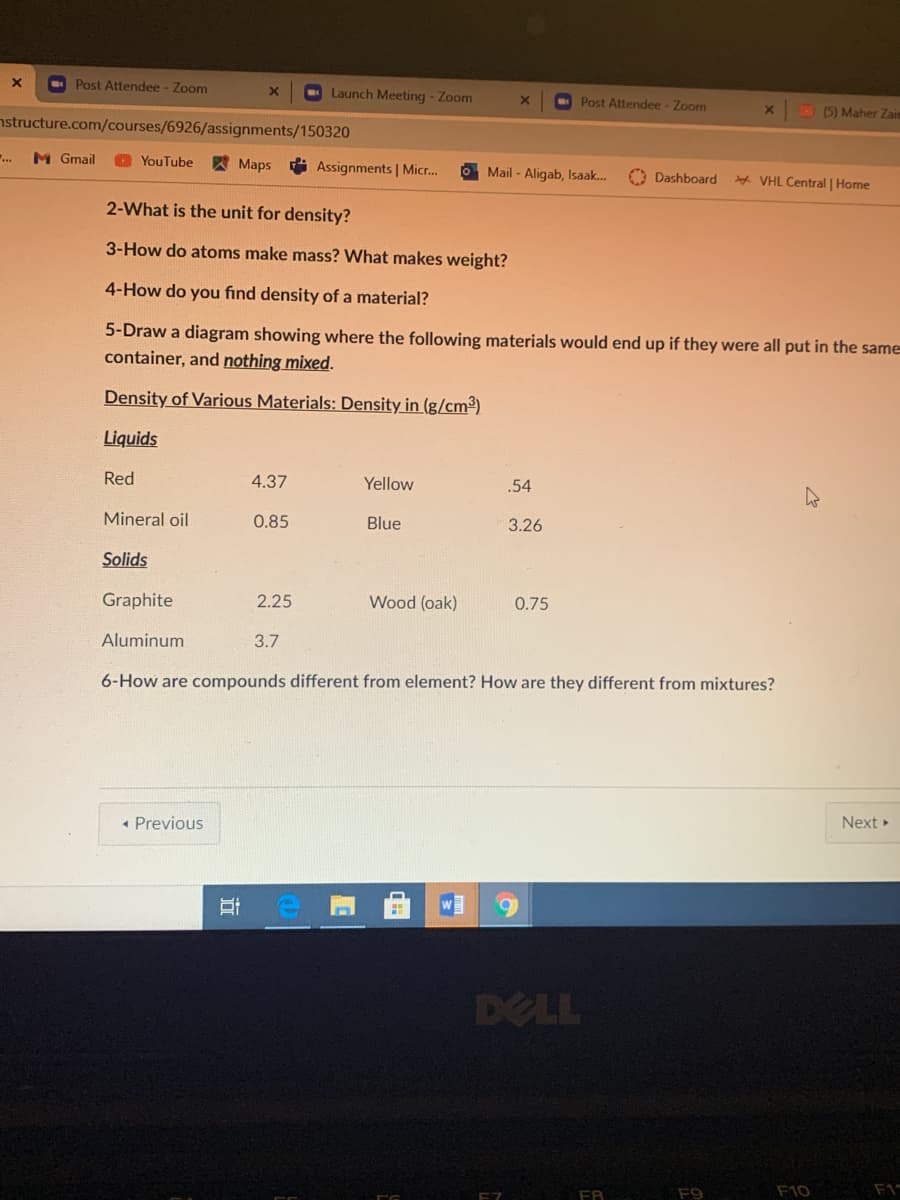
2-What is the unit for density?
3-How do atoms make mass? What makes weight?
4-How do you find density of a material?
5-Draw a diagram showing where the following materials would end up if they were all put in the same
container, and nothing mixed.
Density of Various Materials: Density in (g/cm³)
Liquids
Red
4.37
Yellow
.54
Mineral oil
0.85
Blue
3.26
Solids
Graphite
2.25
Wood (oak)
0.75
Aluminum
3.7
6-How are compounds different from element? How are they different from mixtures?
« Previous
Next
DELL
F9
F10
F1
FB
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning