Q: Adding acetophenone slowly to a cold solution of LDA produces the enolate of acetophenone; but…
A: SOLUTION: Step 1: The organic reaction in which a chemical compound containing a carbonyl group acts…
Q: please explain the reaction and mechanism of sulfide to sulfimide
A: Allylic amine is an organic compound. It is an unsaturated amine. It is colorless.
Q: Esters have two unique sides. When looking at an nmr spectrum the hydrogens alpha to the carbonyl…
A: Esters have two NMR active Hydrogens 1) Alpha to Carbonyl Carbon 2) Alpha to Oxygen atom of Ester…
Q: Why can a NaOH be used to distinguish amines and ammonium salts? Multiple choice 2. In the…
A: Hello. Since your question has multiple parts, we will solve first question for you. If you want…
Q: a. What two amides are obtained from the reaction of acetyl chloride with an equivalent of…
A: SOLUTION: Step 1: The reaction of acetyl chloride with an equivalent of ethylamine and an equivalent…
Q: Aniline is basic, and acetanilide is not basic. Explain this difference. by usig Hic the hydrolysis…
A: a) Basicity is defined as the ability of a compound denotes their lone pair that is ability of base…
Q: 1. Why is acetanilide no longer available as a drug in the market? 2. Why is the synthesis of…
A:
Q: In the 1880's, Acetanilide, sold under the name Antifebrin, was widely used as a pain reliever and…
A: Here we have to write the mechanism of acid catalyzed hydrolysis of acetanilide and synthesis of…
Q: Several additional amine syntheses are effectively limited to making primary amines. The reduction…
A: (a) Reaction between allyl bromide with sodium azide to give allyl aide. This allyl azide is reduced…
Q: Safrole, which is isolated from sassafras (Problem 21.33), can be converted to the illegal stimulant…
A: (a) Please find below a synthesis that begins with safrole and uses a nucleophilic substitution…
Q: Why does p-nitroacetanilide recrystallize in ethanol, but the o-acetanilide remains in the filtrate…
A: The separation of p-nitroacetanilide and o-nitroacetanilide can be done on the basis of its…
Q: Explain why a secondary amine forms a nitrosamine rather than a diazonium ion when it reacts with…
A: To understand the reaction consider the reaction of a primary amine with nitrosonium ion. The…
Q: n-acetylazoles go through hydrolysis more than regular amides. propose a reason for the reactivity…
A: Amide hydrolysis is not so easy because of the donating ability of lone pair of electrons on…
Q: Esters and amides are most easily made by nucleophilic acyl substitution reactions on… A. alcohols…
A:
Q: The reaction of an ester with an amine is not as slow as the reaction of an ester with water or an…
A: Since you have posted a multiple question, we will solve first one for you. To get remaining…
Q: Explain why compound A readily decarboxylates (forming pyrrole) when heated with acid, but compounds…
A: Introduction: Decarboxylation: Decarboxylation reaction is the reaction in which carbon dioxide…
Q: In the Gabriel synthesis of primary amines, phthalimide is used to accomplish the first step –…
A: Solution : The Gabriel synthesis is a chemical reaction that involves the conversion of the alkyl…
Q: Safrole, which is isolated from sassafras (Problem 21.33), can be converted to the illegal stimulant…
A: a) Please find below a synthesis that begins with safrole and uses a nucleophilic substitution…
Q: List the following esters in order of decreasing reactivities towards hydrolysis with reason:…
A: In hydrolysis reaction, water molecule is added to the substrate. Esters can be converted to…
Q: Give the products expected when the following tertiary amines are treated with a peroxyacid and…
A: INTRODUCTION: Cope elimination is defined as the reaction in which amine to oxidised to N-oxide, on…
Q: Rank the following amines based on basicity Rationalize the electronic effects responsible for the…
A: A compound can be said to be basic when it has the ability to donate its lone pair or the non-bonded…
Q: When N,N-dimethylaniline is treated with chlorine and aluminum trichloride, ortho and para products…
A:
Q: What carbonyl and nitrogen compounds are needed to make attached compound by reductive amination?…
A: Reductive amination is the synthesis of amines from aldehydes and ketones. For the given compound…
Q: Explain why a secondary amine forms a nitrosamine rather than a diazonium ion when it reacts with a…
A: Given reaction is:
Q: What product(s) would you expect from the Hofmann elimination of the following amines? If more than…
A:
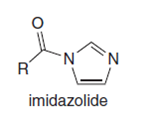
Q: Explain why imidazolides are much more reactive than other amides in nucleophilic acyl substitution.
A: It is known that, imidazolides are much more reactive than other amides in nucleophilic acyl…
Q: Why can a NaOH be used to distinguish amide and ammonium salts? Multiple choice 2. In the synthesis…
A:
Q: With reference to amides A and B, the carbonyl of one amide absorbs at a much higher wavenumber in…
A: The carbonyl groups with amide in it generally shows signal in IR spectrum at 1630-1680 cm-1. This…
Q: Prozac may be prepared via a Mannich reaction using which amine? Benzylamine Methylamine Ethylamine…
A: A chemical reaction in which organic compounds take part is called organic reactions. Organic…
Q: Reduction of aromatic nitro-compounds using Sn and HCl gives :Select one a. aromatic secondary…
A: Given, Reduction of aromatic nitro compounds using Sn and HCl gives = ? Sn/HCl - is a reducing agent…
Q: Match each item to a choice: Choices: alkene Reductive amination Curtius rearrangement Hoffman…
A: Match the following of amine reactions
Q: What kinds of halides cannot be used to alkylate an amine? Why not? a.Tertiary halides form alkenes…
A: Correct Option : d. all of the above
Q: Explain Imine and Enamine Hydrolysis ?
A: Hydrolysis of imines and enamines under acidic condition will result in the formation of an aldehyde…
Q: How would you account for the following :(a) Electrophilic susbstitution in case of aromatic amines…
A: In an electrophile substitution reaction, the electrophile (E+) substitute on of the position in the…
Q: Describe how the following compounds could be prepared from cyclohexanone using an enamine…
A: (a) Cyclohexanone when treated with pyrrolidine in the trace amount of acid, an enamine intermediate…
Q: Imidazole is more basic than pyridine, but more acidic than pyrrole, why?
A: A question based on acidic and basic nature, which is to be accomplished.
Q: What alkyl bromide would you use in a Gabriel synthesis to prepare each of the following amines? a.…
A: a.
Q: Based on the following groups Acid chloride, Amide, Ester a) Select the most reactive group towards…
A: The reactivity of the acyl compound depends on the partial positive charge present on the carbonyl…
Q: Which amines cannot be prepared by the Gabriel synthesis? Explain your choices
A: The Gabriel Synthesis of primary amines: The Gabriel Synthesis consists of two steps and uses a…
Q: Answer both parts plz. Part a) Draw arrow pushing mechanism. Part b) Compare the reactivity of…
A: a) The arrow pushing mechanism for the given reaction is shown below,
Q: Compare the physical properties of acid derivatives, and explain the unusually highboiling points…
A: The carboxylic acid and its derivatives have higher boiling point and melting point than the…
Q: Which of the following reaction or reagent does not produce an amine molecule? A. Curtius…
A: The given reactions are A. Curtius rearrangement B. Reaction of oximes with metallic sodium in…
Q: Give reasons :(a) Aniline is a weaker base than cyclohexyl amine.(b) It is difficult to prepare pure…
A: A strong base is a base that dissociates completely to give ions in solution and a weak base is a…
Q: Which amines cannot be prepared by the Gabriel synthesis? Explain your choices
A: Gabriel synthesis: Gabriel synthesis is a chemical reaction that transforms an alkyl halide into a…
Explain why imidazolides are much more reactive than other amides in nucleophilic acyl substitution.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- N-p-hydroxyphenylethanamide is commonly known as a. acetaminophen b. acetamide c. acetanilide d. formamide High molar mass amines have __________ odor. a.strong ammoniacal b.fruity c.fishy d.obnoxious Trimethyl amine has _________ odor. a.obnoxious b.fishy c. ammoniacal d. fruityEquanil is __________. (i) artificial sweetener (ii) tranquilizer (iii) antihistamine (iv) antifertility drugWhich five benzodiazepines are approved for hypnotic use?
- Why are some physicians are careless when prescribing benzodiazepines for patients suffering from severe anixety?A patient taking an antispasmodic may experience which side effect?Water intoxication is most likely to be a problem among users of: a. SSRIs b. traditional antipsychotics with low anti-ACh activity c. benzodiazepines d. traditional antipsychotics with high anti-ACh activity e. MAOIs f. a and e

