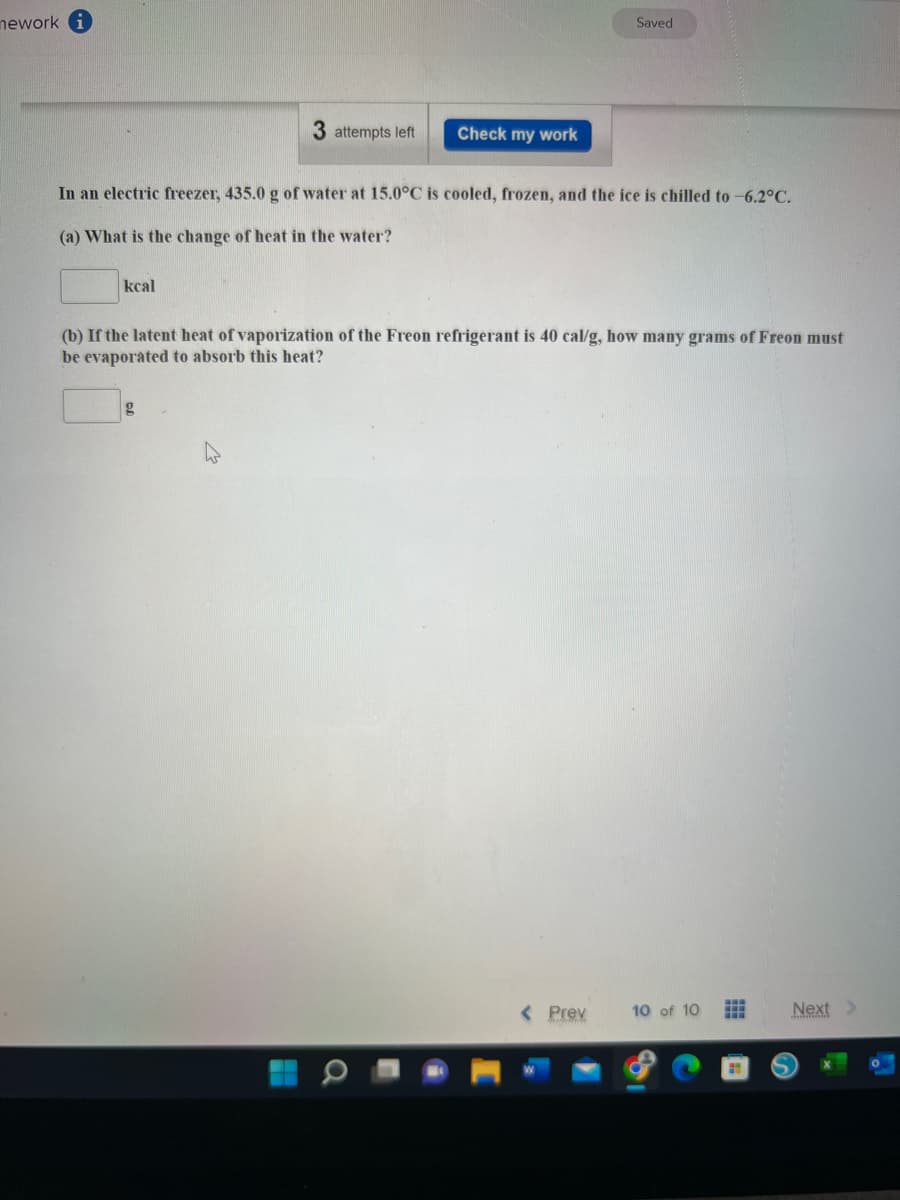
In an electric freezer, 435.0 g of water at 15.0°C is cooled, frozen, and the ice is chilled to -6.2°C. (a) What is the change of heat in the water? kcal (b) If the latent heat of vaporization of the Freon refrigerant is 40 cal/g, how many grams of Freon must be evaporated to absorb this heat? g
In an electric freezer, 435.0 g of water at 15.0°C is cooled, frozen, and the ice is chilled to -6.2°C. (a) What is the change of heat in the water? kcal (b) If the latent heat of vaporization of the Freon refrigerant is 40 cal/g, how many grams of Freon must be evaporated to absorb this heat? g
Chapter1: Temperature And Heat
Section: Chapter Questions
Problem 76P: On a trip, you notice that a 3.50-kg bag of ice lasts an average of one day in your cooler. What is...
Related questions
Question
Transcribed Image Text:nework
Saved
3 attempts left
Check my work
In an electric freezer, 435.0 g of water at 15.0°C is cooled, frozen, and the ice is chilled to -6.2°C.
(a) What is the change of heat in the water?
kcal
(b) If the latent heat of vaporization of the Freon refrigerant is 40 cal/g, how many grams of Freon must
be evaporated to absorb this heat?
g
< Prev
10 of 10 #
Next >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College



College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College
