In Section 12.2, we learned that the Simmons-Smith reaction produces a cyclopropane ring from an alkene. Diiodomethane (CH,I2) is treated with a source of zinc to produce the Simmons-Smith reagent (ICH,ZnI), which reacts with the alkene in a single elementary step. Complete the mechanism below by adding the necessary curved arrows for this elementary step. Hint: The curved arrow notation is very similar to that for epoxidation involving a peroxyacid.) H, CH2 Enantiomer + Simmons-Smith reagent
In Section 12.2, we learned that the Simmons-Smith reaction produces a cyclopropane ring from an alkene. Diiodomethane (CH,I2) is treated with a source of zinc to produce the Simmons-Smith reagent (ICH,ZnI), which reacts with the alkene in a single elementary step. Complete the mechanism below by adding the necessary curved arrows for this elementary step. Hint: The curved arrow notation is very similar to that for epoxidation involving a peroxyacid.) H, CH2 Enantiomer + Simmons-Smith reagent
Chapter9: Alkynes: An Introduction To Organic Synthesis
Section9.SE: Something Extra
Problem 49AP: Occasionally, a chemist might need to invert the stereochemistry of an alkene—that is, to convert...
Related questions
Question
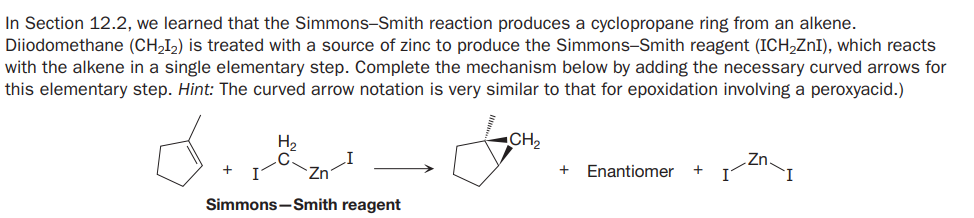
Transcribed Image Text:In Section 12.2, we learned that the Simmons-Smith reaction produces a cyclopropane ring from an alkene.
Diiodomethane (CH,I2) is treated with a source of zinc to produce the Simmons-Smith reagent (ICH,ZnI), which reacts
with the alkene in a single elementary step. Complete the mechanism below by adding the necessary curved arrows for
this elementary step. Hint: The curved arrow notation is very similar to that for epoxidation involving a peroxyacid.)
H,
CH2
Enantiomer
+
Simmons-Smith reagent
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning