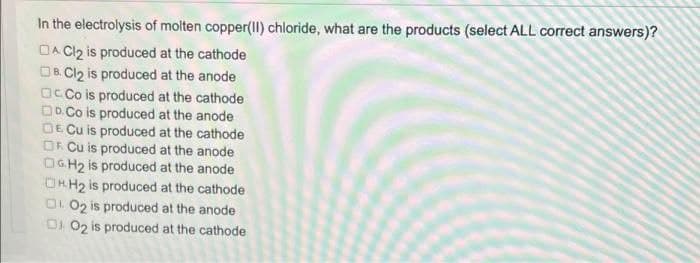
In the electrolysis of molten copper(II) chloride, what are the products (select ALL correct answers)? DA Cl2 is produced at the cathode OB Cl₂ is produced at the anode OcCo is produced at the cathode DD.Co is produced at the anode DE Cu is produced at the cathode OF Cu is produced at the anode OGH₂ is produced at the anode OHH₂ is produced at the cathode 002 is produced at the anode 01 02 is produced at the cathode
Science behind corrosion-test
Corrosion is defined as an activity that transforms refined metals into more chemically stable forms such as oxide, hydroxide, carbonate, or sulfide. It refers to the slow decomposition of things (typically metals); thanks to chemical and/or electrochemical reactions with their surroundings. Corrosion engineering is the science of preventing and controlling corrosion.
Corrosion
Corrosion is defined as an activity that transforms refined metals into more chemically stable forms such as oxide, hydroxide, carbonate, or sulfide. It refers to the slow decomposition of things (typically metals); thanks to chemical and/or electrochemical reactions with their surroundings. Corrosion engineering is the science of preventing and controlling corrosion.
Step by step
Solved in 3 steps with 1 images









