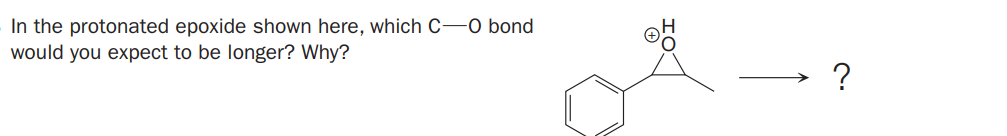
Q: Predict the major product(s) of the following reaction:
A: It is an example of bromination of alkane
Q: From the figure above, what do you expect to observe in these reactions? Which mechanism? Which…
A: It is given that there are three alkyl bromides and two reagents A and B. Which reagent is best for…
Q: Did the overall reaction shown here occur by an Sn2, SN1, E2, or E1 mechanism? How do you know? Draw…
A: Elimination reaction is the reaction in which a small group like water,ammonia is eliminated during…
Q: 7.28 If the anionic species shown were to eliminate a leaving group, the three possibilities would…
A: A group which leaves the molecule and taking the electron pair with it and the electron pair is from…
Q: Draw the major expected product or products of the reaction shown below. If stereochemistry at ar at…
A:
Q: Br H-Br
A:
Q: Which reaction in each of the following pairs would you expect to be faster? (i) Write both…
A: The SN1 reactions are also called the nucleophilic substitution reaction where the attacking…
Q: What is the reagents/conditions to perform the following transformation? Problem viewing the image,…
A:
Q: Draw the complete, detailed mechanism for the addition of hexan-3-ol to dihydropyran and predict the…
A: The given reaction is,
Q: Which one should be considered the major kinetic product of the following reaction?
A: Alkadienes electrophilic addition reactions 1. 1,2-addition : At lower temperature conditions…
Q: For each of the following reactions, draw a complete, detailed mechanism and predict the major…
A: . When aluene react with Bromine in water medium we got addition product . Nucleophite attack…
Q: Provide/ Draw the major product/s of the following reactions
A:
Q: The replacement of bromide ion to the iodide ion as a leaving group in the substitution reaction…
A: In the given problem, the nucleophile attacking is OH- and the leaving groups are Br and I. Since,…
Q: Rank the bold-faced hydrogens for the following compounds from most acidic to least acidic. Explain…
A: Lewis acids are defined as those acids which are able to lose a proton. Acidity is dependent on…
Q: Follow the flow of electrons indicated by the curved arrows in the reaction and predict the product…
A: The possible product for this reaction shown below
Q: 1. Heterolysis ? 2. Coordination involving H,O 3. Proton transfer involving H,0
A: alcohol is formed.
Q: What is the mechanism that leads to the circled, minor product? Is a hydrogen from one of the…
A: The alkyl halides are known for the direct bonding of the halogen to the carbon of the hydrocarbon.…
Q: 9.42 Did the overall reaction shown here occur by an SN2, Sn1, E2, or E1 mechanism? How do you know?…
A:
Q: Draw the curved arrow notation and products for the each elementary step described by the sequence…
A: E2 elimination is elimination reaction where the rate of the reaction depends on both the reactant…
Q: One the molecule below, which hydrogen atom would be abstracted at the fastest rate by a bromine…
A: The molecule given is,
Q: Draw the complete, detailed mechanism for the reaction shown here and predict the major products,…
A:
Q: What is the the resonance contributor(s) for each of the following radicales?
A: we need to draw the resonance structures of the radical given
Q: The requirement for inner sphere mechanism is that one of the reactants is low spin non labile, the…
A: The inner sphere mechanism(ISM) is the mechanism shown by an oxidant and a reductant which forms a…
Q: A thiophene ring is sufficiently activated that bromination may take place without the presence of a…
A: Treatment of alkenes with Br2 gives vicinal dibromides (1,2-dibromides). The bromines add to…
Q: Problem: Consider the addition of HCl shown below. (a) Draw the arrows for the first step of the…
A: Addition of HCl to the alkene result in the formation of haloalkane. In the first step carbocation…
Q: For the reaction below provide all possible product(s) and circle the major product.
A: This reaction is Aldol Condensation reaction in the presence of acid water. You can see details…
Q: Draw the complete, detailed mechanism for each of the following reactions and predict the major…
A: Note - Since you have asked multiple question, we will solve the first question for you. If you want…
Q: Draw the curved arrow notation and products for each elementary step described by the sequence shown…
A: CH3MgBr is Grignard Regent, it is a nucleophilic reagent. They attack the electrophilic carbonyl…
Q: alternate route or reaction? I
A: Your proposed reaction scheme is ok. Only one problem is that F being electron withdrawing group,…
Q: 9.41 Did the following overall reaction occur by an Sn2, Sn1, E2, or E1 mechanism? How do you know?…
A: In this reaction, the Br atom is substituted by the CH3CH2O group. Hence the reaction is a…
Q: Draw the complete, detailed mechanism for the reaction shown here, and explain why nucleophilic…
A:
Q: The following carbocation undergoes a rearrangement by a hydride shift as shown below. The reason…
A: Given, a carbocation which undergoes rearrangement by hydride shift We need to explain the reason…
Q: The reaction shown here is called the pinacol rearrangement. A carbocation rearrangement is believed…
A: When vicinal diols are treated with acids, they can be rearranged to give aldehydes or ketones. This…
Q: Br2 undergoes electrophilic addition to maleic anhydride as shown here. Explain why this reaction is…
A: Here in this compound the C=C double bond is resonance stabilized by two keto group adjacent to…
Q: Which of the listed conditions and nucleophile/s will open the epoxide at carbon A? Which will open…
A:
Q: Explain Please, Thank You: Which of the following substrates can undergo an E2 step with H2N- as the…
A: The reaction occurs via E2-mechanism only if the hydrogen and leaving group are anti to each other…
Q: For the reaction below: OTs KCN DMSO Draw the major organie product(s) in the sketch pad below.
A: OTs is an extremely weak base and a good leaving group. KCN with DMSO leads to cyanation by a…
Q: In the acid-catalyzed aromatic alkylation involving 1-methylcyclohexene and benzene, two isomeric…
A:
Q: Draw the complete, detailed mechanism and predict the major organic product for each of the…
A:
Q: HO Consider the epoxide ring opening reaction shown. Which of the following represents drawn curved…
A: Epoxide ring opening in acidic medium : In the first step oxygen of epoxide ring gets protonated…
Q: Please draw the major product/products of each of the following reaction
A: We will write mechanism of the reaction and estimate the products of this reaction.
Q: Draw the curved arrow notation and product for each of the following when they undergo an SH2 step.
A: It is given a certain reaction of the steps of a free radical mechanism and the curved arrow and the…
Q: What is the complete mechanism using curved arrow formalism of the two products shown below? Explain…
A: Here we are required to find the mechanism for the reaction given
Q: Which of the following reactions in each pair will occur faster?
A: Resonance decreases the nucleophilicity of the compound. strong nucleophile favours SN2 reaction. In…
Q: Which ion in each of the following pairs is better leaving group? Why?
A: Weak bases are good leaving group. They are stable ion. Weaker the bases , better the leaving group.…
Q: Draw the complete, detailed mechanism for the reaction shown here and, using the mechanism, predict…
A:
Q: Draw the product by following the curved arrows. This reaction is an example of a [3,3] sigmatropic…
A: Sigmatropic rearrangement is shifting of sigma bond with the reorganization of pi-bonds in a system.…
Q: Draw both resonance structures of the most stable carbonation intermediate in the reaction shown…
A: No alpha hydrogen increases the stability of carbocation increases.
Q: Which of the following substituents will direct the incoming group in the meta position during…
A: Activating groups :- The electron donating groups present on benzene ring which increase the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Draw a detailed plausible mechanism and all the relevant resonance structures of the reaction below. Make sure to explain the regiochemistry of the product.From the following reactions couples Which of them will go faster ? ExplainDraw a mechanism to account for the reaction shown here, which scrambles the isotopic labeling
- Please explain A, B, and C. Draw the mechanism and the major product(s) of each of the following reactions. Thanks!Please draw the detailed mechanism of this below reaction scheme in 2 steps. Please draw it carefully, I don't understand, for example, why in the first step, the C = O is replaced by C = N and in the second step, the 2 double bonds can react with each other.Draw the curved electron pushing arrow for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bond-making steps
- The reaction shown here is an example of a deiodination. Without AlCl3 present, no reaction occurs. Draw a complete, detailed mechanism forthis reactionWhich nucleophiles will add to the following conjugated enone at the A carbon? Which will add at the B carbon?Draw theproduct of the following reaction.
- Draw all possible alkene products of the following reaction their mechanisms and circle the major productPropose a mechanism for the reaction shown here, which takes place under conditions that favor anDrawing the Product(s) of a Mechanistic Step. For the reactions that have TWO starting materials, indicate/label which species is the nucleophile and which isthe electrophile. Then, by following the curved electron-pushing arrows, draw the product(s) of the following reaction steps. Finally, identify and label what type of elementary step is represented for each mechanism.

