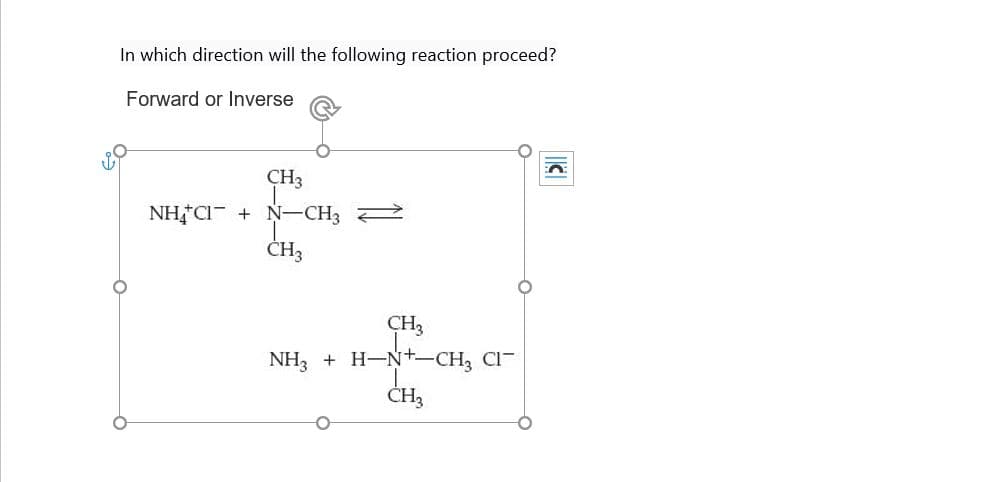
Q: 1. Use curved arrow formalism to propose a mechanism that can explain the formation of the following…
A:
Q: A set of three nucleophilic displacement reactions is shown below: Br Br CH3 CH3CHCH2CH2CH2CH3…
A:
Q: 1. Describe the following chemical reactions as SN1, SN2, E1 & E2. Draw a curved arrow mechanism for…
A: The answer is given below
Q: Provide the ame reaction associated with each reaction below: R-CO-CH3 + N2H/HO" -> Ph-CH2.CH3 Ans…
A: Note: According to our guidelines we are supposed to answer only first three subpart.
Q: Which reaction below give a par of diastereomers and Why? H CH3 CH;COOH А) H CH; HBr B) pегоxides…
A: Diastereomers are stereoisomers that are not mirror images of one another and are non-superimposable…
Q: Which of the following will have the strongest IMFs? CH3OH with H2O CH4 with CH2Cl2…
A: Intermolecular forces (including forces of attraction and repulsion) are responsible for the…
Q: What is the product of the following two-step reaction sequence? Br2 Na CH3C=CCH3 NH3 H CH3 H2N, H3C…
A:
Q: 'hich of the following reagent best accomplish this transformation below? -CH=CH- -CEC- A (1st)Br2,…
A:
Q: Draw the products of each reaction by following the curved arrows. HH + ỘH HỘ: H-C-C-H a. H Br H.…
A: Given,
Q: KMNO4 (CH3)2CHOH Br2/FeBr3 F H heat NaOH LIAIH4 SOCI, CH3O'Na* > K
A: The alkyl group attached with the benzene ring is oxidized into the -COOH group it the key atom…
Q: Rank the following aromatic hydrocarbons below in terms of increasing rates of nitration via…
A: We have to predict rate of electrophilic aromatic substitution.
Q: Which reaction A through D yields hexan-1-ol? Click on a letter A through D to answer. но 1. LIAIH4,…
A: Solution : Organic reduction of carbonyl compounds taken place by a different reducing agent .…
Q: Which of the following anions is the best leaving group in an Sn1 reaction? а. F- b. Но- c. NH2- d.…
A:
Q: dentily the regens necessary FROM BELOW to accomplish each of the following transformation OCH OCH…
A: The reaction will be as follows:
Q: What are the steps to get the answer?
A: In a nucleophilic substitution reaction one nucleophile substitutes the other nucleophile (leaving…
Q: 3. Circle the nucleophile in each pair that undergoes SN2 reaction most rapidly with Ph-CH2-Cl in…
A:
Q: Which of the following is the best method to carry out the following transformation? - OH (a) H₂SO4,…
A: We will draw all the product of each reaction.
Q: Which is not an intermediate or resonance species in the following reaction? A a B b C C D d E e…
A:
Q: estion 6 Select the major product of the reaction sequence below. CI CHNH2 COCNH, 1. (COCI)2 CO,H 2.…
A: In this question, we will Select the correct major product for this reaction. You can see details…
Q: Which of the following is the least reactive towards a nucleophile? O C A Н3С о A Н Н С O B CH3 C…
A:
Q: What Is the mnajor product of this reaction? Click on a letter A through D to answer. 活os %3: CHS…
A: tBuOK is a very strong bulky base that abstracts protons from the less hindered side chain of the…
Q: What will be the major product of the shown E2 reaction: CH3 H3C-C-o CH3 acetonitrile H. Br…
A: The elimination reaction E2 is a bimolecular elimination, which has only one step. A new bond is…
Q: Which compound is a major product of the reaction sequence shown below? (1) benzene COOH ..COOH OCH3…
A: Major product
Q: 2. Rank the following compounds in order of increasing electrophilicity. `NH2 A в D least…
A:
Q: (5 points) Identify the electrophile and the nucleophile in each of the following reaction steps and…
A:
Q: Q6. Draw in all lone pairs and provide the product of each reaction. Use curved arrow notation to…
A: Only three sub parts can be answered pls do repost it.
Q: 1 What is the product of the following sequence of reactions? (1) LİAIH4 PB13 KCN H2O, H*…
A:
Q: Which is the correct order of reactivity towards a nucleophile, from most reactive to least? H3CO F…
A: Given compounds:
Q: CH3 CH3 CH3 H-C-CI CH3-C-CI CH3-C-CI H-C-CI H CH3 H NOP QRS TUV WXY HICIH
A: reactivity of SN2 mechanism decreases on increasing the substituent on the carbon attached to…
Q: 5. The reaction below proceeds through a 4-step mechanism. Classify each step according to the…
A: The arrow in each step has to be provided The each step has to be classified
Q: 1. NaNH2 2. CH3CH21 NH2 А. В. С. D. E. None of the choices is correct. A.
A:
Q: Which of the following is the most stable carbanion? Select one: H NH₂ H O H H :Z:I NH₂ NH₂ NH₂
A:
Q: I-O0 I Complete the curved arrow mechanism for this reaction: H H HO. HO. HO. ОН MeOH Me H. Me Me Me…
A: arrow is shown from filled orbital to unfilled orbital.
Q: .CI NaOH NaNH2 А. В. С. H. `NH2 E. None of the choices is correct. D.
A: Given reaction is : What is the correct product of the reaction = ? Options are :
Q: Q4. The major product(s) of the following E2 reaction is/are: CH CH,ONa Br CH3 E2
A: Biomolecular elimination (E2) reaction:E2 reaction involves a one-step mechanism in which…
Q: 13. Circle the major product of the following reaction (4 points). H,O" / H,0 OH OH он он II II IV
A: At first protonation occurs at alkene carbon to give less stable secondary carbocation which…
Q: :OH H+ terk elimination Q H3O+ heat 1,2-hydride shift Select to Draw Intermediate dissociation…
A: E1 mechanism makes carbocation as an intermediate. Followed by formation of a stable double bond.
Q: Q17. а) Explain the product for each of the reactions below HCI i) TSCI НО ii) рyridine *CH3 Br iii)…
A: Given reactions:
Q: Identify the reagent(s) needed for the following transformation: OH ?. + En 1. BH3 THF 2. H2O2, NAOH…
A: Given: Transformation of a reaction. To find : Best suitable reagents for the transformation.…
Q: 5. Circle the strongest acid in the following group of compounds. PH3 NH3 HS CH4 A В D E 6, Which…
A: Acid is species which can easily give H+ More easily H+ give. More is acidic character
Q: What is the product of the following reaction: N- Br, / FeBr3 ?? Br H O Br H. Br
A: Br2/FeBr3 used forFeBr3 acts as a Lewis acid in this reaction.
Q: Arrange the intermediates below in order of increasing basicity: -c=C -CH=CH -CH;-CH2 a A) a<b<c B)…
A: The basicity depends upon the percentage S character.Higher the S character lesser will be the…
Q: A set of three nucleophilic displacement reactions is shown below: CH3 CH3 CH,CHCH,CH,CCH,CI CH3 CH3…
A: tertiary alkyl halide tends to undergo the SN1 faster than secondary or primary alkyl halide because…
Q: H-S-CH,CH3 (HINT: "S" is a nucleophile ) S-CH2CH3 -S-CH2CH3 C А ОН S-CH2CH3 B D HO CH3CH2-S Select…
A: Here S is a nucleophile,attack at carbon and remove at the oxygen site,so oxygen become -ve charge…
Q: CH3-Br Ag,0 C5H13N N(CH3)3 H20 (heat) (еxcess)
A: Interpretation - To write the reactant in the following reaction sequence which is given as above in…
Q: p) Circle the major product for the following reaction. CH,CH,CI O,N- H2 AICI; O2N- O2N- H2 H2 II…
A: The reaction given is known as Friedel crafts alkylation reaction. In the reactant given, the left…
Q: 4) Rank the following in order of increasing basicity. H3C-O- H2N H20 NH3 A В C a) A < B < D < C b)…
A: Basicity depend upon these factor:- (1) First check Hybridization i.e. % S character is more then…
Q: Which of the following alkyl halides gives the slowest SN2 reaction? CI CH3-CH-CH-CH3 CH3 O…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the mechanism ffrom benzaldehyde to this using: i)NaBH4 ii)TsCl, py iii)NaCN iiii)H+, H2OConsider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:Answer choices for each are the following: unimolecular reaction Br- acetone Cl- substitution reaction elimination reaction CH3OH CH3O- F-
- Draw the favored product(s) of the following reactions (organic chemistry)Which of the following is not a monodentate ligand ? Select one: a. OH- b. H2NCH2CH2NH2 c. H2O d. CN-Match the questions with the correct descriptions below: Sn2, E2 E1cb Sn1, E1 (anti) E2, E1cb Sn1, E1 2 steps poor leaving group 2 carbons removed from a carbonyl group carbanion intermediate 3°>allylic, benzylic>2°>1°>CH₃ carbocation intermediate Ist order reaction (r=k[RX]) H-X anti no intermediate 2nd order reaction (r=k[RX][Nu]) polar aprotic solvent
- Draw the equilibrium chair confirmation of cis1-chloro-3-methylcyclohexane. Interpret the E2 elimination, and draw the structure of the product!In this equilibrium reaction, what is the favored direction? forward, resverse, or equlibrim? CH3CH2CH(OH)CH2CH3 + Na-1CH2CH2CH3 <==>Draw The Major Product Of The Following Reaction. AlCl3 Ay
- Draw the reactant RCHDBr with S absolute configuration, the draw the product of R’OH + RCHDBr (with S-configuration) through an SN2 reactions.What products are formed when each of the following reacts with HO-? a. CH3OH b. +NH4 c. CH3N+H3 d. BF3 e. +CH3 g. AlCl3 d. BF3 f. FeBr3 h. CH3COOHWhich of the following will have the strongest IMFs? CH3OH with H2O CH4 with CH2Cl2 HCl with CH2Cl2 CH3OH with CH2Cl2



