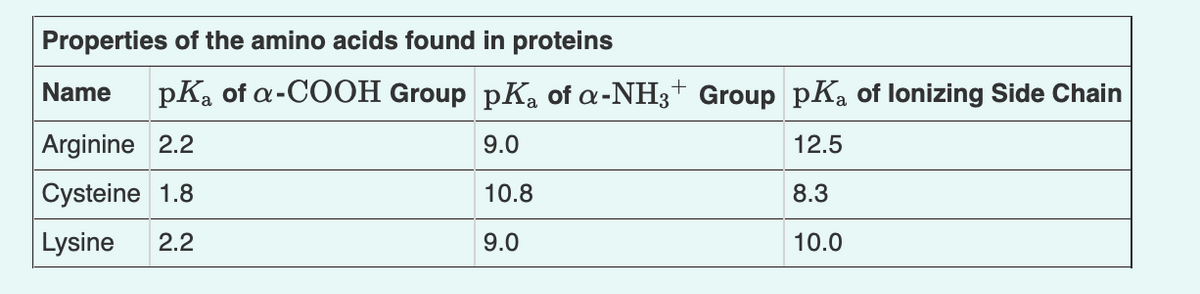
Indicate the net charge of cysteine at pH 9.5
Q: q41 please calculate the unknown concentration of the protein D wih an absorbance value of A412…
A: A linear regression analysis will be required to determine the unknown concentration of Protein D.…
Q: 1. Supposed you used the enzyme lyase in the biomolecular laboratory, identify reaction that you…
A: Enzymes can be described as catalysts of biological origin. Enzymes facilitate the acceleration of…
Q: 852. what is the difference between maltose maltase, sucrose-sucrase and lactoselactase?
A: The body uses carbohydrates for energy. From simple sugars to complex starches, they are found in…
Q: What if C-1 of the oxaloacetate were labeled instead, when would the label be released as CO2?
A: Oxaloacetate is a four-carbon compound, and carbon-1 (C-1) refers to the first carbon atom in the…
Q: A receptor-ligand complex has a dissociation constant of Kd = 20 nM. The rate of receptor-ligand…
A: The reverse rate constant (k-1) is a parameter used in chemical kinetics to describe the rate at…
Q: A patient is suspected of having low stomach acid, a condition known as hypochloridia. To determine…
A: Acids and bases are two chemical substances that react with each other to form a salt and water.…
Q: Sketch a graph showing how the concentration of product P varies with time for…
A: Chymotrypsin is a serine protease. It catalyses the hydrolytic cleavage of the peptide bond next to…
Q: A receptor-ligand complex has a dissociation constant of Kd = 20 nM. The rate of receptor-ligand…
A: The dissociation constant (Kd) for a receptor-ligand complex is related to the rate constants for…
Q: So which form of the arginine side chain is the ionized form? And when the side chain is ionized the…
A: Arginine is an amino acid with a side chain that contains a guanidine group. In its conjugate acid…
Q: HO . -N-H₂₂ OH What is the abbreviation for the fatty acid bond at the number 2 position O…
A: Fatty acids are the components of lipids. A fatty acid is a carboxylic acid with a long aliphatic…
Q: Identify this molecule CH₂OH H HO ОН H nucleic acid saturated fat carbohydrate phospholipid amino…
A: The molecule corresponds to a carbohydrate. It is a simplified representation of a monosaccharide,…
Q: Draw a non-enzymatic mechanism to describe the formation of the imine between retinal and lysine.…
A: Retinal absorbs a photon and this leads to a conformation change. This leads to the formation of an…
Q: Q. Provide two examples of the fate of a metabolite in the pathway
A: Metabolites' fates in pathways are fascinating stories of transformation and purpose in biochemical…
Q: A patient is suspected of having low stomach acid, a condition known as hypochloridia. To determine…
A: Acids and bases are two chemical substances that react with each other to form a salt and water.…
Q: 1.DNA helicase 2.SSBP 3.DNA ligase 4.Primase 5.DNA Polymerase III A. Synthesizes RNA primer B.…
A: Deoxyribonucleic Acid (DNA), is a complex organic molecule is the hereditary material that carries…
Q: 1. In human beings, what is the major control of de novo pyrimidine nucleotide synthesis? A.…
A: Enzymes can be classified as biological catalysts. They enhance the rate of chemical reactions. This…
Q: How do you account for the differences in the amounts of proteins, lipids, carbohydrates, and…
A: Understanding the complex balance of proteins, lipids, carbohydrates, and nucleic acids in a…
Q: The products of the acid hydrolysis of a triacylglycerol (fat) are Select one: a. fatty acids and…
A: When a triacylglycerol (fat) undergoes acid hydrolysis, it is broken down into its constituent…
Q: This is a portion of the periodic table of elements. What is the atomic mass of arsenic (As)? 3 Li…
A: Molecular mass is the total mass of a compound. It is equal to the sum of the atomic masses or mass…
Q: 1. What are at least 2 factors that affect rate of a reaction within a metabolic pathway? Define and…
A: A metabolic pathway is a series of biochemical reactions that occur in a sequence to convert a…
Q: 1. Supposed you want to use phosphoglucomutase to breakdown glycogen. You found out that this enzyme…
A: Enzymes are protein-based biomolecules. They speed up chemical reactions.. Enzymes are extraordinary…
Q: Draw the tripeptide gly-glu-phe with all ionizable groups in their predominant state at pH 7.0.
A: A peptide is a short chain of amino acid residues linked together by a peptide bond. The peptide…
Q: 125. Explain the changes that occur in a G protein as it becomes active and then inactive.
A: G proteins help cell surface receptors send signals to intracellular signaling pathways. Cell…
Q: 2. Comprehend the following statements and identify which one describe ribosomes FALSELY. A. Parts…
A: Ribosomes are intricate cellular organelles. Protein synthesis is heavily reliant on their…
Q: 18. If you make a solution in which the concentration of NaH₂PO4 is 80 mM and the concentration of…
A: pH is the negative logarithm of hydrogen ion concentration in solution. pH measures the acidity or…
Q: Please help! Sketch a titration curve of the peptide Ala-Tyr-Gln-Met-Asp-His from pH=0 to 14 up to 5…
A: Titration is used to determine the amount of acid in a solution.Sodium hydroxide or potassium…
Q: Identify which structures are equivalent to the L-configuration. ☐ CH3 H- H₂N- -NH₂ COOH CH3 -H COOH…
A: In biochemistry, amino acids are the building blocks of proteins, and they come in two different…
Q: Setting up a high throughput system for ultrasound irradiation/sonication?
A: Setting up a high-throughput ultrasound irradiation/sonication system requires several key…
Q: 94. What are the types of non-covalent bonds between the amino acid side chain with the receptor?
A: Molecular interactions between amino acid side chains and receptors underpin many biological…
Q: Choose any/all that apply to the proton-motive force and ATP synthesis. The active pumping of…
A: The active pumping of protons through ATP synthase against their concentration gradient provides the…
Q: Would you expect the pKa to be higher or lower than 8 if the group were buried in the hydrophobic…
A: PKa value is the measure of the tendency of a group to give up a proton. PK a is actually a…
Q: 1. In a catabolic pathway, metabolite X gets converted into metabolite Y, metabolite X metabolite…
A: is the standard change in Gibbs free energy i.e. the change in Gibbs free energy under standard…
Q: Suppose the codon sequence AUGACCCGGCUACUG has a single base pair mutation to AUGACCCGGUUACUG.…
A: Given,Original Codon Sequence: AUGACCCGGCUACUGMutant Codon Sequence: AUGACCCGGUUACUGOriginal Protein…
Q: Which of the following molecules' production is likely to be subject to feedback inhibition in a…
A: Tryptophan synthesis is typically regulated by feedback inhibition in bacteria. This process is…
Q: A monoprotic weak acid, HA, dissociates in water according to the reaction HA(aq) → H+ (aq) + A¯(aq)…
A: A weak acid is an acid that undergoes partial dissociation and produces proton and a conjugate base.…
Q: Select the choice that best describes the stereochemistry of the following amino acid, and rank the…
A: Cysteine is one of the 20 standard amino acids that serve as the building blocks of proteins. It is…
Q: What do you think is the reason why body builders use amino acid to bulk their muscles up? Why do…
A: Essential and non-essential amino acids are the two different categories of amino acids. The body…
Q: 2. DNA Template: TTA - CAT - CAT - ATC - GAT - GAC mrNA: tRNA: Amino acid sequence:
A: All cells divide to give rise to new cells at some specific stage of their life or throughout. The…
Q: +40mV -70mV 2 3₂ OmV 4 ich concet Resting Potential 2 froar points to the dupy Time…
A: Electrical signals are how neurons communicate. Electrical signals are generated when there is a…
Q: 4. Ionic Forms of Aspartic Acid. Aspartic acid is a triprotic acid that can undergo three…
A: Aspartic acid is a triprotic acid, It can give 3 protons from 3 ionizable groups from the carboxylic…
Q: Polypeptides contain two amino acids joined by peptide bonds four amino acids joined by peptide…
A: A peptide bond is a type of chemical bond that forms between the carboxyl group (-COOH) of one amino…
Q: g) Do some research. How does the level of phosphocreatine remain high in muscle cells. Where/how is…
A: Phosphocreatine (PCr) levels in muscle cells are maintained through a process called the creatine…
Q: Select the choice that best describes the stereochemistry of the following amino acid, and rank the…
A: Cysteine is one of the 20 standard amino acids that serve as the building blocks of proteins. It is…
Q: Estimate the apparent slope of the plot for myoglobin. myoglobin apparent slope = Estimate the…
A: The Hill plot is the graph we obtain by plotting at X-axis and at Y-axis. By looking at the value…
Q: Define messenger half-life and how this differs for various messengers based on their…
A: Messenger half-life is the time it takes half of a messenger's molecules to degrade or inactivate.…
Q: 165. Explain why the direction of movement of fluid in the manometer was different during yeast…
A: Respiration, the fundamental process by which living organisms generate energy, has revealed…
Q: 97. Describe the signal transduction mechanisms for enzyme-linked receptors (tyrosine kinases and…
A: Signal transduction converts extracellular signals into intracellular responses. Enzyme-linked…
Q: Q. What would the reverse primer's sequence like if it were intended to bind the area of the DNA…
A: The reverse primer binds to the DNA template's complementary strand from 5' to 3'. If the DNA…
Q: Q. Explain the three main signal transduction mechanisms for GPCRs
A: G-protein-coupled receptors (GPCRs) are transmembrane proteins that transmit signals from the…
Q: Which of the following classes of biological molecules do we commonly use for energy storage?…
A: Energy storage molecules are substances or molecules found in living things that originate with the…
Indicate the net charge of cysteine at pH 9.5
Step by step
Solved in 3 steps with 1 images

- Of the 20 protein-derived amino acids shown in Table 27.1, how many contain Q.) an aromatic ringThe peptide cys-leu-glu-ala-cys-lys is at pH 7 in oxidizing conditions. Part a) Draw the peptide's titration curve. Part b) What would the pI of the peptide be around?Provide for each amino acid the names that will uniquely identify all ionisable groups at pH 11.0 and indicate individual charges associated with these groups and the overall charge of the amino acid. a. Lysine, Leucine, Histidine and Aspartate
- Taurine (2-aminoethanesulfonic acid) is sometimes called an amino acid. (a) Explain why this designation is not valid. (b) From which of the 20 standard amino acids is taurine derived? Describe the chemical change(s) that occurred.Bovine chymotrypsinogen has a molecular massof 25.6 kDa. Amino acid analysis shows that thisenzyme is 4.7% Gly (Mr 75.1).b. Calculate how many glycine residues are present in amolecule of bovine chymotrypsinogenCommercially available high-energy formulas for premature infants are rich in triacylglycerols containing short- and medium-chain fatty acids esterified at the sn-3 position. Why?
- Are triacylglycerols : insoluble in water? very oxidized, contain-glycerol-fatty acids and spingosine, or do they possess a -1charge at 7.0 pH? can you select the true statement?Draw a structural formula for each nucleotide and estimate its net charge at pH 7.4, the pH of blood plasma. Q.) 2'-Deoxyguanosine 5'-diphosphate (dGDP)The peptide aspartyl-glutamyl-leucyl-threonyl-alanine shown in the sketch drawing window, has several ionizable groups. Adjust the charges to show the molecule as it would exist at pH 5.00. (I'm lost, I appreciate any help with explanation. ?)
- identify the amino acid described. state the name, one-letter abbreviation, and draw the structure at pH 12.0 a non-polar essential amino acid capable of donating 1 carbon moiety in metabolismExperimental results describing a protein's amino acid composition are useful for estimating the molecular weight (MW) of the entire protein. A quantitative amino acid analysis reveals that bovine cytochrome c contains 6% alanine (?r 89.1) by weight. Calculate the approximate molecular weight of bovine cytochrome c if there are 6 alanine residues. Please enter your answer with three significant figures. approximate bovine cytochrome c MW: Da Bovine chymotrypsinogen has a molecular weight of 25.6 kDa. Amino acid analysis shows that this enzyme is 11% serine (?r 105). Calculate how many serine residues are present in a molecule of bovine chymotrypsinogen. Round your answer to the nearest whole number. number of serine residues:Calculate the isoelectric point of the tetrapeptide Ser-Leu-Phe-Pro at pH 7.0

