Iron, Fe, will reduce arsenic acid, H3ASO4, to produce highly poisonous arsine gas, AsH3, according to the following equation. 8 Fe*(aq) 3 H3AsOg(aq) + 8 Fe(s) + 24 H*(aq) – 3 ASH3(g) + 12 H20(1) + Molar masses: Fe = 55.85 g mol; H3ASO4 = 141.94 g mol-1 What mass of iron would be required to react with 290 mL of 0.650 mol L¯ H3ASO4(aq)? Enter a mass accurate to 3 significant figures. Number bo
Iron, Fe, will reduce arsenic acid, H3ASO4, to produce highly poisonous arsine gas, AsH3, according to the following equation. 8 Fe*(aq) 3 H3AsOg(aq) + 8 Fe(s) + 24 H*(aq) – 3 ASH3(g) + 12 H20(1) + Molar masses: Fe = 55.85 g mol; H3ASO4 = 141.94 g mol-1 What mass of iron would be required to react with 290 mL of 0.650 mol L¯ H3ASO4(aq)? Enter a mass accurate to 3 significant figures. Number bo
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter11: Chemical Kinetics: Rates Of Reactions
Section11.1: Reaction Rate
Problem 11.3CE
Related questions
Question
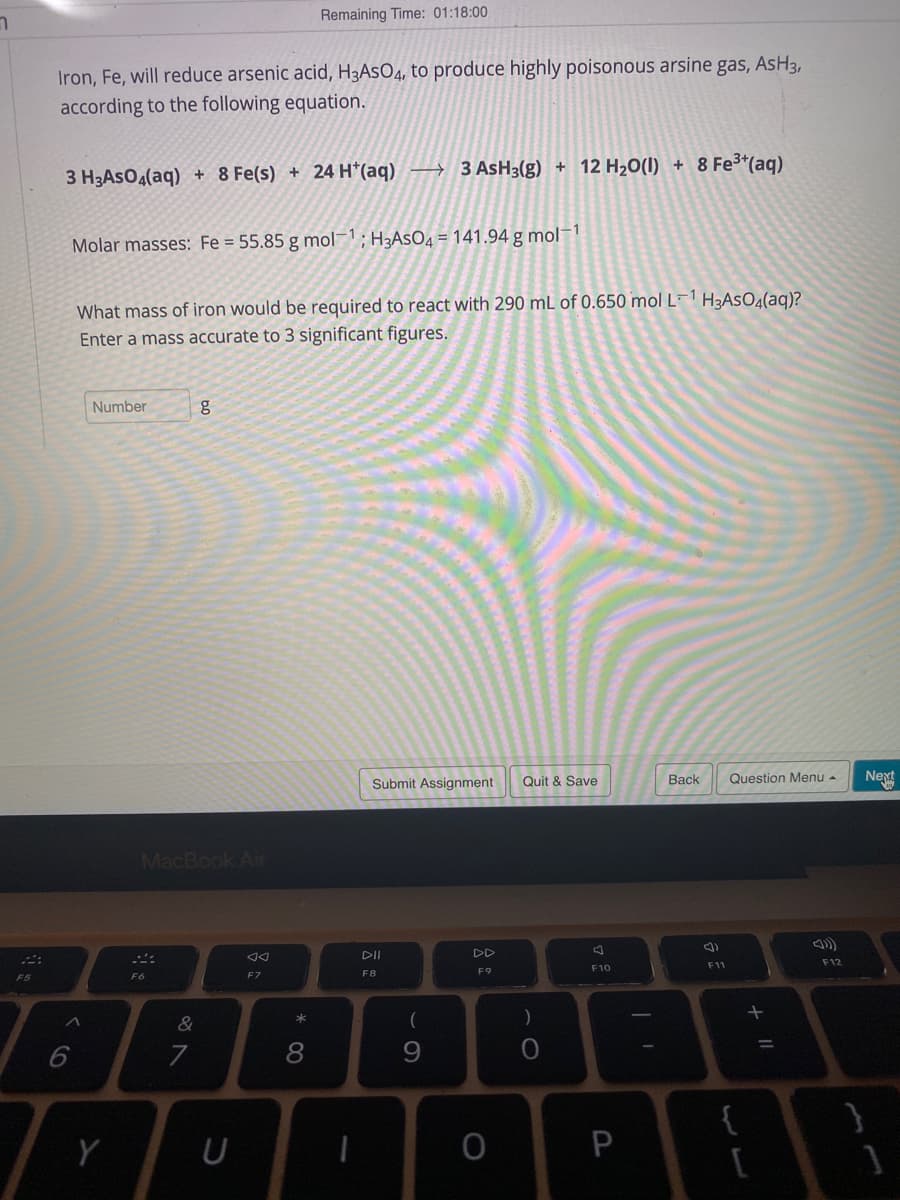
Transcribed Image Text:Remaining Time: 01:18:00
Iron, Fe, will reduce arsenic acid, H3ASO4, to produce highly poisonous arsine gas, AsH3,
according to the following equation.
8 Fe3*(aq)
3 H3ASO4(aq) + 8 Fe(s) + 24 H*(aq) → 3 ASH3(g) + 12 H20(I) +
Molar masses: Fe = 55.85 g mol; H3ASO4 = 141.94 g mol-1
What mass of iron would be required to react with 290 mL of 0.650 mol L¯1 H3ASO4(aq)?
Enter a mass accurate to 3 significant figures.
Number
Submit Assignment
Quit & Save
Вack
Question Menu -
Next
MacBook Air
DII
DD
F12
F11
F10
F8
F9
F6
F7
*
7
8
9.
{
P
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole