IUOIUII Tate varies with pressure, concentratio. Coal gasicacion produces an easily-transportable, clean-burning combination of carbon monoxide and hydrogen from coal by reacting it with C(s)+H,0(g) → Co(g)+H,(g) Suppose an engineer decides to study the rate of this reaction. He prepares four reaction vessels with 94.3 g of solid carbon and 25.2 g of ste. volume and temperature of each vessel is shown in the table below. Arrange the reaction vessels in decreasing order of initial rate of reaction. In other words, select a "1" next to the vessel in which the engineer expect the initial rate of reaction to be highest, a "2" next to the vessel in which the initial rate of reaction would be next highest, and so on.
IUOIUII Tate varies with pressure, concentratio. Coal gasicacion produces an easily-transportable, clean-burning combination of carbon monoxide and hydrogen from coal by reacting it with C(s)+H,0(g) → Co(g)+H,(g) Suppose an engineer decides to study the rate of this reaction. He prepares four reaction vessels with 94.3 g of solid carbon and 25.2 g of ste. volume and temperature of each vessel is shown in the table below. Arrange the reaction vessels in decreasing order of initial rate of reaction. In other words, select a "1" next to the vessel in which the engineer expect the initial rate of reaction to be highest, a "2" next to the vessel in which the initial rate of reaction would be next highest, and so on.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter12: Chemical Kinetics
Section: Chapter Questions
Problem 96AE: Iodomethane (CH3I) is a commonly used reagent in organic chemistry. When used properly, this reagent...
Related questions
Question
100%
Please give me the right answer
Transcribed Image Text:Varles with préssure, concentratio...
Esther v
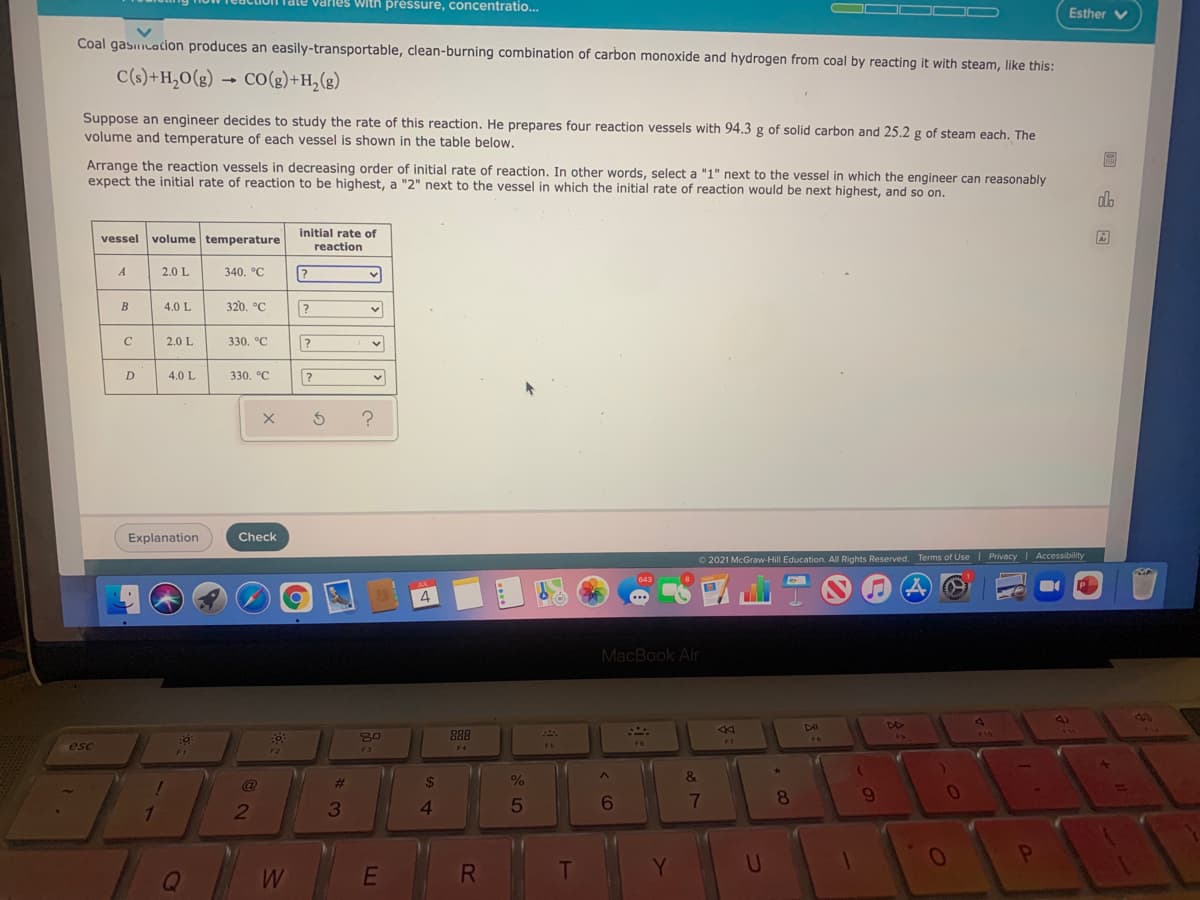
Coal gasicacion produces an easily-transportable, clean-burning combination of carbon monoxide and hydrogen from coal by reacting it with steam, like this:
C(s)+H,0(g) → Co(g)+H,(g)
Suppose an engineer decides to study the rate of this reaction. He prepares four reaction vessels with 94.3 g of solid carbon and 25.2 g of steam each. The
volume and temperature of each vessel is shown in the table below.
Arrange the reaction vessels in decreasing order of initial rate of reaction. In other words, select a "1" next to the vessel in which the engineer can reasonably
expect the initial rate of reaction to be highest, a "2" next to the vessel in which the initial rate of reaction would be next highest, and so on.
do
initial rate of
vessel volume temperature
reaction
A
2.0 L
340. °C
B
4.0 L
320, °C
C
2.0 L
330. °C
D
4.0 L
330. °C
Explanation
Check
O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use I Privacy Accessibility
643
4
*..
MacBook Air
21
DAI
DD
80
888
10
F7
FR
F6
esc
F4
F5
F3
&
%23
2$
7
8
2
W
RI
Y
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning