Q: Br -OCH3 Нeat
A:
Q: 5. Show the mechanism for the Syl reaction between (2R,3R)-2-bromo-3-methylpentane and water.
A:
Q: For 1-(1-methylpropyl)cyclohexane can you show me the mechanisms for the formation of the major…
A: A question based on bromination reaction, which is to be accomplished.
Q: 4. For the multistep synthesis below а. Draw a complete mechanism for this synthesis. b. Predict the…
A:
Q: How many elimination product (s) would you anticipate to form from the reaction below? Br CH,ONa…
A: In the elimination reaction, the base abstracts the hydrogen that is present at beta carbon. So, the…
Q: NC-CH=CH-CN N. CH CH2 CH 3 76 ROOC -C=C-COOR
A: The organic compounds in which carbon atoms are bonded to each other by a double bond or by a triple…
Q: 1. Give the expected Zaitsev and anti-Zaitsev products in the following reactions. Label the Zaitsev…
A: See both type of products in red boxes.
Q: 5. For each of the following molecules, (i) state whether ortho-para or meta substitution will occur…
A:
Q: 1. Show the complete mechanism and product for the transformations below. Show the arrow pushing for…
A:
Q: Describe stereoselective, regioselective, or chemoselective, the reactions below.
A: In case one we write the most stable structure in chair form, now their is two possibility to attack…
Q: For the following reactions below: Give the major product of the reaction. Be specific with stereo-…
A:
Q: 1. Rank the following substrates in order of increasing reactivity in the Suzuki coupling, from most…
A: Rates of Suzuki reaction depends on the transmetallation steps where Halides is replaced by the…
Q: Propose a reasonable mechanism for each of the following transformations. Show clearly the…
A:
Q: .3 Indicate if the following reactions proceed through an S1 or SN2 mechanism. Draw the substitution…
A:
Q: 1. Predict the number of reactive sites that will occur if this compound is reacted with KMNO4 with…
A:
Q: Please identify the most likely product emanating from this series of reactions. 1 Cl2 NaOH
A: ->Addition of Cl2 to alkene give trans alkene which acts as dienophile and give Diels-alder…
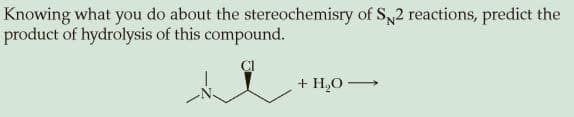
Q: Predict the main mechanism that occur for the following reaction ÇI t-Bu acetone, 50 °C O SN2 SN1 E1…
A:
Q: 5,0<o H20 + NHtz
A:
Q: 4. Sometimes protected species are not as "inert" as we think they might be. Provide a mechanism for…
A:
Q: Give the chemical mechanism and the major product for the following reaction using the curved arrow…
A:
Q: 6) Draw the major organic products generated in the reactions below. Pay particular attention to…
A:
Q: Why do we not see 1,2-hydride shift rearrangements (or 1,2-methyl shift rearrangements) for E2…
A: A reaction which involve removal of a molecule containing atoms or groups give at least one p bond…
Q: Give the chemical mechanism and the major product for the following reaction using the curved arrow…
A: c. This is an example of Hydroboration-Oxidation reaction. Terminal alkyne on…
Q: Which is the best method for carrying out the following two-step transformation? CI 1. O 1.…
A: The given reactant is Benzene and the product formed in the end is 3-chloro acetophenone.
Q: Predict the main mechanism that occur for the following reaction * to MeO MEOH, 50 °C Br O E2 O E1 O…
A: The question is based on the concept of organic reactions. We have to identify the mechanism taking…
Q: Would you expect the reaction below to proceed in conrotatory or disrotatory fashion CH3 CH3 hu CH3…
A: Electrocyclic reactions involves the rotation of bonds in such a manner that the HOMO-LUMO…
Q: Consider the following reactions below: ... Mg ether 1. ether CH, 2. H30 Br IV Compound IV will most…
A:
Q: an S1 reaction
A:
Q: 9) Using curved arrows to indicate electron movement, propose a mechanism tor the following…
A:
Q: 1 eq Br2 ? CCI4 H Br Br Br Br H. Br Br Br Br Br Br Br Br I II III IV V
A: Treating alkynes with dibromide yields vicinal 1,2-dibromides. Carbon tetrachloride is a common…
Q: HO. CH2-NEN: + N2 (CH, =R=R)
A:
Q: 4. Write the product and give mechanism. (i) *CO (ii) RCN/.. -CH3 PMe Rh Me,P
A:
Q: Write mechanisms for each of the following reactions, and carefully indicate general acid / general…
A: Reaction - 1: This reaction is a typical example of aldol condensation that takes place in presence…
Q: Please draw the product below. You need to show stereochemistry using dashes and wedges. OH \CO2CH3…
A: In organic conversions, appropriate reagent converts the reactants into products. Reagents attack…
Q: 4) Propose the reasonable mechanisms for the following reactions. i) CH2CH3 OH + HBr Br-CH2CH3
A: In the given reaction the ether is reacted with HBr to form phenol and ethyl bromide. The reaction…
Q: Indicate the correct set of reactions from the choices below that would accomplish the following…
A: In epoxide ring opening, if there's an acid then the nucleophile attack the ring from the more…
Q: Identify the nucleophile that attacks the carbocation intermediate in the acid-catalyzed hydration…
A:
Q: Propose a reasonable mechanism for the following reactions. HO a. 1) excess CH,MgBr OH 2) H,O b. OH…
A:
Q: Predict the main mechanism that occur for the following reaction * CI t-Bu. acetone, 50 C SN2 O E1…
A: In this reaction we will write the type of the reaction
Q: B-Discuss and clarify the mechanism of this reaction (3 degrees) H* [Co(NH₂)+ [CH₂O) [CoH₂O),+ [C:…
A:
Q: predict the product of this reaction! C₂H₅COCH₃ + CH₃OH
A: Carbonyl compounds react with methanol to form hemiacetal and acetal in presence of an acid…
Q: what is the mechanisnm of these reactions? LiAIH, /THF 2) H30 HO CH3OH /AC HCL, Hz O pells HO…
A: The given reaction is reduction of ester and aldehyde by LiAlH4 followed by hydrolysis or workup.and…
Q: Write the mechanism and predict the most likely product in the following reaction NaOH ICH2CH3…
A:
Q: Draw the structure of compound P and discuss the mechanism of the reaction of compounds N and O to…
A:
Q: redict the major products of the following reactions. If it is possible, write all stereoisomers.…
A: a) Alkenes reacts with water in presence of acid and undergoes addition reaction via carbocation…
Q: CH,OH -Br OCH3 reflux OCH3
A:
Q: 7. The reactions shown below will NOT occur as written. Explain why (briefly), and suggest a ix (A…
A: T he above reaction is the example of oxidation of alcohol in the presence of cro3 which lead to…
Q: 10. According to the 1H NMR spectrum of the product, which of the following is a major product of…
A:
Q: 4. Study the curved arrows in the reactions below and provide the structures of the product(s)…
A:
Q: HCl a. 40.°C HCl 40 °C b.
A:
Step by step
Solved in 2 steps with 1 images

- Describe stereoselective, regioselective, or chemoselective, the reactions below.Conformation control of reaction mechansims. Consider the reaction below: Provide all possible elimination products from this reaction and identify the major elimination product. Provide mechanisms (with curved arrows) that clearly explain the reaction outcome.Arrange the following reactions in order of increasing ΔrxnS and briefly explain your reasoning. a) S(s)+O2(g)⟶SO2(g) b) H2(g)+O2(g)⟶H2O2(ℓ) c) CO(g)+3H2(g)⟶CH4(g)+H2O(ℓ) d) C(s)+H2O(g)⟶CO(g)+H2(g)
- Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpArrange the following reactions in order of increasing ΔrxnS and briefly explain your reasoning.a) S(s) + O2(g) ⟶ SO2(g)b) H2(g) + O2(g) ⟶ H2O2(ℓ)c) CO(g) + 3 H2(g) ⟶ CH4(g) + H2O(ℓ)d) C(s) + H2O(g) ⟶ CO(g) + H2(g)Arrange the following reactions in order of increasing ΔrxnS and briefly explain your reasoning. a) S(s) + O2(g) ⟶ SO2(g)b) H2(g) + O2(g) ⟶ H2O2(ℓ)c) CO(g) + 3 H2(g) ⟶ CH4(g) + H2O(ℓ)d) C(s) + H2O(g) ⟶ CO(g) + H2(g)
- Propose a plausible mechanism for the 1 to 2 conversion as shown, and explain the stereochemical result:predict the main product for the addition of 1 equivalent of HX to the following compounds and write the mechanism of the reactionPropose mechanisms compatible with 1) and 2) of the following reactions.
- Considering the following reaction, while kinetic resolutions limit your yield of, for example, acetate 3 to 50%, in certain cases this can be improved by using a sequence of reactions that convert the undesired enantiomer to the desired one. Propose a series of such reactions that would allow you to convert the (S)-2 to acetate 3. Thank you :)The E1 mechanism (unimolecular elimination) of Elimination ?The diasteromers of 4-t-butylcyclohexanol that you make can be distinguished by the splitting patterns of the proton attached to the carbon bearing th OH group. This is due to the fact that the H that is axial is a triplet of triplets because it is coupled to to sets of non-eqivalent protons, while the equatorial H is coupled to 4 equivalent hydrogens and is a pentet. True? False? Please answer very soon will give rating surely Complete explaination needed

