Laboratory 1: Radioactive Decay Lab Table 1 Number of Un- Number of Number of Half- Total Time Passed decayed Atoms of Decayed atoms of M&Miums lives (seconds) M&Miums (number of shake rounds) O seconds X = 46 10 seconds 30 16 First 10 s + Second 10 s = 2 20 seconds 3 30 16 4 40 13 50 4 60 7 3 70 8. 80 9. 40 10 100 Analysis 1. Plot graph the number of un-decayed atoms vs. half-life for M&M atoms using graph paper. 2. Calculate the fraction of the atoms had not decayed at the second end half-lives.
Laboratory 1: Radioactive Decay Lab Table 1 Number of Un- Number of Number of Half- Total Time Passed decayed Atoms of Decayed atoms of M&Miums lives (seconds) M&Miums (number of shake rounds) O seconds X = 46 10 seconds 30 16 First 10 s + Second 10 s = 2 20 seconds 3 30 16 4 40 13 50 4 60 7 3 70 8. 80 9. 40 10 100 Analysis 1. Plot graph the number of un-decayed atoms vs. half-life for M&M atoms using graph paper. 2. Calculate the fraction of the atoms had not decayed at the second end half-lives.
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter9: Nuclear Chemistry
Section: Chapter Questions
Problem 9.51P
Related questions
Question
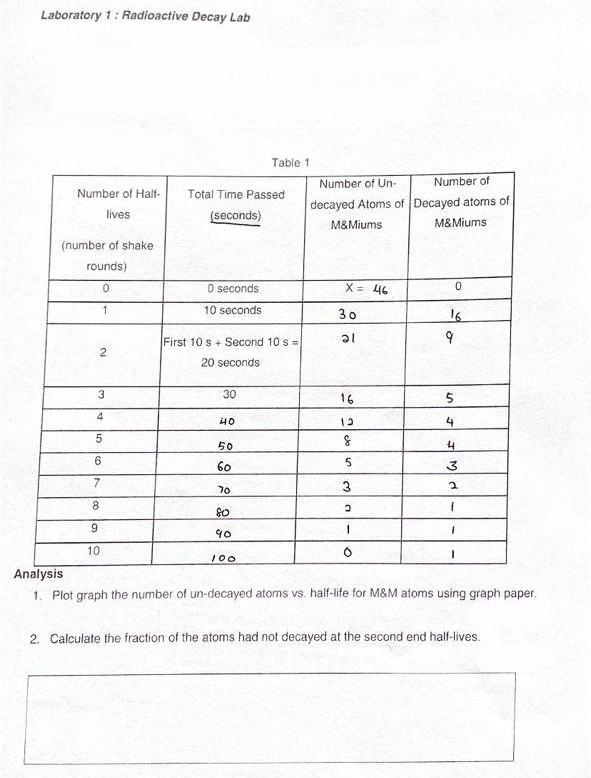
Transcribed Image Text:Laboratory 1: Radioactive Decay Lab
Table 1
Number of Un-
Number of
Number of Half-
Total Time Passed
decayed Atoms of Decayed atoms of
M&Miums
lives
(seconds)
M&Miums
(number of shake
rounds)
O seconds
X = 46
10 seconds
30
16
First 10 s + Second 10 s =
2
20 seconds
3
30
16
4
40
13
50
4
60
7
3
70
8.
80
9.
40
10
100
Analysis
1. Plot graph the number of un-decayed atoms vs. half-life for M&M atoms using graph paper.
2. Calculate the fraction of the atoms had not decayed at the second end half-lives.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning