Lattice Energies for Some Ionic Compounds TABLE 8.1 Lattice Energy (kJ/mol) Compound Lattice Energy (kJ/mol) Compound 1030 LiF MgCl2 2526 LİCI 834 SrCl2 2127 Lil 730 NaF 910 MgO 3795 NaCl 3414 788 Cao NaBr 732 Sro 3217 Nal 682 KF 808 ScN 7547 КСІ 701 Kвг 671 CSCI 657 600 CsI
Lattice Energies for Some Ionic Compounds TABLE 8.1 Lattice Energy (kJ/mol) Compound Lattice Energy (kJ/mol) Compound 1030 LiF MgCl2 2526 LİCI 834 SrCl2 2127 Lil 730 NaF 910 MgO 3795 NaCl 3414 788 Cao NaBr 732 Sro 3217 Nal 682 KF 808 ScN 7547 КСІ 701 Kвг 671 CSCI 657 600 CsI
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter9: Chemical Bonds
Section: Chapter Questions
Problem 9.4QE: What main factors control the magnitude of lattice energies? Give a specific example of a compound...
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
Based on data in Table 8.1, estimate (within 30 kJ/mol) the
lattice energy for (a) LiBr, (b) CsBr, (c) CaCl2.
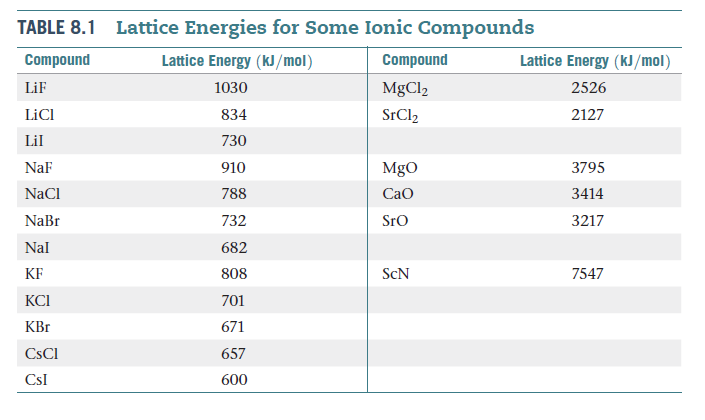
Transcribed Image Text:Lattice Energies for Some Ionic Compounds
TABLE 8.1
Lattice Energy (kJ/mol)
Compound
Lattice Energy (kJ/mol)
Compound
1030
LiF
MgCl2
2526
LİCI
834
SrCl2
2127
Lil
730
NaF
910
MgO
3795
NaCl
3414
788
Cao
NaBr
732
Sro
3217
Nal
682
KF
808
ScN
7547
КСІ
701
Kвг
671
CSCI
657
600
CsI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning