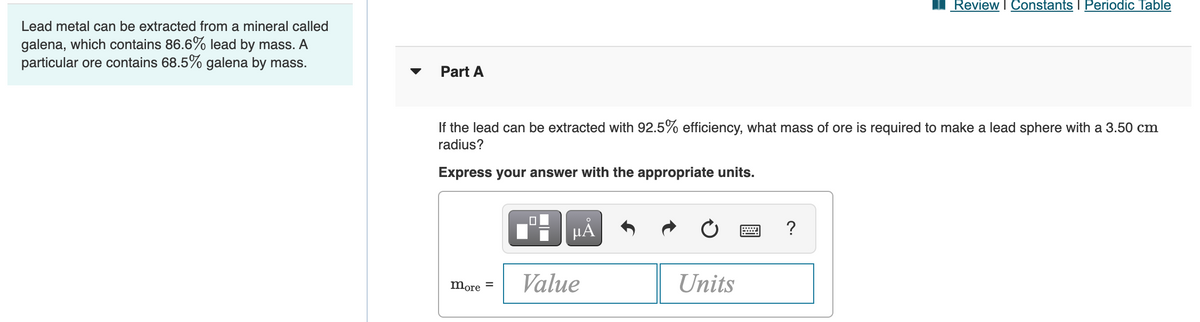
Lead metal can be extracted from a mineral called galena, which contains 86.6% lead by mass. A particular ore contains 68.5% galena by mass. Part A If the lead can be extracted with 92.5% efficiency, what mass of ore is required to make a lead sphere with a 3.50 cm radius? Express your answer with the appropriate units. HA ? Value Units more =
Lead metal can be extracted from a mineral called galena, which contains 86.6% lead by mass. A particular ore contains 68.5% galena by mass. Part A If the lead can be extracted with 92.5% efficiency, what mass of ore is required to make a lead sphere with a 3.50 cm radius? Express your answer with the appropriate units. HA ? Value Units more =
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 7P
Related questions
Question
Please answer question 23 part A
Transcribed Image Text:Review I Constants I Periodic Table
Lead metal can be extracted from a mineral called
galena, which contains 86.6% lead by mass. A
particular ore contains 68.5% galena by mass.
Part A
If the lead can be extracted with 92.5% efficiency, what mass of ore is required to make a lead sphere with a 3.50 cm
radius?
Express your answer with the appropriate units.
HA
?
more =
Value
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

