Q: SECH3 CH3 CH3 SECH3 CH3 H3C H3C H3C. `SECH3 SeCH3 HзC. SECH3 ČH3 CH3 ČH3 A SECH3 В C H3C CH3
A: The given molecule is: The molecule can be converted into various forms of its chair conformations.…
Q: Which of the following is the lowest energy conformer of the following compound looking down the…
A: 1St option is correct.
Q: Draw (a) a Newman projection of the most stable conformation sighting down the C-3 C-4 bond and (b)…
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: What are the chair conformations of chlorocyclohexane and their IUPAC name using R/S or cis-trans?
A:
Q: a) How many degrees of unsaturation (double bond equivalents) are represented by the molecular…
A:
Q: Draw Newman Projections of the following molecule, looking down the C2-C3 bond. Under A, draw the…
A: Generally Anti conformer is most stable and eclipse conformer is least stable
Q: CI
A:
Q: Which is the most stable chair conformer of the following compound? ON
A: For the chair conformations, the most stable conformer is the one in which the bulkier group…
Q: Which Newman projection is the most stable conformed for (S)-4-chlorobutan-2-ol?
A:
Q: 2-methylcyclohexanol
A:
Q: Draw a newman projection of the given compound E viewed from C2-C3 oriented according to the first…
A: In conformational analysis, eclipsed conformers that is having dihedral angle is =0. they faces…
Q: Part 2. In Box 1, use a Newman projection to draw the lowest energy conformation about the C3-C4…
A:
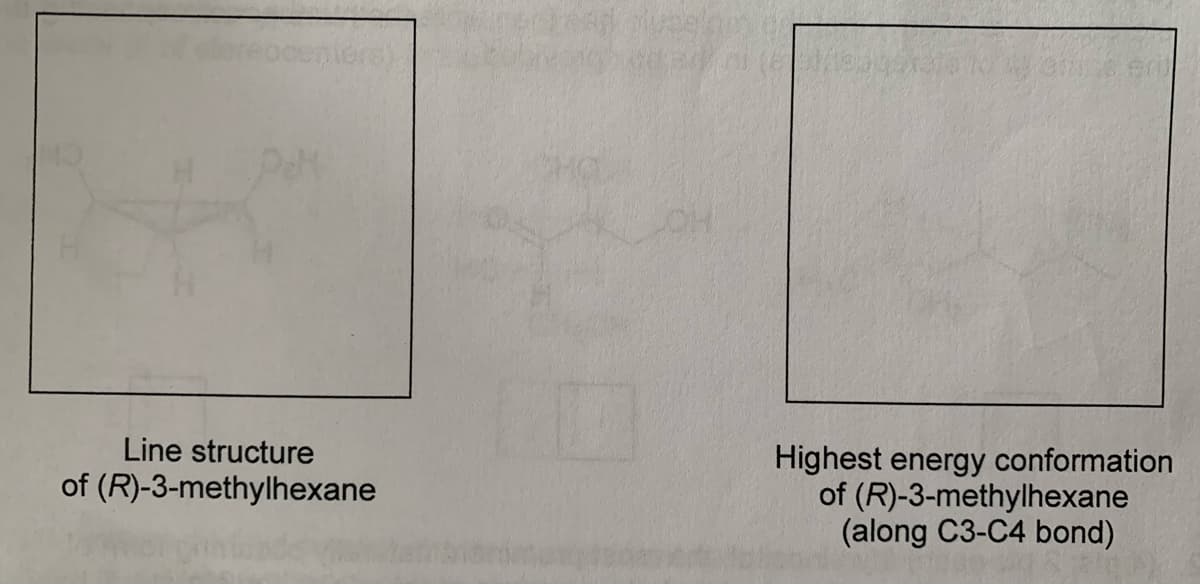
Q: In the space provided below, write a line structure corresponding to the systematic name…
A: Conformational analysis of 3-methylhexane . Stability based on steric hindrance.
Q: Which Newman projection demonstrates the least stable conformation of the C3-C4 bond of…
A: To find: Newman projection that demonstrates the least stable conformation of the C3-C4 bond of…
Q: HỘ, Но In its most stable chair conformation, indicate the orientation of each of the labeled groups…
A: The given chair for is the most stable structure of trans 1,4-dihydroxycyclohexane. Here both the OH…
Q: How many energetically different eclipsed conformations does 1-bromo-2-methylpropane, viewed along…
A: The given Compound is 1-bromo-2-methylpropane. To find: The number of eclipsed conformation viewed…
Q: Provide the most stable (lowest energy) chair conformation of cyclohexan-1-ol Drawing
A: Given Molecule is cyclohexan-1-ol Cyclohexan-1-ol is more stable in chair form.
Q: Draw the most steble cheir conformation of the following cyclohexane.
A: Stability of the cyclohexane mainly depends on syn 1,3- diaxial interaction. Among the conformer…
Q: Draw the Newman conformations of higher and lower energy of (S)-3-methylhexane around the C3-C4 bond…
A:
Q: Which Newman projection for the conformation of 2-methylbutane will have energy Il on the potential…
A:
Q: Draw each compound in its most stable conformation(s). Then draw it in its most symmetric…
A: Given:- The given molecule is trans-1,2-dibromocyclohexane. This cyclohexane has two…
Q: Draw Newman projection: a) for the least stable conformation of 3- Methylbutane, view 2-3 bond b)…
A: Eclipsed conformation has a greater potential energy due to torsional strain, it is less stable than…
Q: Which of these shows the s-trans conformation of (2Z,4Z)-3-ethyl-2,4-hexadiene?
A:
Q: How many alkyl substituents are axial in the lowest energy conformation?
A: Conversion of simple ring presentation to chair form: The groups pointing towards the observer or…
Q: Is the 1st compound ( more, less or equally) stable than B? Pls show through illustrations of chair…
A:
Q: Explain "Gauche conformations are generally higher in energy than anti conformations because of…
A: The stability of any conformation depends upon their energies as higher is the energy ,lesser is…
Q: What structure has the lowest energy?
A: For trans-1,3-substituted cyclohexane, the substituents should be in (a,e) or (e,a) positions to…
Q: Draw the most stable conformation of (a) ethylcyclohexane
A: Cyclohexane exists as two chair conformations that are in equilibrium at room temperature. In…
Q: 5. Please draw out the confirmation of alicyclic compounds for molecular formula C6H12
A: The different conformations for an alicyclic compound having molecular formula = C6H12 can be drawn…
Q: (f) a Newman projection of the following molecule looking down the C2-C3 bond in the anti-…
A: Newman projection visualizes the conformation of a chemical bond from front to back, with the front…
Q: Draw the structure of following compound. (2E,4E)-3-methylhexa-2,4-diene in the s-cis conformation
A: The representation of arrangement of atoms, group of atoms and bonds (double or triple) in the…
Q: Which Newman projection for the conformation of 2-methylbutane will have energy II on the potential…
A:
Q: (e)
A: Complete solution is given below in next step with these conformation.
Q: Draw cis,cis trans-1,2,3-trimethylcyclohexane in its most stable chair conformation and Newman…
A: Given :
Q: In the space provided below, write a line structure corresponding to the systematic name…
A: In line structure we use bond line notation.
Q: Draw the structure of following compound. (Z)-penta-1,3-diene in the s-trans conformation
A: For conjugated dienes the two double bonds are separated by a sigma bond. There is a possibility…
Q: What are the degrees of unsaturation in the following molecule?
A: To find the degree of saturation of any molecule we have the following formula:-
Q: Which Newman projection is the most stable conformer for (S)-4-chlorobutan-2-ol?
A: In this question, we will draw the t stable conformer for the given compound. First we will draw…
Q: Name and draw ALL possible stereoismers of 1,3-dichlorocyclobutane.
A: Given molecule is, 1,3 -dichloro cyclobutane.
Q: Draw the most stable chair conformation of the cis-1-Ethyl-2-methylcyclohexane and estimate the…
A: The two possible chair confirmation of cis-1-Ethyl-2-methylcyclohexane are as follows: Among the…
Q: Draw the chair conformations for cis-1-bromo-3-ethylcyclohexane and circle the most stable…
A:
Q: Which of (a)-(d) is the most stable conformation? CH3 H- H CH2CH3 CH3 a) CH3 CH3YYH H CH2CH3 b) CH3
A: We can determine which Newman projection is most stable from the position of bulky groups. We can…
Q: When you obtained a geometry optimized structure of methyleyclopentane, did you determine the energy…
A: Methylcyclopentane exist in two conformation. Cs Envelope and Cn Half chair.
Q: Molecular Formula: CHgO3 Compute for the DU (degree of unsaturation) Then draw the structure of the…
A: The degree of unsaturation or Double bond equivalence of any organic system represented by their…
Q: Draw an energy diagram for the conformations of ethane during a full 360° rotation of one METHYL…
A: In this question, we will draw the energy diagram for the conformation of ethane during a full 360°…
Q: Draw the most stable chair conformation of the following cyclohexane.
A: The most stable conformation of cyclohexane is formed by placing bulky molecules at the equitorial…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Fill in the blanks: cis-1,3-Dimethylcyclohexane has two different chair conformations: one withboth methyl groups in __________ positions and one with both methyl groups in ____________ positions.What are the chair conformations of chlorocyclohexane and their IUPAC name using R/S or cis-trans?translate the bond-line notation structure to the Newman projection by filling int the missing groups (A, B, C, D or E) on the lines in the Newman projection so they match the conformation given in the original structure. Circle if the conformation is a staggered or eclipsed.
- In the lowest energy chair conformation of cis-1,3-dimethylcyclohexane, how many axial positions are occupied by methyl groups?Draw most stable chair conformation for cis-1-tert-butyl-4-methylcyclohexane in the image below.Draw the highest and lowest energy conformations. In cases where two or three conformations are degenerate, choose only one as your anwser. Use Pr, Me, H, Et in the structures.
- a. Draw Newman projections of the two conformers of trans-1,3-dimethylcyclohexane. b. Which of the conformers predominates at equilibrium?What are the chair conformations of 1,2-dichlorocyclohexane and their IUPAC name using R/S or cis-trans?Is the 1st compound ( more, less or equally) stable than B? Pls show through illustrations of chair conformations
- a) i) Draw the Newman projections for the conformations of ethane (staggered and eclipsed) Which is higher in energy? Which is more stable? Next to each conformer, draw the equivalent ‘sideways’ view, using wedged bondsDraw two possible chair conformation of Trans-1,3-dihydroxyclohexane they differ energy?Draw the most stable conformation

