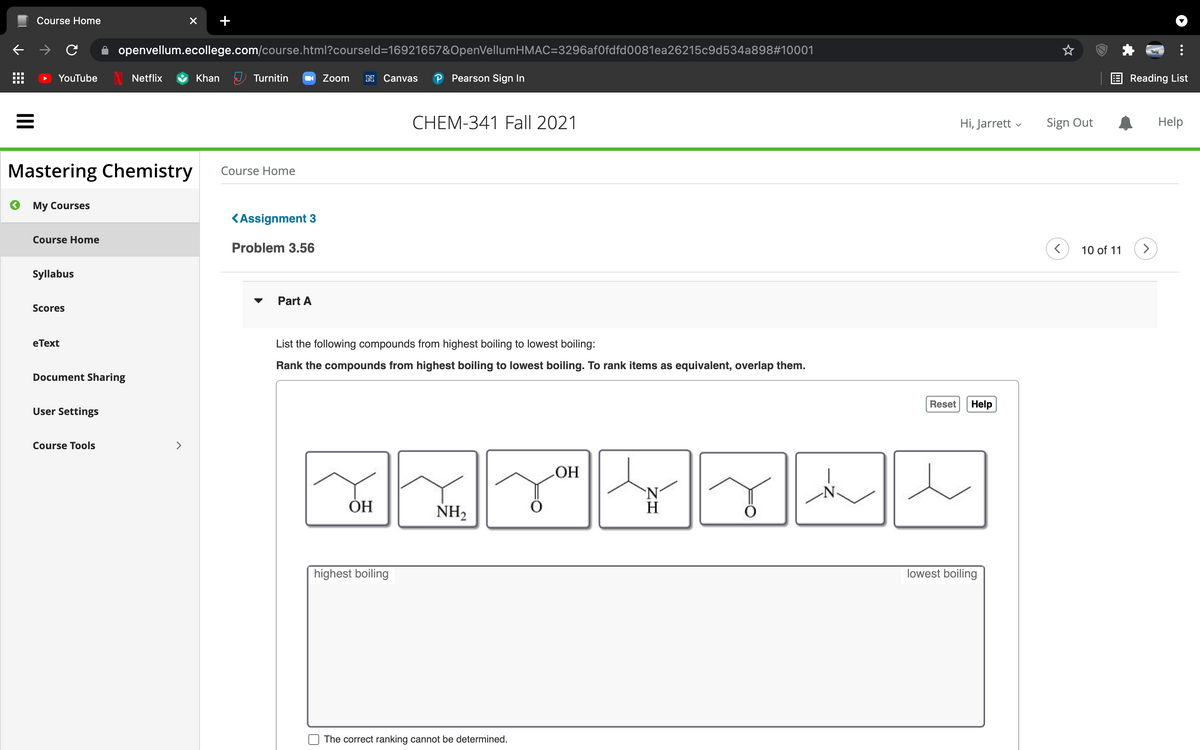
List the following compounds from highest boiling to lowest boiling: Rank the compounds from highest boiling to lowest boiling. To rank items as equivalent, overlap them. Reset Help HO ОН `N H NH2 highest boiling lowest boiling
List the following compounds from highest boiling to lowest boiling: Rank the compounds from highest boiling to lowest boiling. To rank items as equivalent, overlap them. Reset Help HO ОН `N H NH2 highest boiling lowest boiling
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Transcribed Image Text:Course Home
openvellum.ecollege.com/course.html?courseld=16921657&OpenVellumHMAC=3296af0fdfd0081ea26215c9d534a898#10001
YouTube
N Netflix
Khan ) Turnitin
O Zoom
N Canvas
Pearson Sign In
E Reading List
...
CHEM-341 Fall 2021
Hi, Jarrett
Sign Out
Help
Mastering Chemistry
Course Home
My Courses
<Assignment 3
Course Home
Problem 3.56
10 of 11
>
Syllabus
Part A
Scores
eТext
List the following compounds from highest boiling to lowest boiling:
Rank the compounds from highest boiling to lowest boiling. To rank items as equivalent, overlap them.
Document Sharing
Reset
Help
User Settings
Course Tools
>
OH
N-
ОН
`N'
H
NH2
highest boiling
lowest boiling
The correct ranking cannot be determined.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

