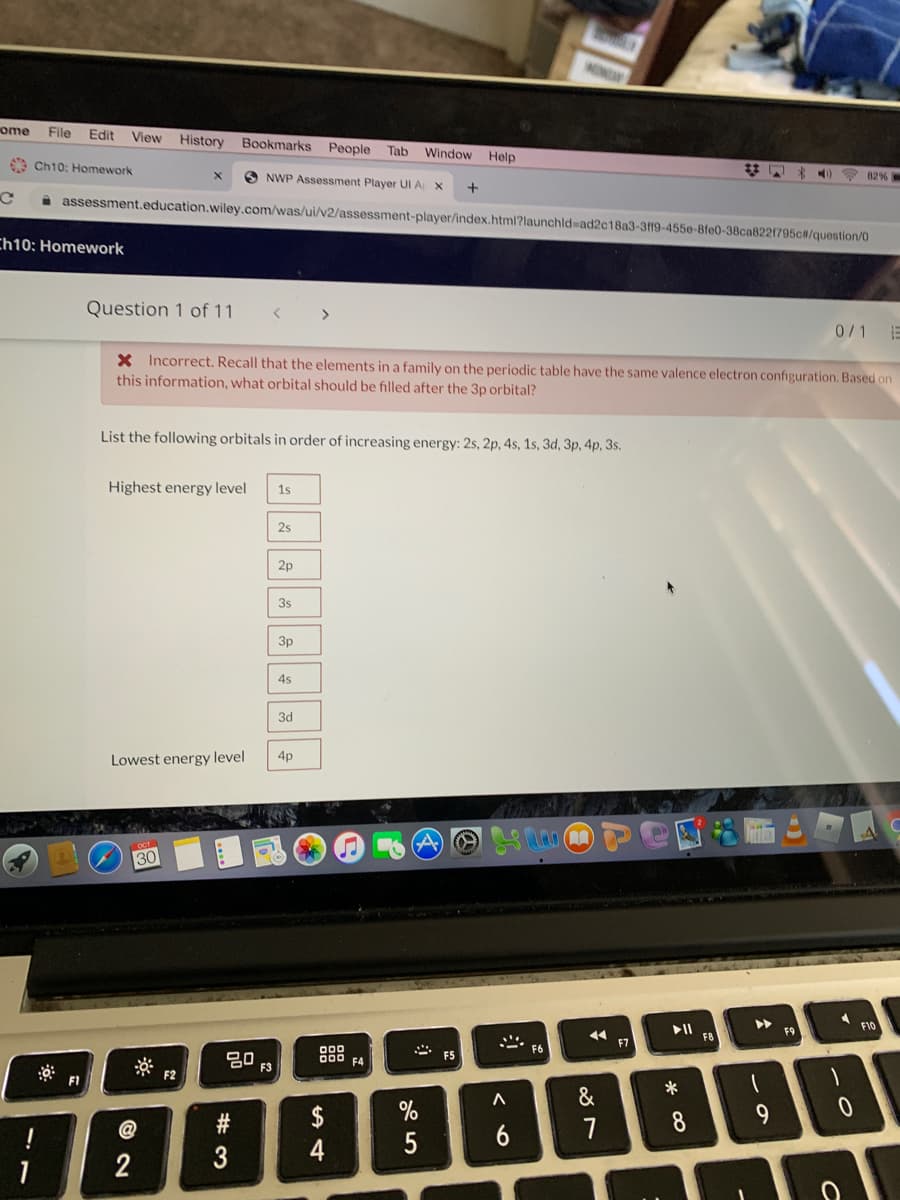
List the following orbitals in order of increasing energy: 2s, 2p, 4s, 1s, 3d, 3p, 4p, 3s. Highest energy level 1s 2s 3s 3p 4s 3d Lowest energy level
Q: Which of the following quantum number sets would represent an electron within an orbital in the 4d…
A: Principal quantum number (n)- Represents the shell of the electron. Possible Values of 1,2,3,......…
Q: How many of the following are accurate orbital diagrams? (A) 1 1s 2s 2p 11 1 1 (B) 1s 2s 2p (C) 1s…
A: We know, the general rules of electronic configuration are
Q: 回国 6.8.3 Which orbital diagram is correct for the ground-state S atom? a) 1L 1s 2s? 2p° 3s? 3p* b)…
A: The correct orbital diagram for the ground state of sulfur has to be given,
Q: How many p orbitals are in each energy level, except n=1? a. 1 b. 3 c. 5 d. 6
A:
Q: Q16. Which one of the following is an incorrect orbital notation? B. 2d C. 3s D. 2p A 4f
A: 2d orbital can’t exist in an atom. We can provide an explanation for it from its subsidiary quantum…
Q: n = 4, 1 = 2 %3D
A: Quantum Number: The set of number which gives every information related to the position, shape, and…
Q: 8. Which of the following is an incorrect designation for an atomic orbital? a) b) 3d с) 2р d) 3f 1s
A:
Q: Write the A) orbital diagram of the following. From the diagram, how many unpaired electrons are…
A: as question part A is already solved we are going for question part B in this part the given…
Q: 6.6.4 What is the total number of orbitals in the shell designatedby n = 3?a) 1 d) 6b) 2 e) 9c) 3
A: Orbitals in a shell are the location where electrons accommodate. Each orbital can have 2 electrons.…
Q: Describe the relative size and energy for each of the following orbitals 1. 2s vs 3s and 1s 2. 2p…
A: In order to compare the relative size and energy of the given orbitals, the use of quantum numbers;…
Q: Which of the following orbitals has the highest energy in a multielectron atom? The quantum numbers…
A: We know that, The highest energy of the orbital is for that orbital for which the sum of n and l is…
Q: Look at the following orbital diagrams. What principle/rule do they break? Explain. а. 1s 2s 2p b.…
A: According to the given diagram, which principle/rule break --
Q: 4. For the following sets of quantum numbers, what is the maximum number of electrons or orbitals…
A: Hello. Since you have posted multiple questions and not specified which question needs to be solved,…
Q: List the four quantum numbers associated with the last electron added in the following orbital…
A: n = 3 l = 1 ml = -1, 0, 1 ms = +1/2 and -1/2
Q: How many orbitals are associated with each of the following types? 1s 5f 4f
A: Orbitals is the 3D space around the nucleus where probability of finding an electron is maximum is…
Q: ml values
A:
Q: Which of the following orbital designations is (are} not possible?msp; a . 3 f c . 4 d b . 5 s…
A: Principal quantum number (n): It signifies about name, size, energy of the orbit to which electron…
Q: How many orbitals in an atom can have each of the follow-ing designations: (a) 5f; (b) 4p; (c) 5d;…
A: An atom is the basic and fundamental unit of matter. An atom consists of electron and the nucleus in…
Q: Write the A) orbital diagram of the following. From the diagram, how many unpaired electrons are…
A: Electronic Configuration : This gives us information about the shape of orbitals and energy of the…
Q: What are the possible orbitals for n = 4? s, p, d, f s s, p, d s, p
A: l <(=) n - 1 For n = 4 , l <(=) 3 Therefore, l = 0, 1, 2, 3
Q: Match the quantum numbers associated with each of the following orbitals: 4p Choose... 1s Choose...…
A:
Q: Which one of the following sets of quantum numbers represents an electron with the highest energy? A…
A: Given : Set of quantum number To find : set of quantum number which represents electron in higher…
Q: Which orbital in each of the following pairs is lower in energy in a many-electron atom: (a) 2s, 2p;…
A: The pair of electrons are given a question. We know that, The orbital having the lower sum of n and…
Q: Which is the correct orbital notation of a lithium atom in the ground state? 2p1 1. 1s1 2s2 182 2s2…
A:
Q: Which ground-state atom has an electron... Which ground-state atom has an electron configuration…
A: The electron configuration is given as : [Kr] 5s2 4d10 5p2
Q: Which combination of quantum numbers is possible for an atom with five orbitals in one subshell?…
A:
Q: Look at the following orbital diagram. What principle/rule does it break? 1s 2s 2p Choose all that…
A: From the given orbital diagram : Which principle/rule it breaks = ?
Q: What is the maximum number of electrons associated with any orbital? (a) 6…
A: We have one orbital in s, three in p, 5 orbitals in d and 7 orbitals in f- subshell. Orbitals is the…
Q: Which electron orbital diagram represents a violation of the Aufbau principle? 1s 2s 2p B 1s 2s 2p…
A: Given that : We have to determine which of the following electron orbital diagrams represents a…
Q: 5. Determine the maximum number of electrons that can be found in each of the following subshells:…
A: Maximum number of electrons in a subshell = 2(2l+1)For s subshell (l = 0)Maximum number of electrons…
Q: 4. Identify the maximum number of orbitals for the following: a. the 3rd energy level b. n35 c.…
A: An atom is the basic and fundamental unit of matter. An atom consists of electron and the nucleus in…
Q: 3. Which of the following relates to the phenomenon of electrons occupying orbitals at the same…
A: Aufbau principle states that in the ground state of atom , electrons are filled in order of…
Q: The orientation in space of an orbital is designated by which quantum number? Select one: а. 1 b. n…
A: Principal quantum number specifies size of the orbital, Azimuthal quantum number specifies shape of…
Q: What is the total number of orbitals in the Third energy level (n = 3.)? a) 4 c) 8 e) 16 b) 18 d) 9…
A: Option-d There are total 9 orbitals present in third energy level of an atom.
Q: In an atom of oxygen, there are two electrons in the 2s orbital. Where is the next electron located?…
A: Oxygen is the chemical element . It is member of the chalcogen group. It is highly reactive…
Q: Which one of the following orbital designations is INCORRECT? A. 5d В. 25 ©3f D. 7s Е. Зр (2)
A: To solve this question first of all see what are the rules that we need to solve this question.…
Q: Here are sketches of four electron orbitals: Are any of them s orbitals? List the s orbitals, if…
A:
Q: For a principal quantum number n, how many atomic orbitals are POSSIBLE? Select one: a. n b. n2…
A: If the principal quantum number is n, the number of atomic orbitals which are possible is n2. For…
Q: Label each of these orbital designations as “possible” (exists) or “impossible” (does not exist).…
A: a. n = principal quantum number (1,2,3....n) only positive b . l = orbital angular momentum…
Q: Q16. Which one of the following is an incorrect orbital notation? A. 4f B. 2d C. 5p D. 65
A:
Q: 4. Which of the following subshell has five degenerate orbitals? (a) 2s (b) 3d (c) 4f (d) 5p 5. What…
A: 4) since degenerate orbitals means that the energy of orbitals are same And since we know that d has…
Q: Which of the following orbital designations is incorrect? a) 1p b) 2d c) 3f d) all of the above
A:
Q: Here are sketches of four electron orbitals: 88 A Are any of them s orbitals? List the s orbitals,…
A: We have to categorise the orbitals diagram given in the question. There are 4 different type of…
Q: A certain shell is known to have a total of 4 orbitals. Which shell is it? Select one: O a. n 8 O,b.…
A: Number of orbitals in a shell = n2 Where, n = principal quantum number
Q: n = 3, 1 = 0
A: The quantum number helps in determining the property of an electron. There are four quantum numbers-…
Q: How many orbitals in an atom can have each of the following designations? (a) 7s (b) 5d (c) 6p…
A: The number of orbital in the given shell and subshell is determined by the quantum number. The four…
Q: According to the Aufbau principle, which orbital should be occupied first? A. 4s B. 3d C. 3p
A: Applying n+l rule to determination of energy of orbitals.
Q: quantum numbers and orbital notation. Explain why these are impossible. a. n=4, =2, m²-3, m,=-½ b.…
A: ->n,l,ml,ms are quantum number then l =0 ( s orbital) l = 1 ( p-orbital ) l = 2 ( d-orbital) l =…
Q: 1. State the maximum number of electrons that the following sublevels may contain. а. 7s b. 5p с. Зd…
A: Please note: As per our company guidelines we are supposed to answer only three parts of one…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- Must answer all questions eslse downvote A. MULTIPLE CHOICE. Choose the BEST answer. PLEASE HELP ME ANSWER EVERYTHING THANK YOU Q)What is used for heating small amounts of solids at a high temperature?a) Mortar and pestleb) Evaporating dishc) Crucible and coverd) Clay triangleQ)Which of the following is used in separation techniques?a-Rubber policemanb-Graduated cylinderc-Volumetric flaskd-Filter paperQ)Which of the following describes a centrifugate?a-Always clearb-Supernatant liquidc-Discarded via decantation onlyd-Solid particlesQThe inward force that pulls substances towards its center is called .a-Gravitational forceb-Centrifotal shiftc-Continental shiftd-Centrifugal forceQA "slippery floor" is considered a .a-hazardb-riskc-flash pointd-toxicantQ)Mrs. Lily Potter is 24 weeks pregnant. She was exposed to a chemical while making a potion. What is the type of the chemical she should be avoiding?a-neurotoxicantb-asphyxiantc-teratogend-carcinogenQ)Which of the following should be…In a river with a constant flow rate, the factory releases wastewater containing NaCl at a concentration of 200 mmol / L to the river at 20.0 L / s. The Na+ and Cl- concentrations at the downstream observation point were 1.00 mmol / L and 0.800 mmol / L, respectively. What is the river flow Q and Cl- concentration before the inflow of drainage? The river originally contains 0.500 mmol / L of Na+.A. B. and C. already solved (https://www.bartleby.com/questions-and-answers/chemistry-question/a89691d6-2162-4677-b0cc-dc84fcceda08). Please answer E, F and GCircle in pencil in the image on th eirght cide of the diagram is a "2"( Pb(NO3)2 ).
- Please answer fast it’s very important and urgent I say very urgent so please answer super super fast please For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.466A 120. MW coal plant wants to add a SOXemissions control system to reduce its emissions. The emission control system has a capitalcost of $815 per kW and reduces SOX emissions by 98% to 0.0918 kg/MWh. Assume thelifetime of the emission control system is 20 years and the power plant produces 650GWh/y. a)What is the annualized cost of this system in dollars per year if the discount rate (interestrate) is 7.0 percent/year? What if the discount rate is 4.0 percent/year? b)What is the cost per 1.0399 x 104 kWh of electricity usage (the average annual U.S.residential electricity consumption in 2017 according to the EIA) at each of the discountrates specified in part a? c)What is the cost per metric ton of SOx removed at each of the discount rates specified inpart a? In a metered-dose inhaler (MDI), such as those used for asthma medication, medicine isdelivered by a compressed-gas propellant. (The device is similar in concept to a can of spraypaint.) When the inhaler is activated, a fixed amount of the medicine suspended in thepropellant is expelled from the mouthpiece and inhaled. In the past, chlorofluorocarbons(CFCs) were used as propellants; however, because of their reactivity with the Earth's ozonelayer, they have been replaced by hydrofluorocarbons (HFCs), which do not react withozone. Now HFC use is also being reduced due to their high global warming potential. In one brand of inhalers, the original CFC propellant was replaced by HFC 227ea (C3HF7,heptafluoropropane). The volume of the inhaler propellant reservoir is 1.00×102 mL, and thepropellant is charged into the reservoir to a gauge pressure of 4.443 atm at 23°C. An onlinesearch for properties of HFC 227ea yields the information that the critical temperature andpressure of the substance…
- A clinic had 25 patients on Friday morning. If 21 patientswere given flu shots, what percentage of the patientsreceived flu shots? Express your answer to the ones place.Sugar Cane Solution Evaporation. Evaporator is used to thicken sugar cane solution. The feed rate is 10,000 kg / day, wherein the solution contains 25% (w / w) solid sugar. The result of thickening is that of a 65% (w / w) solution of solid sugar. Calculate the weight of the solution produced and the amount of air removed. (a) The weight of the solution produced = Answerkg (b) The amount of air removed = AnswerkgChemistry (i) For T = 298K ΔG° = -8.314 x 298 x ln (2.83x10-3) = 14.524 KJ/mol (ii) For T = 308.15K ΔG° = -8.314 x 308.15 x ln (9.619x10-3) = 11.891 KJ/mol (iii) For T = 318.15K ΔG° = -8.314 x 318.15 x ln (3.14x10-2) = 9.147 KJ/mol (iv) For T = 328.15K ΔG° = -8.314 x 328.15 x ln (8.82x10-2) = 6.621 KJ/mol (v) For T = 338.15K ΔG° = -8.314 x 338.15 x ln (2.42x10-1) = 3.990 KJ/mol Based on the measured ΔG° values, is this equilibrium spontaneous at room temperature? Which factor, entropy, or enthalpy, has the greater impact on spontaneity in this case? Explain your answers.

