Q: What are the advantages of evaporative-light scattering detector when used with high-precision…
A: ELSDs are aerosol-based HPLC detectors which work by converting the eluents of target components…
Q: 4000 100 IR Spectrum (quid film) 88888 80 Få 60 40% 8 3000 CO 80 40 13C NMR Spectrum (50.0 MHz.…
A: 1H-NMR spectrum gives complete information about structure of the molecule. Form the position of the…
Q: What is the melting point of the substance modeled by this heating curve? F. 150 D E 135 120 105 90…
A:
Q: Based on the molecular structure of thymol, which region is considered to be electrophilic? HO The…
A: The answer is as follows:
Q: Br2, H₂O major product
A: Identify the final products of the given reactions.
Q: Chemistry below. Which Consider the two compounds would you expect to be more persistent in the…
A: Because of presence of phosphate group in second compound. It is more persist in the environment. If…
Q: Harriet Rowki prepared 100.0 mL of 2.50 M HBO3-NaH2BO3 buffer solution (pH = 8.50). The pKa value of…
A:
Q: The "blood alcohol concentration" (BAC) is given in grams of alcohol (ethanol) per 100 ml of blood.…
A: Given, BAC of blood = 0.10 We have to find the Molarity of ethanol in blood=?
Q: If a voltaic cell is made from [Cr₂O72/Cr+³] and [Cu+²/Cu] half-cells. What will be the total moles…
A: If a voltaic cell is made up of [Cr2O72-/Cr3+] and [Cu2+/Cu] half cells. Total moles electrons…
Q: Which of the following can be used to couple with Ni+2/Ni to produce a Ni-Metal based battery with…
A: K
Q: From the following list, select all salts that would form an acidic aqueous solution. LiClO2…
A: For these salts , we would write their dissolution in water and check the strength of acid and base…
Q: What is the mass used in each reactant to create stock solution Reactant #1 Potassium carbonate -…
A:
Q: 0=$=0 ÓH Ko
A: The given problems are based o basics in organic chemistry.
Q: Aluminum hydroxide is one component of antacids for hyperacidity. If one tablet contains 178 mg,…
A: Given : Ksp = 1.9 x 10-33 Volume = 1 litre Mass of aluminium hydroxide in one tablet = 178 mg
Q: CH3 AlCl3 + CH3-C-Cl CH3 Draw the molecule by placing atoms on the grid and connecting them with…
A: Here we are required to predict the product of the reaction
Q: At what position, and on what ring, would you expect the following substances to undergo…
A: ✓Given reaction is an example of electrophilic substitution reaction.An electrophilic substitution…
Q: H₂C CH₂ Show how the compound can be synthesized from benzene and alkyl chlorides.
A: Here we have to synthesize the following given compound from the starting material benzene and alkyl…
Q: What do we call a distinct substance that contains two or more elements chemically combined in…
A: 1.Element are the simplest form of matter Example ; lithium , barilium
Q: ow might you use a Grignard reaction to prepare the following alcohols: ) ОН (b) ОН (c) ОН -CH3…
A:
Q: Which reactant is the reducing agent? * This item is only equivalent to one point. KI О HCI O…
A:
Q: 2089 IR Spectrum (liquid film) LL 4000 100 80 of base peak 3000 63 60 405 20 40 13C NMR Spectrum…
A:
Q: Give the a) of the name Compounds given below 애 LOH
A: The key functionality present in the given compounds are alcohol and aldehyde respectively.
Q: QUESTION 14 Consider the reaction scheme below. Compound E PBr3 KMnO4/ H* + HCN Compound A Compound…
A:
Q: 1. Calculate the observed rotation, a one would measure for a 20.0% (m/v) D-glucose solution if a 2…
A: We have find out the observed rotation of D- glucose and L- glucose solution. Note -As per our…
Q: 4. The americium isotope Am is unstable and emits a 5.538 MeV alpha particle. The atomic mass of Am…
A: #4: An alpha particle, He24 has 2 protons (Z = 2) and 4 unit mass. Hence when an alpha particle,…
Q: From this chromatogram, which of the following compounds is the MOST polar?
A: Both C18 and C8 columns are used in reverse phase chromatography.
Q: At what position, and on what ring, would you expect the following substance to undergo…
A: In this question, we will see that which ring and what position will give expected electrophilic…
Q: What will be the minimum voltage required for the electrolysis of SnCl₂(aq) to just begin to…
A:
Q: What is true when acids and bases neutralize each other? O The amount of H* and OH is the same O The…
A: When acid is react with base it will form salt and water called neutralization
Q: Gas particles have random _____. speed speed and direction mass temperature direction
A: We can answer the above question by using Kinetic theory of gases.
Q: H₂C CH3 Show how the compound can be synthesized from benzene and alkyl chlorides.
A: In this question, we will synthesized the given carbonyl compound from the starting materials…
Q: A student finds the average Keq to be 370. (a) Calculate the approximate [FeSCN²+]/[SCN¯] in Part 3…
A:
Q: Assuming the same concentration of solution and all solutions at the same temperature, when…
A: The pH of a solution is the negative logarithm of hydrogen ion concentration. The pKb is the…
Q: Which among the following is a compound? salt crystals O charcoal O sulfur powder O iron filings
A:
Q: The following are intermediate products in the stepwise synthesis of compound 1 from benzene. Give…
A:
Q: C. Using the generic Michaelis-Menten plot below, describe the reaction's behavior in the following…
A: We know the michaelis menten equation r = (k2[E][S])/([S]+Km) Where r = rate of reaction [E]= intial…
Q: values : Experimental values of IR Peaks compared to literature values for Benzoic Acid (ATR…
A: A question based on IR spectroscopy that is to be accomplished.
Q: 3.5 moles of methane reacts with 12 moles of oxygen gas in a combustion reaction. How many moles of…
A: In a balanced Chemical reaction, Limiting reagent is the one which has less value of:-…
Q: What is the pH of the solution when 100 ml 0.02 M HCI is added to 100 ml 0.04 M sodium hydrogen…
A:
Q: CH₂Cl www
A: In this question, we will see the reaction mechanism for this transportation. You can see the…
Q: 5. Glutaraldehyde is a germicidal agent that is sometimes used to sterilize medical equipment. In…
A:
Q: What is the principle behind reverse-phase ion-pairing chromatography?
A: Reverse Phase ion chromatography use the principle of Solute ions instead of molecules .
Q: At what position, and on what ring, would you expect the following substance to undergo…
A: Aromatic electrophilic substitution reaction: Aromatic electrophilic substitution reactions are the…
Q: A solution of 0.25 M potassium hydroxide, KOH is added from a burette to a flask containing 25.0 mL…
A: For given pH curve , incorrect statement is to be determined,
Q: A piece of metal thought to be either chromium , nickel or lead was weighed and found to have a mass…
A:
Q: Determine the concentration of sodium hydroxide if 21.39ml of sodium hydroxide is required to…
A: In this question, we will determine the concentration of NaOH solution during the titration with…
Q: CH, — CH, CH,— CH,—NH, Your answer CH₂-CH₂-CH₂-O-C-CH₂-CH₂ 0 Your answer CH₂-CH₂-CH-CH₂-CH-CH₂ I OH…
A:
Q: Select the false statement for an organic reaction. In an electrophilic addition reaction, the O…
A: In elimination reaction, Bita hydride elimination occur not the base proton abstract at that carbon…
Q: She fills a reaction vessel with H3PO4 and measures its concentration as the reaction proceeds: time…
A:
Q: Determine the gravimetric factor of CrO4 in K₂Cr2O7. The atomic weight of chromium is 52g/mol 0.7891…
A:
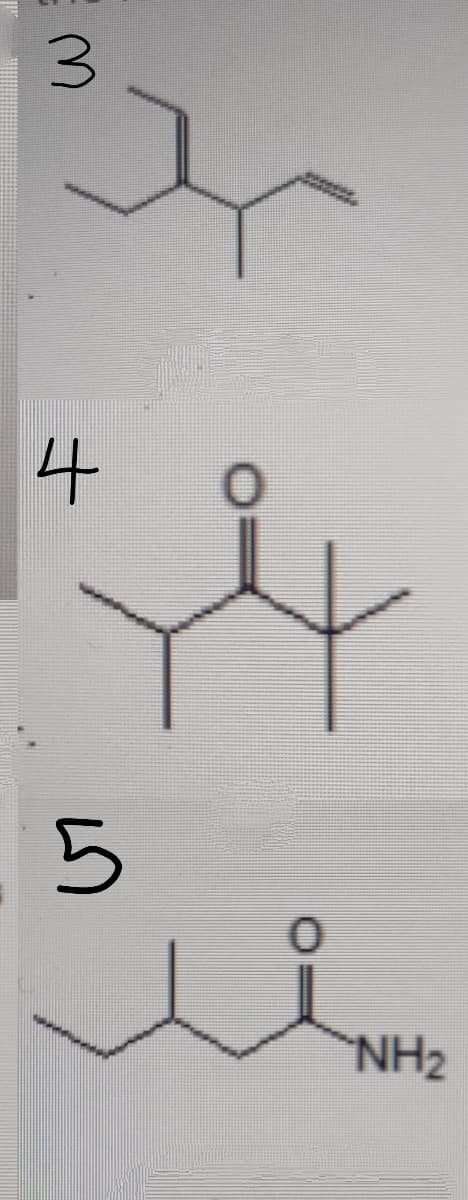
Step by step
Solved in 2 steps with 1 images


