Q: 8. Au adopts a fcc crystal structure with lattice constant a=4.08Å and AHsub=324.4 kJ/mol. Calculate…
A: The objective of the question is to calculate the value of surface energy, for the given surfaces…
Q: Give an IUPAC name for the compound below. Accepted names for branched alkyl groups are isopropyl,…
A: In IUPAC nomenclature, we first find the parent name or the longest continuous chain.In #1, we find…
Q: In the following solubility curve, the substance with the highest solubility in 100 g of water at 20…
A: Step 1:In the solubility curve the point where a straight line drawn from the temperature axis meets…
Q: HOW MUCH 1 M NAOH SOLUTION SHOULD BE ADDED TO 100 ML OF A 0.1 M H3PO4 AQUEOUS SOLUTION FOR IT TO…
A: Please see the attached image for the solution. If there are queries, please do not hesitate to ask.…
Q: Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer…
A:
Q: CONCEPT QUESTION 1. Retrosynthesis reaction. For the product given below, show the retrosynthesis…
A: Step 1:Step 2:The retrosynthesis and the forward synthesis of the target molecule are given. Step 3:…
Q: A balance has a sensitivty requirement of 4.0 mg. Explain how you would weigh 15 mg of a substance…
A: Step 1: Step 2: Step 3: Step 4:
Q: Table NU.1 lists some sources of household background radiation, all of which emit gamma radiation…
A: The objective of the question is to write nuclear equations for the decay of various radioactive…
Q: H3Cm Br cyclohexane I CH3
A: The objective of the question is to find out the both conformer of the given cyclohexane molecule…
Q: None
A: Reaction: Answer:
Q: None
A: Please see the attached image for the solution.If there are queries please do not hesitate to ask.…
Q: Please correct answer and don't use hend raiting
A: The two structures are the cyclic forms of sugars in their pyranose forms. The sugar at the left is…
Q: Please help with Organic Chemistry problem. What is the product of reaction shown? (what should be…
A: Step 1: The lone pairs on nitrogen of hydroxylamine attack the acyl chlorides. Step 2: The attack…
Q: A solution is prepared at 25 °C that is initially 0.41 M in chloroacetic acid (HCH2CICO2), a weak…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: a) This molecule has two functional groups, the C=O i.e. ketone and the methyl CH3 group.b) This…
Q: 0
A: The objective of the question is to find out the synthetic procedure of the given molecule from the…
Q: The correct lewis structure fir carbonate acid H2CO3
A: Thank you.
Q: None
A: a. Alanine and Leucine The interaction between alanine and leucine side chains involves a…
Q: ->> CaCl2(aq) + H2O(1) но(1) Predict the reactants of this chemical reaction. That is, fill in the…
A: The reactants are: HCl and Ca(OH)₂ The products are: CaCl₂ and water (H₂O) The balanced equation is:…
Q: 7. What is the equilibrium constant, K, at 25°C for this redox reaction under standard conditions?…
A: Step 1:Relation between standard cell potential and equilibrium constant…
Q: None
A: Avogadro's Law describes how gases behave at Standard Temperature and Pressure (STP), which is…
Q: Sm. Don't provide handwriting solution
A: Step 1:1) what is the pH ?It is negative log of hydronium ion concentration. 1) reaction as…
Q: Cp = 2.07+ T-673 1480 kJ kgK T-673 kJ Cy=1.6085+ 1480 kgK The specific heat of superheated water…
A: Given Data:The specific heat of water vapor at constant pressure is, .The specific heat of water at…
Q: pH i got from previous question which was calculate the pH of a 1.00L buffer solution that is 0.100M…
A: To calculate the pH of the buffer solution after the addition of NaOH, we need to consider the…
Q: A chemist titrates 240.0 mL of a 0.7078M acetic acid (HCH3CO2) solution with 0.3641 M KOH solution…
A: Given: VHCH3CO2=240.0mL;[HCH3CO2]=0.7078M;[KOH]=0.3641M;pKa=4.76Step 1: Solve for pKb and Kb.…
Q: Calculate the ionic strength of a solution containing 0.2 M NaCl in a 0.2 M Na2CO3. Consider all the…
A: The ionic strength (I) of a solution is calculated using the formula: I = 21∑Mi zi2whereMi is the…
Q: Give me the calculating steps thank you! here is the answer 1. a) 5.24 a b) 2 a
A:
Q: Please correct answer and don't use hend raiting
A:
Q: i.) Ester hydrolysis can occur in acidic or basic conditions. Predict the product AND show the…
A: c)This reaction proceeds without the need for an external acid or base catalyst because water itself…
Q: Give the major organic product of the following reaction. NaOCH NaOH 0 ? CH₂OH H₂O heat கு OMe OH…
A: Step 1 : In presence of base (NaOH in CH3OH) esters do self condensation reaction by forming a…
Q: 15. Which of the following is the strongest acid? H OH OH О 16. Which of the following is the…
A: Step 1:Step 2: Step 3: Step 4:
Q: Show the steps necessary to transform the acid on the left into the compound on the right. Be sure…
A: Answer:
Q: Using the aromacy rules determine if the following compounds A ,B are aromatic and explain why
A: Step 1The 1st one is antiaromatic because it has got 4 pie electron and due to huckel rule ie 4n+2…
Q: You want to determine Fe in a solution via manganometric titration. To determine the exact…
A: Relevant Reaction Equations:Standardization Reaction:Balanced equation: Na2C2O4 (aq) + 2KMnO4 (aq) +…
Q: Answer step 1 step 2 step 3
A: Step 1:Calculate the total cost associated with purchasing the land: Cost of land = Purchase price…
Q: (3 points) Using the dissociation constant(ka), calculate the equilibrium concentrations of Cd2+ and…
A: The complex ion Cd(CN)42- dissociates into its component ions when dissolved in aqueous solution:…
Q: :):)):;););)/(/);$:$
A:
Q: X INCORRECT; see section 18.1 A piece of a wooden tool found at a Northwest Indian fishing village…
A:
Q: Ascorbic acid, H₂C6H6O6, is a diprotic acid usually known as vitamin C. For this acid, pk1 is 4.10…
A:
Q: None
A: To solve this problem, we need to use the Arrhenius equation, which relates the rate constant (k) of…
Q: None
A: Option A is incorrect because NH3 is a weak base, and thus the titration will not result in a pH of…
Q: Draw the mechanism arrows for the reaction between an acid chloride and an alcohol.
A:
Q: 3) Venlafaxine is a Wyeth antidepressant that is synthesized on a commercial scale from the starting…
A: Step 1:
Q: dont provide handwritinng solution...
A: Approach to Solving the Question:1. Understand the Problem: Begin by carefully reading and…
Q: How many sites of diversity are there in this combinatorial library? NH 2 6 4 3 HN NH2
A: Step 1To determine number site of diversity , we would check the positions on which different atoms…
Q: Draw the major organic product of the reaction shown below. OH + HBr You do not have to consider…
A: 1) In the presence of strong acids, alcohols can be converted into their corresponding alkyl…
Q: Draw the Major Organic product of the following reaction. Do NOT use abbreviations such as Ph. Do…
A: Step 1:The Nucleophile attack to the electrophilic carbon centre. Step 2: Since C=O has high bond…
Q: draw an 8 membered ring that would be considered anti-aromatic. include the molecular orbital…
A: Step 1:Frost's CircleDraw a circle of radical 2 beta and centered as alphaInside the circle,…
Q: Consider the reaction: 2SO2(g) + O2(g) ⇌ 2SO3(g) K = 1.8 x 102The reaction chamber was initially…
A: The objective of the question is to calculate the partial pressure of O2(g) at equilibrium given the…
Q: What is shown below is an interesting way of making lactone B from lactone A. The reaction takes…
A: Step 1: Step 2: Step 3: Step 4:
Give the organic reaction product(s) for the following
reactions:m
I need answer expert solution
Need answer step by step
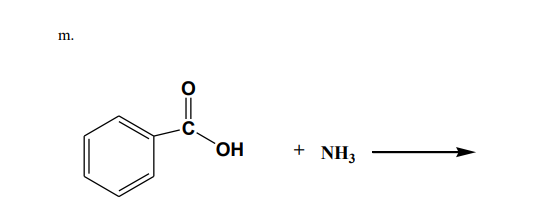
Step by step
Solved in 2 steps with 1 images

- Can you help me to explain step by step with number 2? Can you explain it to me?Hi! I'd just like to confirm if I understood it correctly. :) K = [CS2] [H2]4 / [CH4] [H2S]2the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (e) and (f).

