Macmillan Learning Organic Chemistry Maxwell presented by Macmillan Learning Select the correct IUPAC name for the following organic substrate, including the R or S designation where appropriate, and draw the major organic product(s) for the SN 1 reaction. Include wedge-and-dash bonds and draw hydrogen on a stereocenter. ……...H H₂O Select G 5 Draw Rings More Erase Q2Q
Macmillan Learning Organic Chemistry Maxwell presented by Macmillan Learning Select the correct IUPAC name for the following organic substrate, including the R or S designation where appropriate, and draw the major organic product(s) for the SN 1 reaction. Include wedge-and-dash bonds and draw hydrogen on a stereocenter. ……...H H₂O Select G 5 Draw Rings More Erase Q2Q
Chapter9: Alkynes: An Introduction To Organic Synthesis
Section9.SE: Something Extra
Problem 46AP: A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the...
Related questions
Question
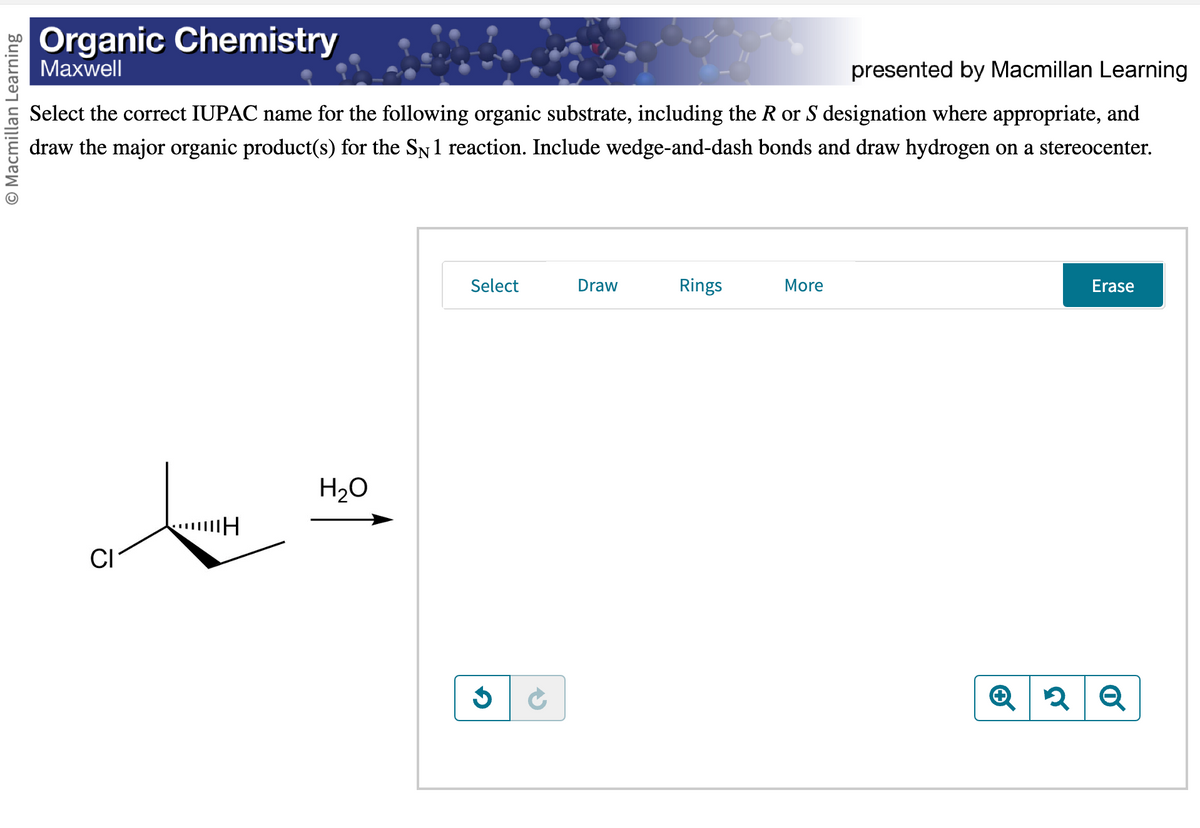
Transcribed Image Text:O Macmillan Learning
Organic Chemistry
Maxwell
presented by Macmillan Learning
Select the correct IUPAC name for the following organic substrate, including the R or S designation where appropriate, and
draw the major organic product(s) for the SN 1 reaction. Include wedge-and-dash bonds and draw hydrogen on a stereocenter.
t
H
CI
H₂O
Select
G
Draw
Rings
More
Erase
Q2 Q
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

