Q: How many mL of 0.400 M KCl would be required to make a 0.0795 M solution of KCl when diluted to 200....
A: M1V1 = M2V2 Where, M1 = initial molarity, V1 = initial volume, M2 = final molarity, V2 = final volu...
Q: Consider the following reaction: COCI,(g) CO(g) + Cl,(g) If 1.79×10 moles of COCI,(g), 0.470 moles o...
A: Equilibrium constant of any reaction can be obtained by dividing the multiplication of concentratio...
Q: 22,23,24,25
A: Use formula Mole= mass÷molar mass Then use Mole-mole realtionship
Q: Nitrate ion (NO31-) h) determine the formal charge of all the atoms in the nitrate ion i) is the st...
A: h) calculation of formal charges in Nitrate ion formal charge on nitrogen formal charge =V.E - N.E -...
Q: Consider the following equation in the gas phase: Carbon Monoxide + Hydrogen d...
A: The initial concentration of carbon monoxide is 0.500 atm and of hydrogen is 1.00 atm. The equilibr...
Q: Coal can be converted to methane, a chief component of natural gas, according to the following react...
A: Use relation ∆G= ∆H-T∆S And ∆G= -2.303RTlogK ∆G=23943.95kj/mol Kc= 6.3×10-5
Q: Calculate the percent yield of alum if a student starts with 1.61 g of Al and produces 18.9 g of alu...
A: The molecular formula of alum states that one molecule of alum contains one atom of aluminum. The m...
Q: http://chemcollective.org/activities/vlab/3 Virtual lab
A: Given that: Volume of the Solution to be made = V1= 250.0ml Molarity of the solution to be made = M1...
Q: Calculate the pH of a 0.025 M solution of propanoic acid (K, = 1.3 x 10-5). 7.48 0.025 O 5.6 x 104 1...
A:
Q: The amount of energy required to raise the temperature of a substance by 1 K (1 °C) is its heat capa...
A: We have to conclude what is the term called in chemical thermodynamics when 1g of substance is heate...
Q: For the reaction: NO (g) + O3 (g) ---> NO2 (g) + O2 (g) the value of A= 8.7 x 1012 s-1 and Ea = ...
A: Use K= Ae-E/RT K=11.74×10-83S-1
Q: Chemistry Question
A: Limiting reactant: The reactant species which is totally consumed is the reaction is allowed for com...
Q: When 0.625 grams of ethanol(C2H5OH) undergoes combustion in a bomb container calorimeter attaining a...
A: We are given 0.625 g of ethanol which undergoes combustion to yield carbon dioxide and water and the...
Q: How many moles of NaOH need to be added to 200.0 mL of a 0.200 M solution of HF to make a buffer wit...
A: Given NaOH added to 200.0 mL of a 0.200 M solution of HF to make a buffer with a pH of 4.20 Ka for...
Q: How many milliliters of 0.200 M FeBr3 are needed to react with an excess of Na2S to produce 0.172 g ...
A: Molar mass of Fe2S3is 207.9 g/mol Mass of Fe2S3 is 0.172 g.
Q: calculate the mass of primary standard sodium chloride (MM=58.44) necessary to react with 25 ml of 0...
A: Molarity is a term used for concentration which shows the moles of solute dissolved per liters of so...
Q: An acetylation mixture containing 3.5 g acetic anhydride is reacted with 4.5 g salicylic acid. The e...
A: Given,Mass of acetic anhydride=3.5gMass of salicylic acid=4.5gExperimental yield=5.1gMolar mass of a...
Q: alculate Keq for the reaction A (aq) + B (aq) = 2C (aq) if at equilibrium [A] = 0.26 M, [B] = 35 M, ...
A: Equilibrium constant is defined as ratio of concentration of to the concentration of reactant 3)Kc=0...
Q: . What is the molarity concentration of CuCl2 in a solution prepared by dissolving 33.6g of CuCl2 in...
A: Molarity of a solution can be determined by dividing the number of moles of solute by volume of the ...
Q: PROBLEM-SOLVING PRACTICE 3-6 What is the mass percent of each element in hydrated nickel(II) chlorid...
A:
Q: Which of the following are strong acids? Select all correct answers. H2SO4 OHCI HBr OHI OHC2H3O2
A: There are different concept which tells about the nature of acids. According to Arrenhius concept, a...
Q: 2. Potassium fluoride has a solubility of 92 g of KF in 100 g of H2O at 18 °C. Determine if each of ...
A: Record the given data, Solubility of potassium fluoride (KF)=92 g Mass of water =100 g Temperature= ...
Q: 4. Identify X in the reactions below a) 23°Pu + 15n → 254FM + X X= b) 23®U (Y, 7e) 253ES Y =
A: Balancing of a nuclear reaction: In a nuclear reaction, the total mass and charge of the reactants a...
Q: Selenic acid, H2SeO4, is produced from elemental selenium in a two-stage reaction. First the seleniu...
A:
Q: Please give full information
A: Constitutional isomers: Compounds with same chemical formula but different structures are called con...
Q: Which of the following statement best describe the potassium permanganate test on hydrocarbons? Pota...
A: Potassium permanganet acts as the oxidizing agent in the acidic medium. Hence we have to find which ...
Q: To what volume (in mL) would you need to dilute 25.0 mL of a 1.45 M solution of KCl to make a 0.0405...
A: Given, 25.0 mL of a 1.45 M solution of KCl and the solution diluted to make a 0.0405 M solution of K...
Q: A scuba diver ascends too quickly and develops the bends. A nitrogen bubble has formed in the patien...
A: Interpretation - To determine the volume of bubble at the surface at constant temperature and mole...
Q: 2 when 3,269g of Zn is heated in puve Onggen the Sample gal Oareoy of oxide product Cale the fmpinca...
A: Empirical formula is defined as the simplest whole number ratio between the constituting atoms of a ...
Q: „(1) + 15 0,(g) → 12 CO,(g) + 6 H,O(1) + 6542 kJ H, is burned and the heat produced from the burning...
A: First calculate heat release in 1 mole of benzene then use mole mole relationship After this calcula...
Q: What atom has a ground-state electron configuration [Kr]4d105s25p1?
A:
Q: Identify the spectator ions. Sr(OH)2+K2S=2KOH+SrS
A: The reaction given is, => Sr(OH)2 + K2S --------> 2 KOH + SrS
Q: Many important metals occur as sulfide, arsenide, and antimonide minerals, especially in the Sudbury...
A: The balanced reaction taking place is given as, => 2 Ni11As8 + 31 O2 → 22 NiO + 8 As2O5 Given : ...
Q: How many grams of carbon dioxide, CO2 are released from a 6.492 g sample that has 38% magnesium carb...
A:
Q: At 25C and pH 7, a solution containing compound A and its reduced form AH2 has standard electrode ...
A: (i) The standard potentials are given for A and B which can be represented as; A+2H++2e-→AH2 Eoc...
Q: What is the molecular formula of a compound with the empirical formula CH2CH2 and a formula mass of ...
A: Empirical formula of compound=CH2 Mass of empirical formula=1 x atomic mass of C + 2 x atomic mass o...
Q: Molar Conc, Ionic Strength, and Activity...
A: The ratio of moles of solute in per liter of solution is known as molar concentration.
Q: Calculate the mass in grams of 1.39x10^24 molecules of methanol. The chemical formula for methanol i...
A: We are given 1.39×1024 molecules of ethanol are present and we have to calculate the mass in grams p...
Q: When a 3.26g of Zinc is heated in pure oxygen, the sample gains 0.800g oxygen in forming an oxide. C...
A: Mass of Zinc (Zn) in the compound = 3.26 g Mass of oxygen (O) in the compound = 0.800 g. Hence, the...
Q: An unknown substance has a mass of 56.8 g and a volume of 23.4ml. What is the density of this unknow...
A: Mass of the unknown substance is 56.8 g. Volume of the unknown substance is 23.4 mL.
Q: Please question A&B
A: Since your question has multiple parts, we will answer the first question for you. If you want the r...
Q: A student carried out the reaction shown by reacting 10.0g P4 with excess Cl2 and obtained 35.0...
A: Given: Mass of P4 = 10.0 g Mass of PCl3 (actual yield) = 35.0 g Molar mass of P4 = 123.89 g/mol Mola...
Q: An alloy is a solid solution between two metals. A 15.0 grams nugget of an alloy made from gold and ...
A: Assuming mass of gold i.e Au in alloy = y. Given : Mass of alloy = 15.0 g Initial temperature of all...
Q: Use the conversions : 4.184Joules = 1calorie, 1000cals=1kcal, 1000joules = 1kJ 18 kcalories...
A: Unit conversion is one of the basic tool for the measurement. Unit conversion can be used to convert...
Q: Calculate the pH of a 0.045M sulfurous acid solution. H,SO, → H + HSO, HSO3-→ H+ + SO,? Ка, = 1.7 x ...
A: The equilibrium dissociation reactions given are, Given : Concentration of H2SO3 = 0.045 M Since th...
what is the name of the major product
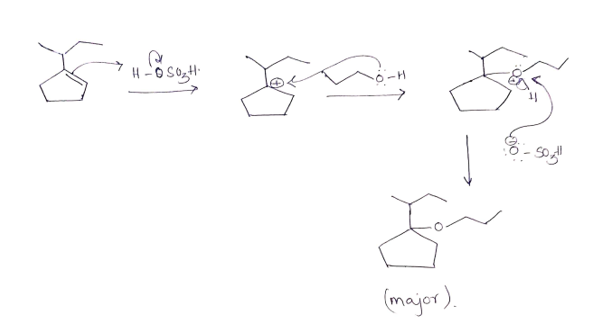
Step by step
Solved in 2 steps with 1 images


