Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter4: Introduction To Gases
Section: Chapter Questions
Problem 4.2TC
Related questions
Question
Transcribed Image Text:Knowledge Check
Question 8
BryAnna
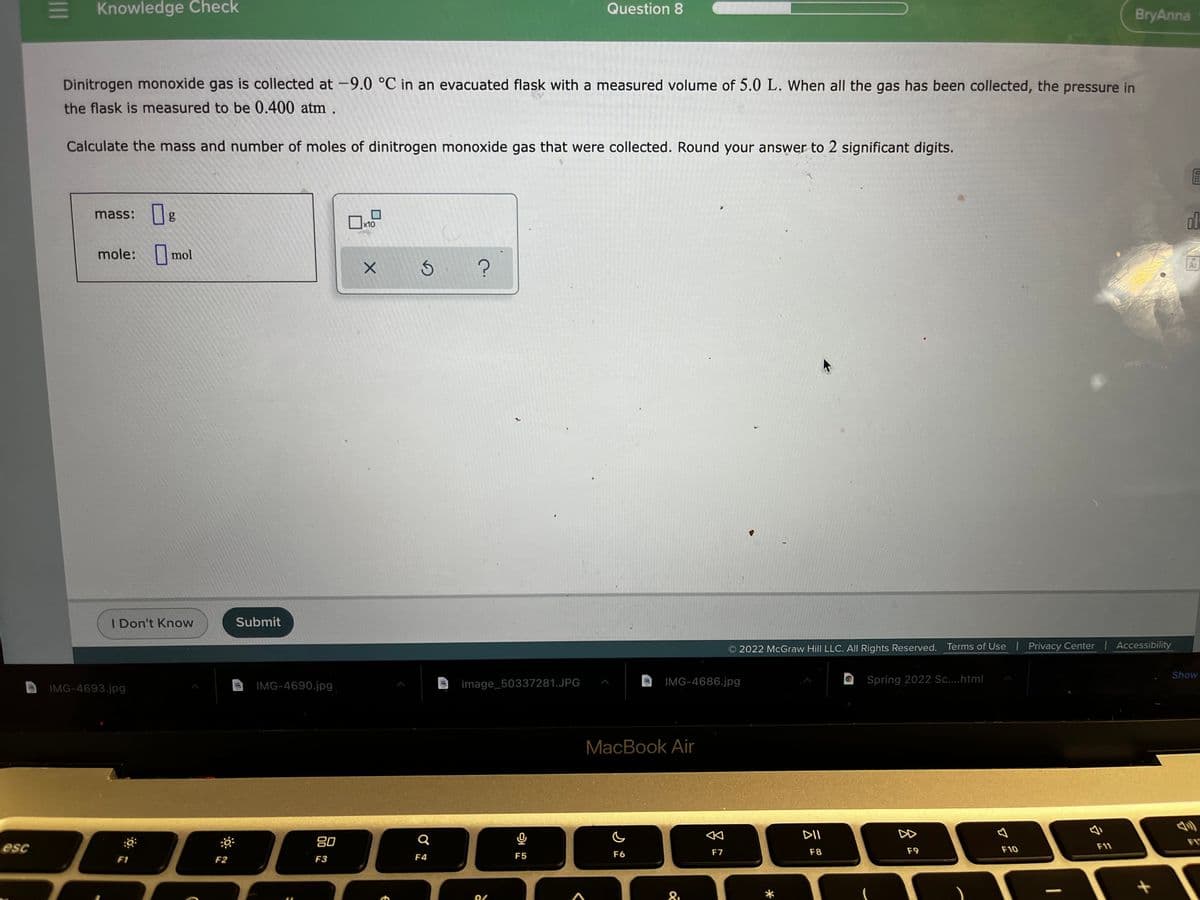
Dinitrogen monoxide gas is collected at -9.0 °C in an evacuated flask with a measured volume of 5.0 L. When all the gas has been collected, the pressure in
the flask is measured to be 0.400 atm .
Calculate the mass and number of moles of dinitrogen monoxide gas that were collected. Round your answer to 2 significant digits.
mass: g
x10
mole: mol
I Don't Know
Submit
2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center | Accessibility
Show
IMG-4690.jpg
image_50337281.JPG
IMG-4686.jpg
Spring 2022 Sc....html
IMG-4693.jpg
MacBook Air
DII
DD
80
F1
esc
F10
F11
F6
F7
F8
F9
F1
F2
F3
F4
F5
8.
了
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning