Mass of NaOH = SC Submit Part E Ascorbic acid, or vitamin C (C6H₂O6, molar mass= 176 g/mol), is a naturally occurring organic compound with antioxidant properties. A healthy adult's daily requirement of vitamin C is 70-90 mg. A sweet lime contains 2.81x10-4 mol of ascorbic acid. Mass of ascorbic acid= Submit Provide Feedback To determine whether the ascorbic acid in a sweet lime meets the daily requirement, calculate the mass of ascorbic acid in 2.81x10 mol of ascorbic acid. Express the mass in grams to three significant figures. View Available Hint(s) alt 7 WwD option VG ΑΣΦ Q A N IVE ΑΣΦ 3 → P @ 2 W F2 S 27 #3 X command 20 F3 E D 54 ? $ C B F4 70 ? F 8 % 5 S V F5 T G MacBook Air A 6 B Y & 7 #tv H U N * 00 8 J AO 1 M ( 9 ➤➤ K O < 1 0 L F10 P command A 4) : ¡ FIT { + 11 [ alt = ? option 4) FIZ 1 Next > 1 (
Mass of NaOH = SC Submit Part E Ascorbic acid, or vitamin C (C6H₂O6, molar mass= 176 g/mol), is a naturally occurring organic compound with antioxidant properties. A healthy adult's daily requirement of vitamin C is 70-90 mg. A sweet lime contains 2.81x10-4 mol of ascorbic acid. Mass of ascorbic acid= Submit Provide Feedback To determine whether the ascorbic acid in a sweet lime meets the daily requirement, calculate the mass of ascorbic acid in 2.81x10 mol of ascorbic acid. Express the mass in grams to three significant figures. View Available Hint(s) alt 7 WwD option VG ΑΣΦ Q A N IVE ΑΣΦ 3 → P @ 2 W F2 S 27 #3 X command 20 F3 E D 54 ? $ C B F4 70 ? F 8 % 5 S V F5 T G MacBook Air A 6 B Y & 7 #tv H U N * 00 8 J AO 1 M ( 9 ➤➤ K O < 1 0 L F10 P command A 4) : ¡ FIT { + 11 [ alt = ? option 4) FIZ 1 Next > 1 (
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter8: Chemical Composition
Section: Chapter Questions
Problem 128CP: itamin B12 , cyancobalamin, is essential for human nutrition. Its molecular formula is...
Related questions
Question
![Mass of NaOH =
SC
Submit
Part E
Ascorbic acid, or vitamin C (C6H8O6, molar mass = 176 g/mol), is a naturally occurring organic compound with antioxidant properties. A healthy adult's daily requirement of vitamin C is 70-90 mg. A sweet lime contains 2.81x10-4
mol of ascorbic acid.
Mass of ascorbic acid U
Submit
Provide Feedback
To determine whether the ascorbic acid in a sweet lime meets the daily requirement, calculate the mass of ascorbic acid in 2.81x104 mol of ascorbic acid.
Express the mass in grams to three significant figures.
►View Available Hint(s)
7
alt
[VD] ΑΣΦ
option
Q
A
N
VG ΑΣΦΑΦ
16,388
2
-
F2
W
S
27
X
H
command
ㅁㅁ
#3
F3
E
D
$
?
54
C
www
B
DOD
F4
R
?
F
19
g
%
5
0
V
F5
T
G
MacBook Air
6
B
F6
Y
&
7
#tv NA
H
F7
U
N
* 00
8
J
F8
M
(
9
K
F9
O
H
дв
0
L
X
F10
P
command
>
W
î
{
[
alt
+ 11
option
F12
Next >
1
3](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F506bbbf0-95df-4280-b676-0179f995f37e%2F037a2f4a-347d-4de4-b1c2-a6b2ed7c2de8%2F6a4gzl8_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Mass of NaOH =
SC
Submit
Part E
Ascorbic acid, or vitamin C (C6H8O6, molar mass = 176 g/mol), is a naturally occurring organic compound with antioxidant properties. A healthy adult's daily requirement of vitamin C is 70-90 mg. A sweet lime contains 2.81x10-4
mol of ascorbic acid.
Mass of ascorbic acid U
Submit
Provide Feedback
To determine whether the ascorbic acid in a sweet lime meets the daily requirement, calculate the mass of ascorbic acid in 2.81x104 mol of ascorbic acid.
Express the mass in grams to three significant figures.
►View Available Hint(s)
7
alt
[VD] ΑΣΦ
option
Q
A
N
VG ΑΣΦΑΦ
16,388
2
-
F2
W
S
27
X
H
command
ㅁㅁ
#3
F3
E
D
$
?
54
C
www
B
DOD
F4
R
?
F
19
g
%
5
0
V
F5
T
G
MacBook Air
6
B
F6
Y
&
7
#tv NA
H
F7
U
N
* 00
8
J
F8
M
(
9
K
F9
O
H
дв
0
L
X
F10
P
command
>
W
î
{
[
alt
+ 11
option
F12
Next >
1
3
Transcribed Image Text:tab
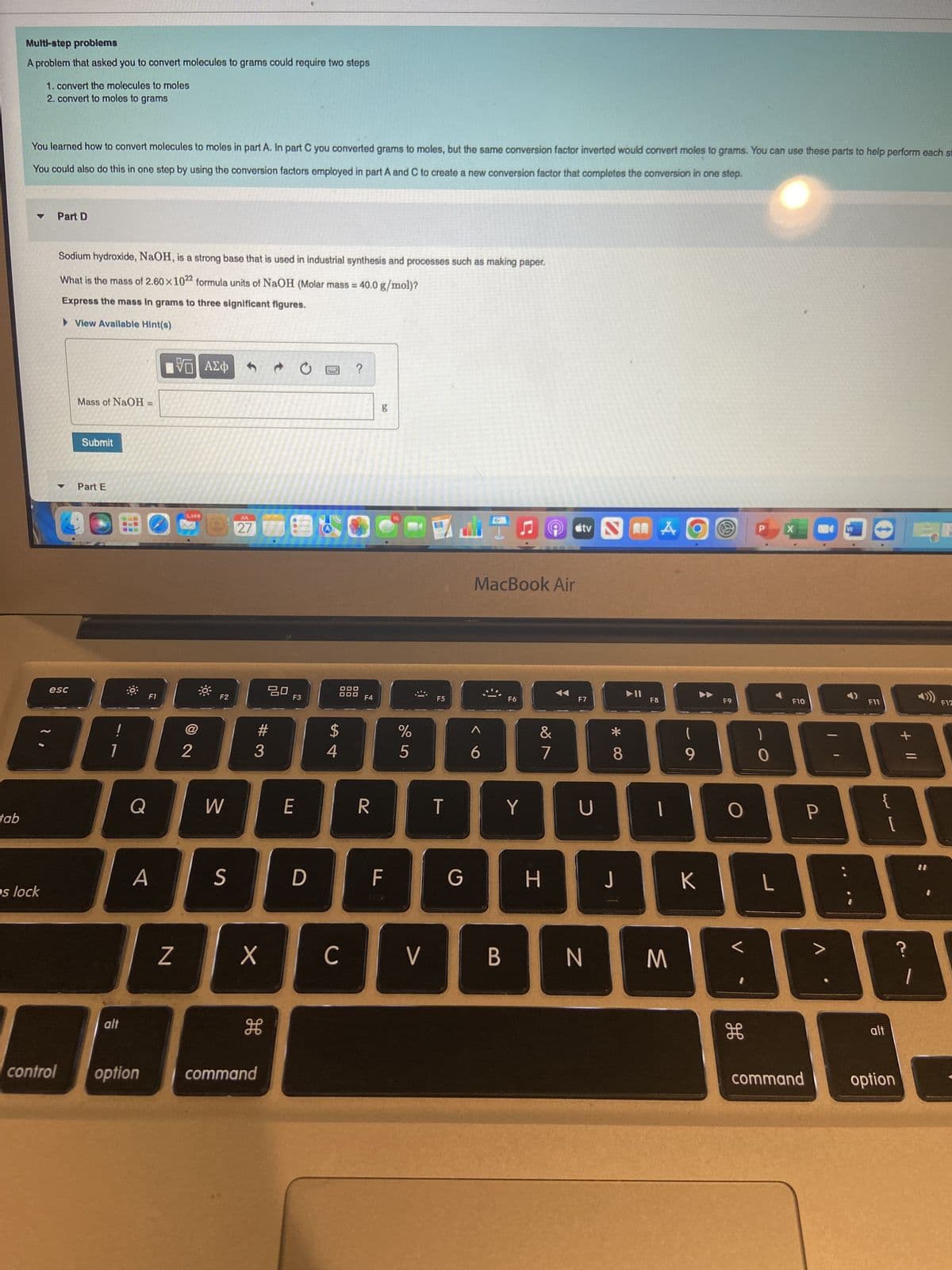
Multi-step problems
A problem that asked you to convert molecules to grams could require two steps
1. convert the molecules to moles
2. convert to moles to grams
You learned how to convert molecules to moles in part A. In part C you converted grams to moles, but the same conversion factor inverted would convert moles to grams. You can use these parts to help perform each s
You could also do this in one step by using the conversion factors employed in part A and C to create a new conversion factor that completes the conversion in one step.
ps lock
Part D
control
Sodium hydroxide, NaOH, is a strong base that is used in industrial synthesis and processes such as making paper.
What is the mass of 2.60×1022 formula units of NaOH (Molar mass = 40.0 g/mol)?
Express the mass in grams to three significant figures.
► View Available Hint(s)
esc
Mass of NaOH
Submit
Part E
1
alt
T
Q
A
option
IVE ΑΣΦ
Z
6,888
2
F2
W
S
JUL
X
#3
H
command
20
F3
E
D
P
DO
$
4
C
?
F4
R
g
F
16
%
5
V
F5
T
G
MacBook Air
6
B
F6
Y
&
7
H
tv
F7
U
N
*
8
J
➤II
F8
A
M
O
(
9
K
F9
H
0
L
X
F10
command
P
W
î
:
F11
alt
option
+ 11
F12
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER