Chapter25: Instruments For Optical Spectrometry
Section: Chapter Questions
Problem 25.20QAP
Related questions
Question
2
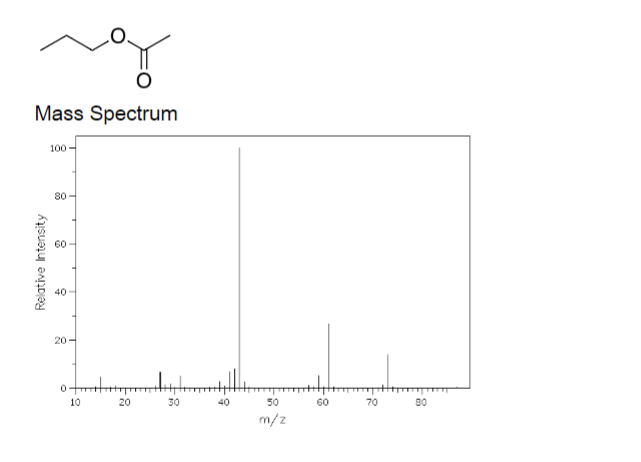
Transcribed Image Text:Mass Spectrum
100
80
60
40
20 -
ott
10
20
30
40
50
60
70
80
m/z
Relative Intensity
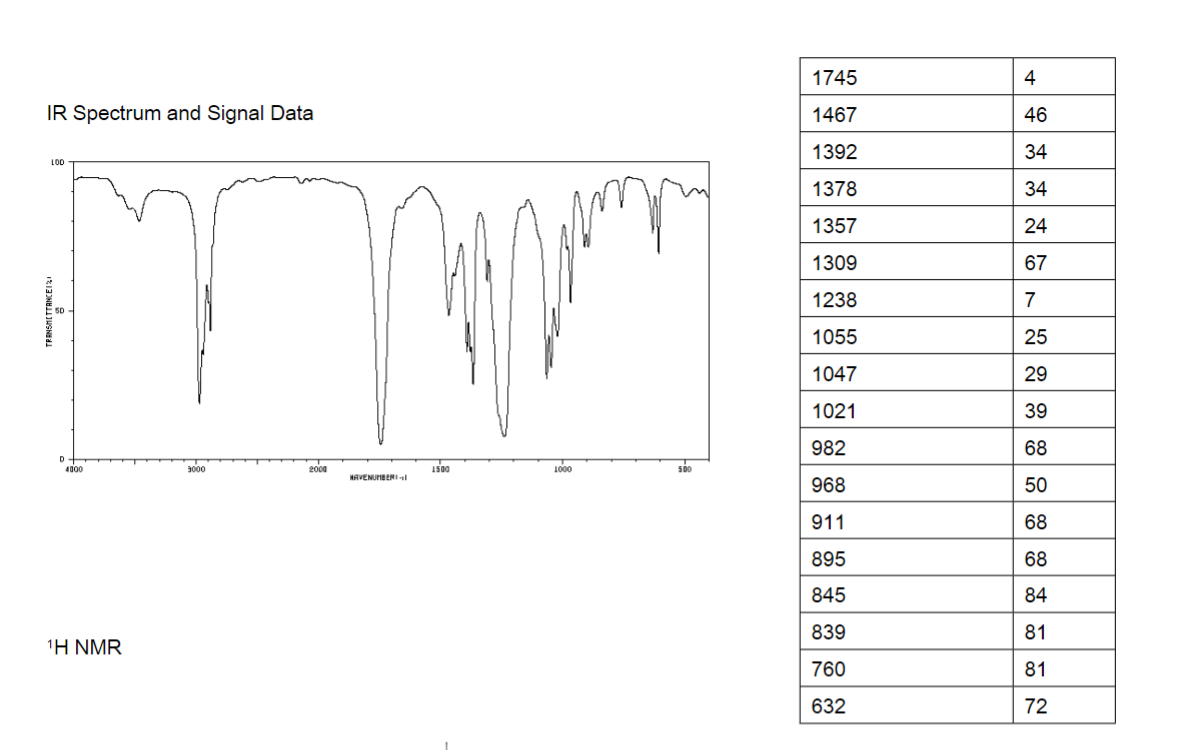
Transcribed Image Text:1745
4
IR Spectrum and Signal Data
1467
46
1392
34
LOD
1378
34
1357
24
1309
67
1238
7
1055
25
1047
29
1021
39
982
68
4000
20
1500
a000
NAVENUMBER al
968
50
911
68
895
68
845
84
839
81
1Η ΝMR
760
81
632
72
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning