Q: 28. If an enzyme with a Vmax of 4.3 µmol.min', and a Km of 150 µM was investigated under pre-set…
A: The Michaelis- Menten equation helps in estimating the relation between the rate of the reaction and…
Q: ant by replicate samples 7 Why is replicatio
A: As the question shows that we have replicate and how they are important for chemical analysis. This…
Q: 25 15 B 10 A Reaction progress - Use the reaction energy diagram above to answer the following…
A: A faster step requires lesser activation energy. After gaining activation energy,reactant forms an…
Q: 4. Derive a modified Michaelis-Menten equation for the following enzymatic reaction E+SES E+PEP…
A: The Michaelis–Menten equation is the rate equation for a one-substrate enzyme-catalyzed reaction.…
Q: 17 The equilibrwm constent Fer the fellewiny reachon Ts 5.0 xIc -8 at 25°c N3 Cg) F 3H, Lg 2 NHs Cg)…
A: There is a direct relationship between equilibrium constant and free energy of a reaction. Using…
Q: 2) a)Determine AGrxn° values for the following COmbustion reactions at 25°C b) Decide whether this…
A: Temperature = 25°C = 25 + 273 = 298K
Q: CN inhibits the oxidation of cytochrome aa3, what will be its consequence? O No electron loss for 02…
A: Ans. Option (d) is right. During electron transport system, NADH donates electrons to ubiquinone…
Q: 34. A key step in the formation of sulfuric acid from dissolved SO, in acid precipitation is the…
A: Given that, the mechanism of formation of sulfuric acid from the dissolved SO2 in acid precipitation…
Q: PGF2α is synthesized in cells using a cyclooxygenase enzyme that catalyzes a multistep radical…
A: Given:
Q: Calcelate Acteuatoin everge Tenp CE) 3१२ 451 Rate conschant (M-!s-1) 0,15 こO2333
A:
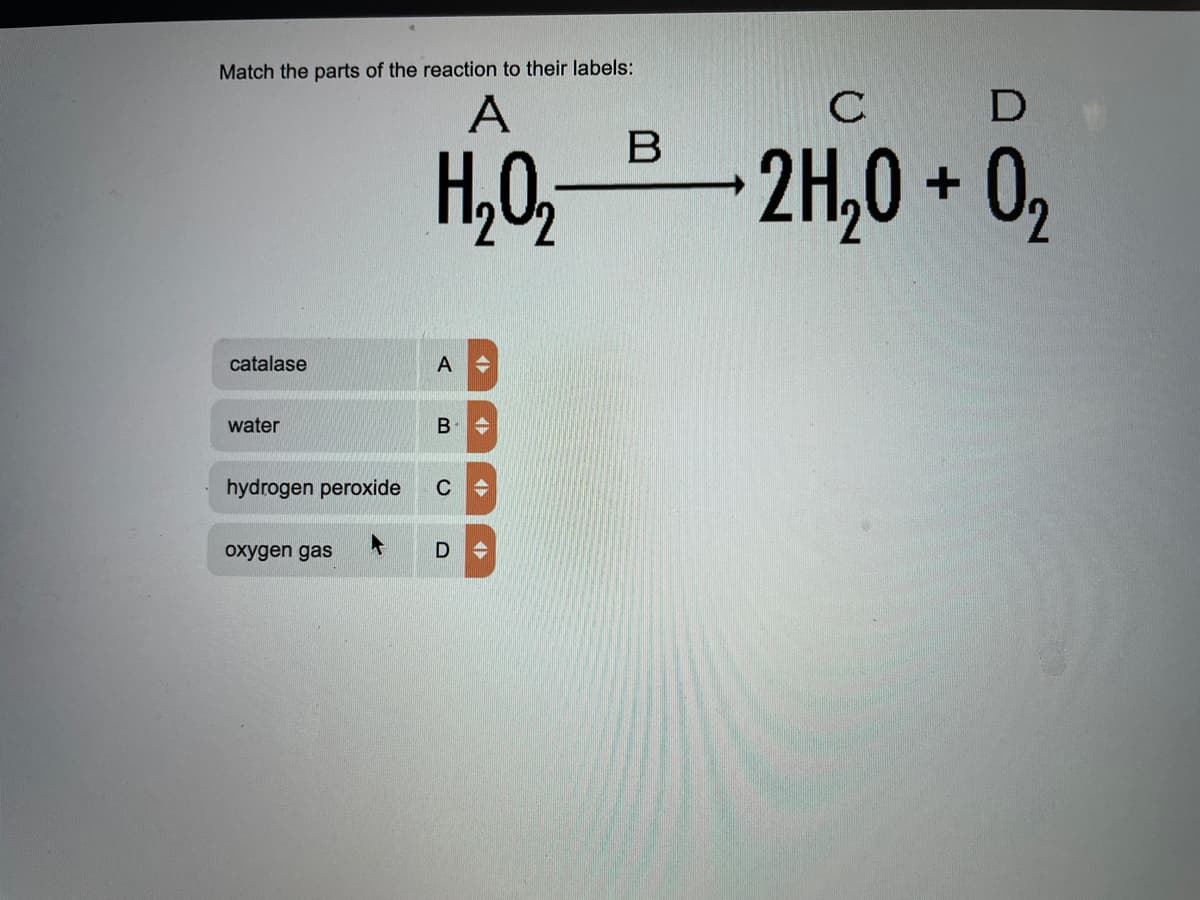
Q: From this activity, 1.) Which potato sample decomposed the most with hydrogen peroxide? 2.) Which…
A: ( a ) Boiled Potato section decomposed the most with hydrogen peroxide because with increase in…
Q: ow much is left 2.0 s later?
A:
Q: 4. The proton transfer step for the deprotonation of acetaminophen with potassium carbonate is shown…
A: The strength of an acid determines on the basis of pKa value. The pKa is defined as follows: pKa =…
Q: 3. A bacteria is able to grow on hexadecane as a substrate: C16H34 + 16.28 02 + 1.42 NH3 → 1.65…
A:
Q: 5. Will the reactions shown below go the direction as indicated? -H- + CH3MgBr CH4 pKa = 25 pka = 50…
A: The reactions taking place are given as,
Q: (d) What are the main hazards of disperse blue 14? (e) What are the main hazards of quinazarin…
A:
Q: Complete any two of the following reactions with mechanism. Name the reaction involved: сно NaOEt,…
A: The organic reactions are carried out with the help of various reagents. Every molecule has a…
Q: Describe the following 1. Hydrocarbons ( saturated) 2. Hydrocarbons ( unsaturated) 3. Aldehydes,…
A: Saturated hydrocarbons are the hydrocarbons that are made of Carbon and hydrogen. There is no double…
Q: 70 100 56 MS-NW-0546 80- 43 60 40 29 31 20 83 98 10 20 30 40 50 60 70 80 90 100 Rélative Intensity
A: Alpha cleavage in mass spectrometry is a characteristic fragmentation of the molecular ion derived…
Q: + HS- SH + I a. -AG b. K<1 c. Reactants are favored d. Ituction progeas
A:
Q: ritical reaction in the production enosine triphosphate, ATP, to aden ATP(aq) + H,O(1) → ADPO which…
A: Reaction ATP (aq) + H2O (l) →ADP (aq)+HPO42- (aq) G = ΔΔG0 + RT ln K = ΔG0 + 2.303 RT log K…
Q: Which of the following is related to adsorption? I AH= - ve II A S= + ve III -TAS= - ve AG= - ve A)…
A:
Q: Theophylline was administered to a patient at dosing rates of 600 mg/day and 1200 mg/day and the…
A: In the drug pharmacokinetics, the relation between the drug concentration and the rate of the…
Q: Solve the attached file
A: Benzene undergoes Nitration in the presence of nitric acid and sulphuric acid. NO2+ acts as an…
Q: 120 100 80 PE 60 40 reaction pathway Jse the above potential energy diagram to answer the following…
A: Applying concept of potential energy representation in the diagram potential energy versus to the…
Q: Note these acronyms: THF = tetrahydrofuran; D = heavy H ( 21H one extra neutron) Ph = Phenyl = C6H5;…
A:
Q: 20.23 The mutarotation of glucose is first order in glucose con- centration and is catalyzed by…
A: The values of k0 and kA are written below:-
Q: PO-8. What is the major product of this reaction? JI HA for at X OH (A) (B) (C) (D)
A: Since, this question is a multiple questions. So, I will solved first question-8 for you. we will…
Q: Xylitol, which is used as a sugar substitute, is produced by the reduction of D-xylose. Modify the…
A: The conversion of the aldehyde group (-CHO) to alcohol (-CH2OH) is the reduction reaction. In this…
Q: Which of the following is false regarding the iodine clock experiment? a. when [KCI] and [K2SO4]…
A: We know Reaction rate is directly proportional to the no. Of effective collisions,which depends upon…
Q: PROPOSE THE MECHANISM FOR REACTION. EXPLAIN HOW THE REACTION OCCURS AND WHAT KIND OF REACTION. 1)…
A:
Q: Explain why pyrrole (pKa ~ 17) is a much stronger acid than ammonia (pKa = 36).
A: There are some definitions which can define acids and bases on the basis of their behaviors. Three…
Q: A chemist tries the reaction below and is surprised to find the target is not made. Provide two…
A: The given reaction is friedal craft acylation, in this there are 2 steps i.e. generation of an…
Q: A certain drug has a half-Iife in the body of 2.5 h. What should the interval between doses be, if…
A: The half-life of the drug in the body is = 2.5 h The interval between doses be, if the concentration…
Q: the mathematical expression of Ksp of calcium (II) of phosphate is: Select one: O a. Ksp [Ca²+1³…
A: The equilibrium between the soluble ions and insoluble salt can be described by the solubility…
Q: NEW August 15, 2020. What is the pNa of a solution that is 100 ppm in Na,SO,? Select one: O a. 2.85…
A: Given data contains, Concentration of Na2SO4 solution is 100 ppm. Now, we have to calculate pNa of…
Q: NaH O Meo- Ome
A: The reactions involving organic compounds are called organic reactions. There are different types of…
Q: 0.4 Inhibitor A Inhibitor B 0.35 0.3 No inhibitor 0.25 0.2 0.15 -3 -2 -1 3. 5 -0.05 1/(S] (10, M)…
A: Equation of line of inhibitor B is (in the form of y=mc+c) y = 0.75x + 0.15 From graph and this…
Q: „Br K EtO EC-Li+ L+M (inorganic byprodu Br
A:
Q: Draw the correct products, in either order, for the following oxidative cleavage reaction (1) кМпO,,…
A: The basic KMnO4 followed by treatment of acidified water cleaves the double bond forming carbonyl…
Q: 1.Predict the chemical name of B 2. What is the reaction name for the chemical transition of A to…
A: The reaction with products are as shown below.
Q: Which of the following binding constants has the highest affinity?
A:
Q: Trypsin is a serine protease that has a KM of 15 mM. At an initial substrate concentration of 250 µM…
A: Given that, for a serine protease trypsin The Michaelis-Menten constant is KM = 15 mM = 15×10-3 M,…
Q: The reaction: Mn(CO);Me + CO - Mn(CO)s(COME) is an example of: Select one: O a. an oxidative…
A: This given reaction is one of the step in the organometallic reaction . In this reaction CO ligand…
Q: Deuterium oxide, D20, is water that contains the stable hydrogen isotope ?H, usually shown as D.…
A: Correct option is (D)
Q: Carbonic anhydrase is one of the fastest enzymes known. If 10 µg of this enzyme catalyzes the…
A:
Q: "N+H;C+;He Calculate the energy released (MeV) in the reaction. Given,
A: Energy is calculated by the given energy= energy of products+releasee energy.
Q: structures has activity like acetylcholine? Et HC H3C C.
A: Answer : Structure has activity like acetylcholine : The correct option is 4th option i.e…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Hydrolysis of 1 M glucose 6-phosphate catalyzed by glucose 6-phosphatase is 99% complete at equilibrium (in other words, only 1% of the substrate remains). Calculate the DG0 of the reaction by considering all standard factors had been fulfilled2 1. Calculate the number of moles of Pd that were added. The catalyst weighed is 10% by mass Pd. Mass of Palladium carbonate: 0.903g 2. How many molar equivalents, versus 5-iodovanillin, of Pd were added? Mass of 5-iodovanillin: 0.355gFor the reaction 5Ce4+ + Mn 2+ + 4H2 O → 5Ce3+ + MnO4- + 8H+ , given E0 (Ce4+ /Ce3+ ) = 1.70V,E0 (MnO4- /Mn2+ ) = 1.507V, calculate ΔG0 for this reaction ?
- What are the dissociation reaction, including phases, of AICI3?Explain how the light naphtha isomerization catalyst is bifunctional.What is the location in the catalyst of the different kinds of sites and what function do they serve?one experiment involved the addition of magnesium metal to concentrated hydrochloric acid. 1. what type of reaction is this? 2. describe how the product(s) of the reaction was detected
- An example of a heterogeneous catalyst is: a. Wilkinson’s catalyst for the hydrogenation of alkenes b. Grubb’s catalyst with varied substituents for olefin metathesis in THF c. Pt and Pd nanoparticles on ceramic honeycombs in catalytic converters d. Pd(PPh3)4 in toluene solvent used for Miyaura-Suzuki coupling reactions( plz asap within 5 min....plz dont answer after 9.00 PM ..i will write plagarism if u answer after 9.00 pm6. Sodium chloride, β-D-fructofuranosyl α-D-glucopyranoside or sodium chloride - β-Dfructofuranosyl α-D-glucopyranoside solutions are used for dehydration of pumpkin.a) Estimate the water activity of 30% β-D-fructofuranosyl α-D-glucopyranoside solution.b) Estimate the water activity of 30% sodium chloride solution.c) Estimate the water activity of solution 20% sodium chloride and 20% β-Dfructofuranosyl α-D-glucopyranoside solution.d) What do you think about the effectivity of these solutions if the water activity of pumpkins are 0.945?
- How did the rate change as the concentration of the S2O8-2 was halved. Expalin at molecular level.Talk about how you might tell whether the oxidation of [Fe(OH2)5SCN]2+ by Cr2+(aq) occurs by an inner or outer sphere mechanism. Write the chemical equations for key steps of these competing pathways.The ampicillin monograph states that the reconstituted suspension is stable for 14 days in a refrigerator (50C). If the product is left at room temperature (250C) for 6 hours, what is the reduction in the expiration period? (Assume Q10 = 3) Ans is 11.75 days please help Do not give handwriting solution.

