Melting Boiling Point Point Molar Density (g/mL) Chemical Notes Mass (g/mol) (°C) (°C) Acetone CAS: 67-64-1 Ethanol CAS: 64-17-5 CH;CH,OH 46.07 -114 78 0.789 2,4- Dinitrophenylhydrazine (2,4-DNP) CAS: 119-26-6 Chromium(VI) oxide CAS: 1333-82-0
Melting Boiling Point Point Molar Density (g/mL) Chemical Notes Mass (g/mol) (°C) (°C) Acetone CAS: 67-64-1 Ethanol CAS: 64-17-5 CH;CH,OH 46.07 -114 78 0.789 2,4- Dinitrophenylhydrazine (2,4-DNP) CAS: 119-26-6 Chromium(VI) oxide CAS: 1333-82-0
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Can you please fill the rest of this chart
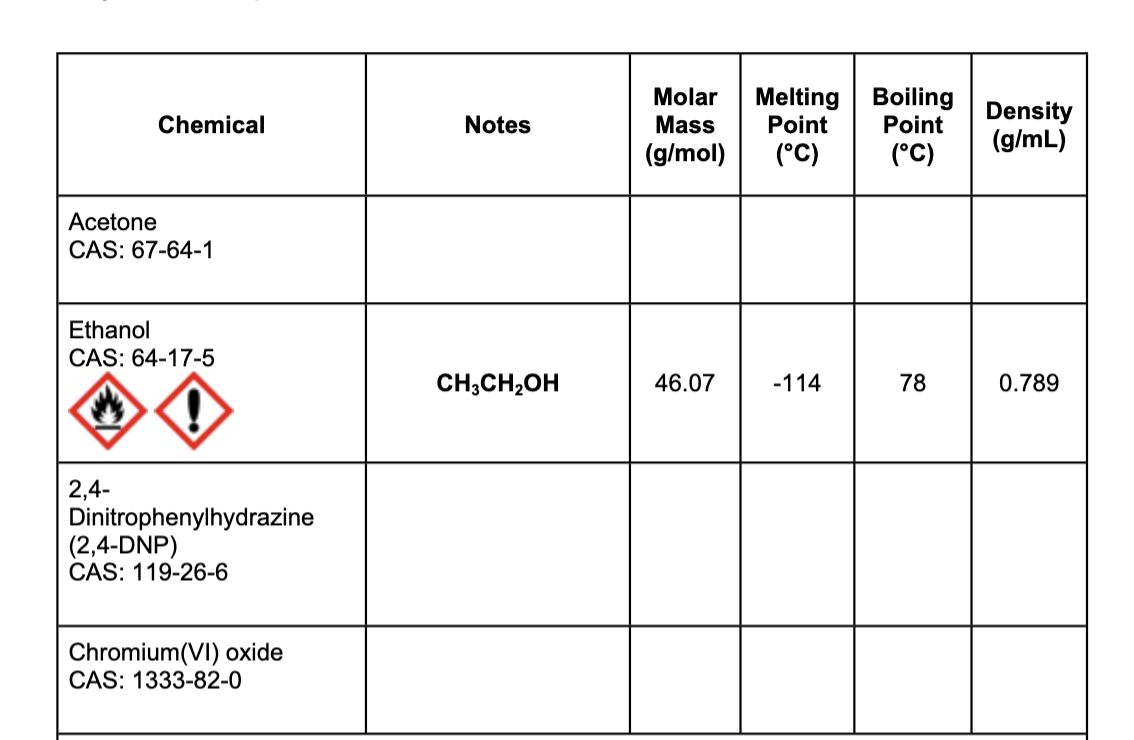
Transcribed Image Text:Molar
Melting
Point
Boiling
Point
Density
(g/mL)
Chemical
Notes
Mass
(g/mol)
(°C)
(°C)
Acetone
CAS: 67-64-1
Ethanol
CAS: 64-17-5
CH;CH,OH
46.07
-114
78
0.789
2,4-
Dinitrophenylhydrazine
(2,4-DNP)
CAS: 119-26-6
Chromium(VI) oxide
CAS: 1333-82-0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

