Q: Which of the following is not a property of aromatic hydroca O A. Reactive to electrophilic reagents...
A: Option E Non Planar is the only property which are not acquire by Aromatic Hydrocarbons. 1)As all we...
Q: Use the interactive to measure the densities of several different solids and liquids. LIQUID SOLID T...
A: All the transition metal are malleable and ductile.
Q: Convert 16.3 L/cm to units of milliliters per meter. Show the unit analysis by dragging the conversi...
A: Given :- 16.3 L/cm To convert :- 16.3 L/cm to mL/m
Q: 10 - The opposite diagram is related to the change of energy states in the octagonal complexes of wh...
A:
Q: 5. Which of the following represent isotopes of the same element? А: 34 B: 79x C: 78x 34 D: 81x 35 3...
A:
Q: Identify the following spectra. label 5-6 peaks. Label the peaks on the spectrum place the structu...
A: Interpretation: To identify the IR peaks on the spectrum and identify the structure.
Q: 2. As the director of a research laboratory you are paid to decide if there is a significant differe...
A:
Q: A solution of HNO3 is standardized by reaction with pure sodium carbonate: 2H+ + Na2CO3 → 2Na+ + H2O...
A:
Q: Calculate the amount of work done, in joules, when 2.5 mole of H2O vaporizes at 1.0 atm and 25°C. As...
A:
Q: This is a Brønsted-Lowry acid-base reaction. Draw structures for all the products, showing any non-z...
A:
Q: Consider the reaction: CH4(g) + CCl4(g) → 2CH2Cl2(g) Given an initial mass of 18.25 g CH4, an exce...
A: Given that - balanced Chemical equation - CH4(g) + CCl4(g) ---------> 2CH2Cl2(g) Molar masses...
Q: Classify these substances. More than one answer may apply in each case. H2 O, pure substance element...
A: Given substances are : a). H2 b). O2 c). H2O d). Salt water They are pure substances or element ...
Q: The following initial rate data are for the reaction of ICl with hydrogen: 2 ICl + H2I2 + 2 HCl E...
A: Given reaction is 2 ICl + H2 ----> I2 + 2 HCl Given experimental data is Experiment [ICl]o, ...
Q: Method 1 Fill a burette with the solution of NaOH(aq) of unknown concentration. Pipette 25.00 cm' of...
A: Given: Volume of HCl = 25.00 cm3 Molarity of HCl = 0.10 mol dm-3 Volume of NaOH = 24.7 cm3
Q: When 18.5 g of HgO(s) is decomposed to form Hg(l) and O2(g), 7.75 kJ of heat is absorbed at standard...
A:
Q: What is the electronegativity of Ne?
A:
Q: what is the ph of a buffer solution composed of 0.50m formic acid and 0.70m sodium formate
A: Answer: 3.90 Given buffer solution contains formic acid, HCOOH , a weak acid, and sodium formate, ...
Q: I need the answer as soon as possible
A: Dear student, this question is related to orgel diagram.
Q: Draw the Lewis structure of the missing reactant.
A: Lewis Structure is defined as a representation of the valence shell electrons present in a molecule....
Q: Please provide the 2 conversion factors (CF) that correspond to each equality: 1L = 1000 ml .......(...
A: A conversion factor is an expression used for the change in units of a measured quantity. The conver...
Q: elect an orthorhombic unit cell that indicates the locations of thel-0 and- 4 3 4 point indices. Not...
A: (1,1/2,0) represents ( x,y,z) coordinates .
Q: A metal, M, of atomic mass 56 amu reacts with chlorine to form a salt that can be represented as MCl...
A: Given - Formula of salt = MClx Atomic mass of M metal = 56 amu Mass of salt used , MClx = 35.5 g M...
Q: a) Draw the electron dot diagram for the molecule CH20 b) Predict the shape around the central atom ...
A:
Q: Assume the recommended single dose of ibuprofen for children over the age of two is 4.5 mg per pound...
A: Given that - The assigned dosage of ibuprofen is = 4.5 mg per pound Here one pound body weight req...
Q: eaks are assigned on the spectrum (~5-6 peaks). Label the peaks on the spectrum and place the struct...
A: The answer is given below-
Q: Draw the partial (valence-level) orbital diagram and write the symbol, group number, and period numb...
A: Electronic configuration represent the arrangement of an electron in an orbital in increasing order ...
Q: An element has a density of 10.25 g/cm3 and a metallic radius of 136.3 pm. The metal crystallizes in...
A: For BCC lattice root 3 a = 4r where a = edge length and r = radius No. of atoms in BCC (Z) = 2 form...
Q: Results and Discussion: Schiff's Test: Samples: tyrosine, lysine, arginine Reagents: rosanilin...
A: A question based on qualitative analysis, which is to be accomplished.
Q: What were the main needs to develop colloidal technique for powder processing, explain your argument...
A: Main needs to develop colloidal technique for powder processing.
Q: 12.Consider this reaction, which occurs in the atmosphere and contributes to photochemical smog: P2...
A: 1-First we will convert grams of P2O5 to moles : Molar mass of P2O5 = 142gmol-1 Moles of P2O5 = ( ...
Q: Gasoline is composed of a variety of different liquid hydrocarbons, which do not separate as time pa...
A: Given information, Gasoline is composed of a variety of different liquid hydrocarbons, which do not ...
Q: What mass of K2CO3 is needed to prepare 80.0 mL of a solution having a potassium ion concentration o...
A: We have to calculate the mass of potassium carbonate.
Q: 7- Full Blue Prussia KFe[Fe(CN)6] Intense absorption bands in It has 500 and 25,000 cm! What type of...
A: Following is the appropriate answer of the given question.
Q: A sample of an unknown pure substance X is analyzed and found to contain 8.40 g of Element E,, 9.60 ...
A:
Q: 7- Full Blue Prussia KFe[Fe(CN)6] Intense absorption bands What type of electronic in It has 500 and...
A: Following type of transition is seen incase of Blue Prussia.
Q: Nickel has a face-centered cubic structure with an edge length of 352.4 picometers. What is the dens...
A:
Q: BIUA A - 4. Fluorine Neon Nitrogen
A: The modern periodic table was created by Mendeleev.
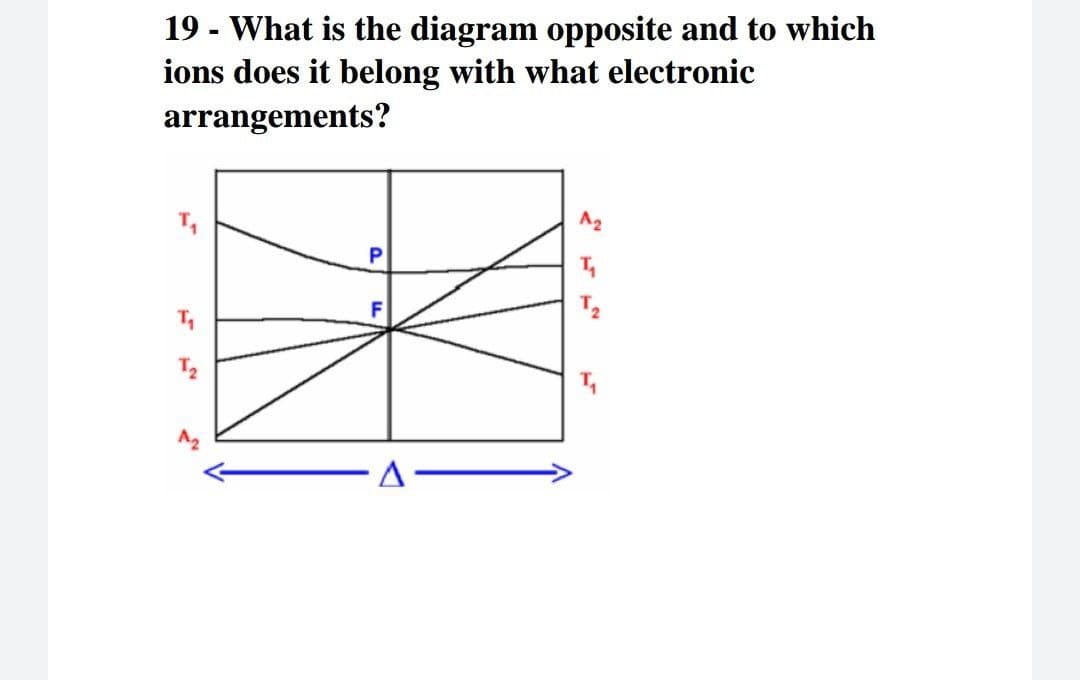
Q: 17- Electron spectrum Ti (H2O)6*³ compared to the electron spectrum Nd*3 It is very wide (Several na...
A: Question is based on the concept of inorganic chemistry. We have to explain the electronic spectra ...
Q: I need the answer as soon as possible
A: Dear student this question is related to crystal field splitting.
Q: What are the indices for the direction represented by the vector that has been drawn within a unit c...
A:
Q: The unbalanced equation for the reaction between scandium metal and hydrochloric acid is: Sc(s) + HC...
A:
Q: 11- In cases A to C, specify the weakest electron transfer. A) Ni (H2 O)6+2 : A2g → Tıg or A2g → Tig...
A: From A to C the weak electronic transfer is A2g3 to T1g1 Since According to the selection rule spin ...
Q: 2 .What is the 2nd most prevalent element in soil (name, not symbol)? 3. What is the name of the...
A:
Q: The incomplete Lewis structure below shows all the atoms and sigma bonds for a particular molecule, ...
A: In this question, we will show the complete Lewis structure for given incomplete Lewis structure. Y...
Q: +3 16- Electron spectrum Cu (H20),* compared to the electron spectrum Ti (H2O)6*³is +3. very wide Wh...
A: The question is based on the concept of coordination chemistry. we have to describe the nature of ba...
Q: MnO2 + 4HCl ---> MnCl2 + Cl2 + 2H2O If 0.86 mole of MnO2 reacts with 48.8 g HCl, and 24 g of...
A: Mole of MnO2 = 0.86 mol Mass of HCl = 48.8 g Mass of Cl2 = 24 g Percent yield = ?
Q: For which of these reactions will the difference between ΔH° and ΔE° be the smallest? Multiple C...
A:
Q: For your new candle-making business, you need to purchase a large amount of a unique scented wax. Yo...
A: This question involve unit conversion.
Q: 17- Electron spectrum Ti (H2O)6*³ compared to the electron spectrum Nd** It is very wide (Several na...
A: We explain the reasoning for different electronic spectra of neodymium and titanium complexes.
Q: Give the formula of a compound which will give a precipitate with KI and K3PO4? SOLUBILITY RULES ...
A: (a) ZnCl2 + 2 KI --------> ZnI2 + 2 KCl All salts of Cl-, Br-, I- are soluble in water (except A...
Step by step
Solved in 2 steps with 1 images

- Define the Mechanism- Addition of H and BH2—Hydroboration ?I. Molecular rearrangementsA) How are 1,2-hydride shifts in carbocation intermediates formed during SN1 or E1-type processes? How are carbocations formed by treating alcohols with water? How can you predict when a hydride shift will occur and how can you show it using arrow-pushing strategies?(a) A compound known to be a substituted cyclohexanone derivative has lamda max of 235 nm. Could this compound be a conjugated dienone? explain (b) (i)For this compound, how many nm must be accounted for by substituents? (ii) What are the substituents and the points of substitution that may occur having accounted for the 20nm?
- Which is higher in energy? Q.) Infrared radiation of 1715 cm-1 or of 2800 cm-1?When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.(a) Propose structures for products A and B. (Hint: In many cases, an allylic carbocationis more stable than a bromonium ion.)This is an example of a [1,3] sigmatropic rearrangement. Can someone explain how this is 1,3 (number the carbons) and draw the anion / cation transition state
- Give detailed answer- Provide example of compound that fulfil each criteria below. Explain why-A)1,2-disconnectionB)1,4-disconnectionC)1,5-disconnectionD)1,6-disconnectionThere is a different, thermally allowed cycloaddition of two butadiene molecules. Showthis reaction, and explain why it is thermally allowed. (Hint: Consider the dimerizationof cyclopentadiene.)15-13 UltravioletThe molecular formula of unknown compound B is C10H16.Quantitative brominationof a 0.473-g sample of compound Brequired 48.22mL of a 0.216MBr2/CCl4to produce a color change. How many rings and pi bonds does compound Bcontain?Include any explanations/calculations to justify your answers.
- at 25C, which of the following reactions of PbS and H2O2 is more favourable?1) PbS + 4H2O2 -> SO2 + PbO2 + 4H2O2) PbS + H2O2 -> PbSO4 + H2OArrange the following reactions in order of increasing ΔrxnS and briefly explain your reasoning. a) S(s)+O2(g)⟶SO2(g) b) H2(g)+O2(g)⟶H2O2(ℓ) c) CO(g)+3H2(g)⟶CH4(g)+H2O(ℓ) d) C(s)+H2O(g)⟶CO(g)+H2(g)Arrange the following reactions in order of increasing ΔrxnS and briefly explain your reasoning. a) S(s) + O2(g) ⟶ SO2(g)b) H2(g) + O2(g) ⟶ H2O2(ℓ)c) CO(g) + 3 H2(g) ⟶ CH4(g) + H2O(ℓ)d) C(s) + H2O(g) ⟶ CO(g) + H2(g)



