Method 1 Fill a burette with the solution of NaOH(aq) of unknown concentration. Pipette 25.00 cm' of the standard solution of 0.10 mol dm3 HCI(aq) onto a clean conical flask. 3 Add a few drops of the indicator bromothymol blue solution to the conical flask and stand it on a white tile or, white paper. 4 Titrate the NaOH against the HCI, until the endpoint of the indicator is observed, and record the volume added from the burette. 5 Repeat the titration until final values within 0.05 cm3 are obtained. Results Volume NaOH Titration 1 Titration 2 Titration 3 end volume / cm3 + 0.05. 24.7 24.8 24.6 start volume / cm3 + 0.05 titre / cm3 + 0.10 24.7 24.8 24,6 average titre/ cm' ± 0.10 = 24.7 Also, record your qualitative data. Analysis Calculate the number of moles of HCI that reacted. By reference to the reacting ratio of HCl and NaOH, deduce the number of moles of NaOH required to reach equivalence.
Method 1 Fill a burette with the solution of NaOH(aq) of unknown concentration. Pipette 25.00 cm' of the standard solution of 0.10 mol dm3 HCI(aq) onto a clean conical flask. 3 Add a few drops of the indicator bromothymol blue solution to the conical flask and stand it on a white tile or, white paper. 4 Titrate the NaOH against the HCI, until the endpoint of the indicator is observed, and record the volume added from the burette. 5 Repeat the titration until final values within 0.05 cm3 are obtained. Results Volume NaOH Titration 1 Titration 2 Titration 3 end volume / cm3 + 0.05. 24.7 24.8 24.6 start volume / cm3 + 0.05 titre / cm3 + 0.10 24.7 24.8 24,6 average titre/ cm' ± 0.10 = 24.7 Also, record your qualitative data. Analysis Calculate the number of moles of HCI that reacted. By reference to the reacting ratio of HCl and NaOH, deduce the number of moles of NaOH required to reach equivalence.
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.48QAP
Related questions
Question
By reference to the reacting ratio of HCl and NaOH, deduce the number of moles of NaOH required to reach equivalence.
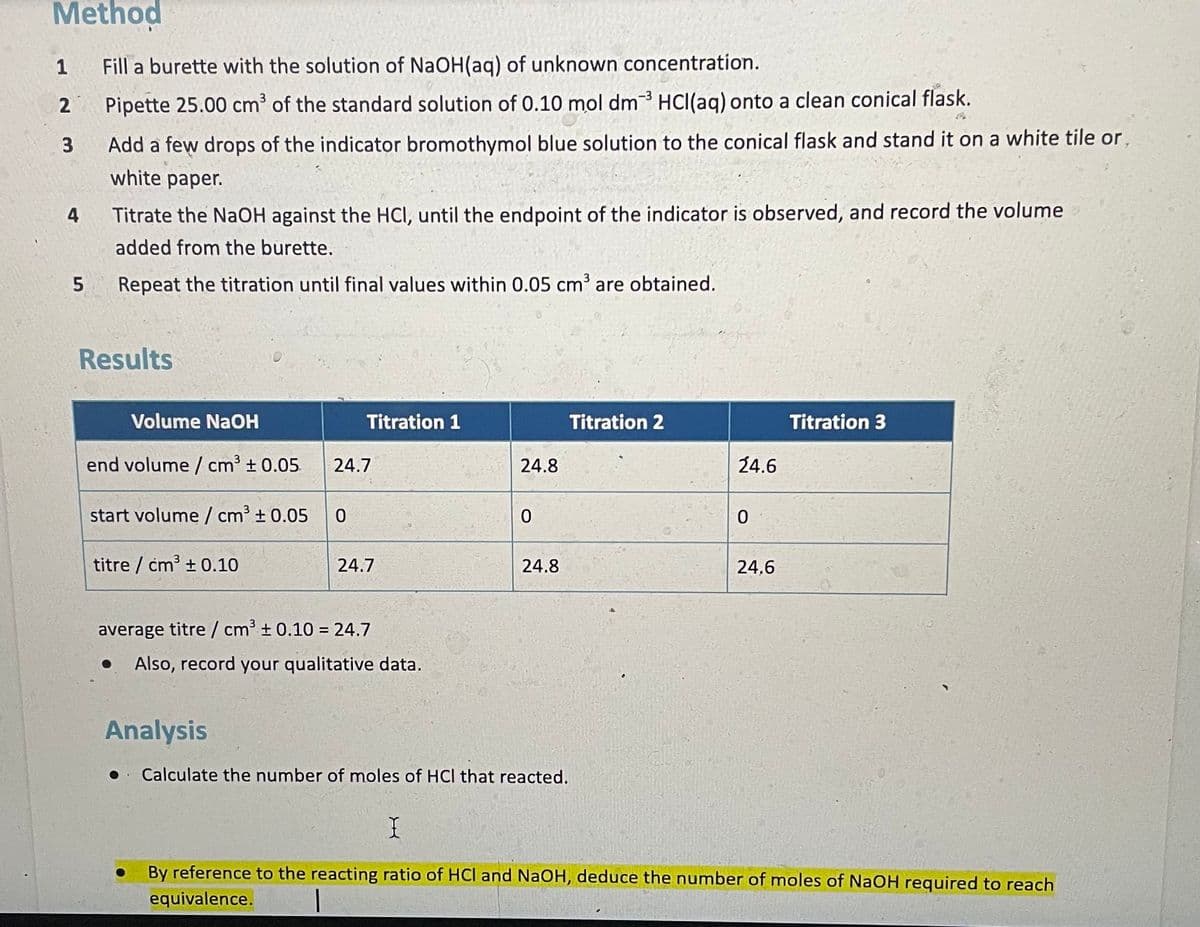
Transcribed Image Text:Method
1
Fill a burette with the solution of NaOH(aq) of unknown concentration.
-3
2
Pipette 25.00 cm of the standard solution of 0.10 mol dm HCI(ag) onto a clean conical flask.
3
Add a few drops of the indicator bromothymol blue solution to the conical flask and stand it on a white tile or,
white paper.
4
Titrate the NaOH against the HCI, until the endpoint of the indicator is observed, and record the volume
added from the burette.
Repeat the titration until final values within 0.05 cm³ are obtained.
Results
Volume NaOH
Titration 1
Titration 2
Titration 3
end volume / cm³ ± 0.05
24.7
24.8
24.6
start volume / cm³ ± 0.05
titre / cm³ + 0.10
24.7
24.8
24,6
average titre / cm' ± 0.10 = 24.7
Also, record your qualitative data.
Analysis
Calculate the number of moles of HCl that reacted.
By reference to the reacting ratio of HCl and NaOH, deduce the number of moles of NaOH required to reach
equivalence.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning