Q: How does one determine the expected Salinity of a water sample via applying constant proportions for…
A: Salinity is a measure of the concentration of dissolved salts in seawater and is a crucial factor in…
Q: The Quantum Mechanical Model of the atom was proposed by:
A: This model explains the recent understanding about any atoms, which is commonly described as wave…
Q: MS: M+ is not seen but equals 144. i. Relative Intensity IR: 120 100 80 20 0 27 29 30 373840 40 M…
A: Since its a multiple subpart question, only first-three subparts needs to be answered if not…
Q: What does the following equation represent? Ca5(PO4)3OH -----> 5 Ca²+ + 3 PO4³- + OH the production…
A:
Q: 10: 14) Recall that the bidentate ligand, ox² = Draw the structure of Fe(ox)(NH₂). Draw the metal…
A: A bidentate ligand is one that is capable to donate two pairs of lone pairs to the central atom.…
Q: Rank from lowest to highest entropy. A. 1 mol of CCl4 gas at 273 K and 40 L. B. 1/2 mol of liquid…
A: We need to Rank from lowest to highest entropy. A. 1 mol of CCl4 gas at 273 K and 40 L. B. 1/2 mol…
Q: Why do we say the two enzyme-catalyzed reactions taking place in your test tube are "coupled"? O…
A: First glucose oxidase produce glconic acid and hydrogen peroxide from glucose. Then these peroxide…
Q: 2) Lowry's assay requires the sample protein concentration under the range of
A: Introduction The Lowry protein assay is a widely used technique that enables the quantification of…
Q: 12. a) Consider Ozone, Os. 1) Draw the Lewis structure. Include all resonance structures and formal…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: P2.34 One mole of an ideal gas is subjected to the following changes. Calculate the change in…
A: Answer: These questions are based on first law of thermodynamics which states that: ∆U=q+w Here:…
Q: To answer the questions, interpret the following Lewis structure for CO3²-. For the central carbon…
A: Lewis dot structure is diagrammatic representation of molecules in which bonding and non bonding…
Q: Consider a double replacement reaction between Zinc (ll) chloride and sodium hydroxide: ZnCl2+NaOH…
A: Given that - Following half reaction - ZnCl2 + NaOH ==> Here zinc (II) chloride is a salt…
Q: The autoionization of water can be expressed with the following balanced chemical equation: 2H₂O (1)…
A:
Q: What are the formal charges on these I am lost
A: Formal charge is charge assigned to the atoms in a molecule after formation of bonds.
Q: You add 15 ml of 0.15 M NaCl to a container containing 15 ml of 0.15 M HCI. What is the pH of the…
A: In this question, we will determine the pH of the HCl solution when we adding NaCl No effect of…
Q: How much glucose do you need to make 500 ml of 5% glucose?
A:
Q: Why is it that the ionic activity is equal to the molality (mol/kg) in DHLL (Debye-Hückel Limiting…
A: Why is it that the ionic activity is equal to the molality (mol/kg) in DHLL (Debye-Hückel Limiting…
Q: 4. A galvanic cell consisting of a magnesium electrode and a standard hydrogen electrode is…
A:
Q: solution is made by mixing 40.0 mL of ethanol, C₂H,O, and 60.0 mL of water. Assuming ideal behavior,…
A:
Q: A common misconception about vapor pressure lowering is that the solute occupies some of the surface…
A: Vapor pressure lowering is the dercrease in pressure exerted by solvent molecules due to the…
Q: particular element has two naturally occurring isotopes. The first makes up 20.000% of the total and…
A:
Q: 9. One step in the isolation of pure rhodium metal (Rh) is the precipitation of rhodium(III)…
A:
Q: F F X CF₁
A: Hydrogen bonding has a significant effect on the volatility of a substance. Hydrogen bonding is a…
Q: 2. Name the following compound: 32
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of…
Q: Nitrogen dioxide gas has what volume @STP when its mass is 491.2g? round to 2 decimal places.
A: In this question, we will determine the volume of NO2 in L unit at STP. At STP, T = 0°C = 273K P =…
Q: How many atoms of all kinds are contained in 42.1 grams of calcium carbide (Ca2C)? Ans: 8.25 x 10^23…
A: Given : Mass of Ca2C = 42.1 gm Molar mass of Ca2C = 92 gm mol-1…
Q: Be sure to complete step ONE before moving on to the other steps. Break each step down. You should…
A: Introduction A double replacement reaction is a type of chemical reaction that occurs when two…
Q: solve these problems 1. Determine the work done by the system against a pressure of 7.3 atm given a…
A:
Q: Calculate Mn, Mw and the polydispersity index for a hypothetical polymer sample that contains…
A: The molecular weight of a polymer is an important characteristic that determines its physical and…
Q: Make a ramp table with the following chemicals: -Hexanes -Acetone -Dichloromethane
A: We need to Make a ramp table with the following chemicals: -Hexanes -Acetone -Dichloromethane
Q: According to the nutritional information on a package of jasmine rice, each serving (1/4 cup or 45.0…
A: According to the nutritional information on a package of jasmine rice, each serving (1/4 cup or 45.0…
Q: A solution of urea (MW = 60 g/mole) in water has a concentration of 2.41 m (where m = molality,…
A: Molality of urea solution = 2.41 m Density of solution = 2.0 g/mL Molarity of solution = ?
Q: This part is very confusing.
A: Amount of heat,Q required to change the temperature of a substance of mass ,m and specific heat…
Q: 6. For the following sets of compounds, show using resonance structures, how the formal charges are…
A:
Q: Consider 1.70 mol of carbon monoxide and 3.00 mol of chlorine sealed in a 3.00 L container at 476…
A: Dear student since you have posted multiple questions; As per BARTLEBY QnA guidelines we are allowed…
Q: Postlab Question: * 1. Under each compound, indicate its most important attractive force. Which of…
A: Note: As per the guidelines, solutions of first question has been made. For the expert solution of…
Q: Draw the Newman projection of cis 1,2-dimethyl-4-t-butyl cyclohexane, and place the t-butyl group in…
A:
Q: What mass of HCl is contained in 27.1 mL of an aqueous HCl solution that has a density of 1.19 g/mL…
A: Given, density of HCl solution = 1.19 g/mL volume of HCl solution = 27.1 mL we know, density = mass…
Q: Predict the position of the equilibrium for the reaction and provide a brief explanation. (The…
A: The equilibrium of acid base reaction always favours the direction where weaker acid and weaker base…
Q: 4. Find the temperature at which water boils on a day in the mountains when the barometric pressure…
A: we have to calculate the boiling point of water at the mountains where pressure is 593 mmHg
Q: 2. Identify which of the following may represents an element, a molecular compound, or an ionic…
A: Matter exists in various forms. In terms of molecular scale, the type of bonding, and the type of…
Q: НО.
A: An organic compound in which C-O-C linkage is present is classified as an ether. Ether can be formed…
Q: How many molecules are in a 7.5L sample of H2 @ STP? Round to 2 decimal places.
A:
Q: why is the blue particle the solvent and the red particle is the solute?
A: In a solution, the solvent particles are typically greater in number than the solute particles…
Q: 9. Write the correct formulas for the following compounds and state if they are molecular or ionic.…
A: we have to fill the given table
Q: ▼ Part A Why is vaporization endothermic? Why is condensation exothermic? Match the words in the…
A: The process in which energy is released is known as exothermic and the process in which energy is…
Q: A pharmacist mixes 100 mL of 38% w/w hydrochloric acid with enough purified water to make 360 mL. If…
A: A pharmacist mixes 100 mL of 38% w/w hydrochloric acid with enough purified water to make 360 mL. If…
Q: Assuming ideal solution behavior, what is the freezing temperature of a solution of 118.4g of…
A:
Q: Assuming 100% dissociation, calculate the freezing point (T₂) and boiling point (T) of 1.81 m CaCl₂…
A: Given that The molality of aqueous CaCl2 solution, m = 1.81 m Assuming 100% dissociation, The…
Q: Which compound in the following pair is the stronger acid? Select the single best answer. Br Br Br…
A: Answer: Strength of the acid depends upon its ability to lose H+ ion, the compound that lose H+ ion…
Please help in answering this
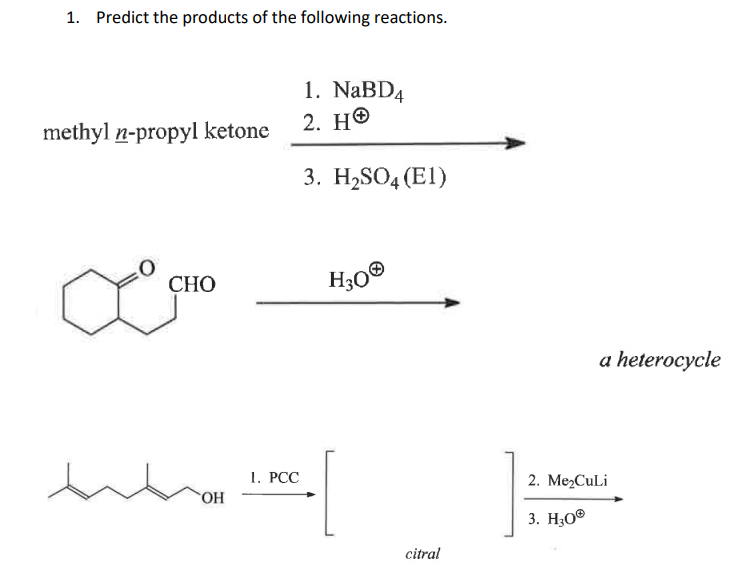
The three chemical reactions are given as follows:
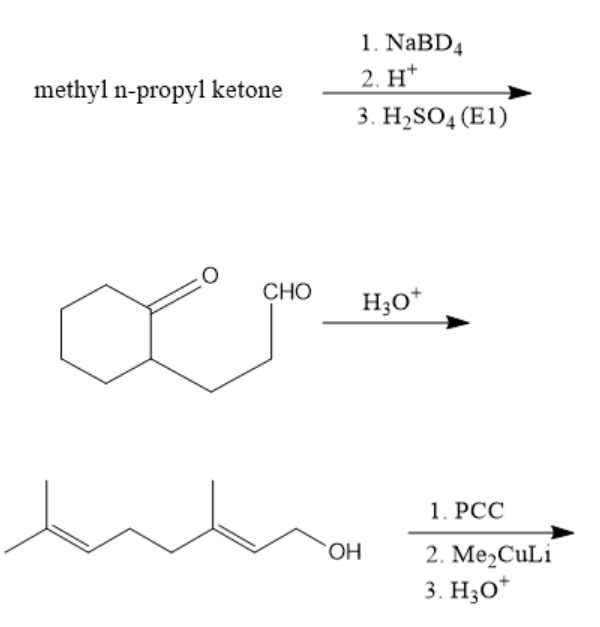
We have to determine the products of each of the above three reactions.
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 7 images

- KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanoneComplete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Predict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the reaction mechanism the given alkene reactionsAlcohols can undergo a lot of different reaction mechanims. If the alcohol group (OH) is attaached to an aromatic core, how will the chemistry change as compared to a typical alkyl alcohol? A) The OH group will become more polarised and more nucleophilic. B) The OH group will become more susceptible to oxidation C) The OH group will become more polarised and therefore basic D) The OH group will become more polarised and therefore acidic.
- When (CH3CH2)3CBr is added to CH3OH at room temperature, the major product is (CH3O)C(CH2CH3)3 and a minor product is CH3CH=C(CH2CH3)2. Propose a mechanism for the product that is formed by the substitution reaction. Use curved arrows to show the movement of electrons.Complete the flowchart by drawing the resulting structures of each reaction. A. B. C. D. E. F. G. H. I.If NH4OH reacts with H2SO4, what will the products be?
- What steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?Give the following reaction A+3B --->2C.1. What type of reaction is occuring in step 3? (halogenation, hydrohalogenation, reduction, keto–enol tautomerism, dehydrohalogenation, acid-catalyzed hydration, base-catalyzed hydration) 2. Which reagent is necessary for step 3? (Br2, HBr, H2/Pt, NaNH2, H20/H2SO4/HgSO4)



