MISSED THIS? Read Section 20.4 (Pages 903-913); Watch KCY 20.4 Decide whether or not each of the following metals dissolves in 1 M HNO₂. For those metals that do dissolve, enter a balanced net lonic redox reaction showing what happens when the metal dissolves Consider the table of standard electrode potentials at 25 C Reduction hat-reaction B (V) 1.50 NO(g) + 2H₂O(1) 096 0.34 0 -0.76 -1.06 Au (aq) + 3e NOS (aq) + 4H(aq) + 3e Cu (aq) +201 21' (aq) + Za (aq) + 2e → Zn(s) AP (aq) + 3+ Al(a) →Au(n) → Cu(s) H₂(g) Y Part A Cu Express your answer as a balanced net ionic equation including phase NOREACTION if there is no reaction. | ΑΣΦ X Submit A chemical reaction does not occur for this question . Previous Answers Request Answer 4
MISSED THIS? Read Section 20.4 (Pages 903-913); Watch KCY 20.4 Decide whether or not each of the following metals dissolves in 1 M HNO₂. For those metals that do dissolve, enter a balanced net lonic redox reaction showing what happens when the metal dissolves Consider the table of standard electrode potentials at 25 C Reduction hat-reaction B (V) 1.50 NO(g) + 2H₂O(1) 096 0.34 0 -0.76 -1.06 Au (aq) + 3e NOS (aq) + 4H(aq) + 3e Cu (aq) +201 21' (aq) + Za (aq) + 2e → Zn(s) AP (aq) + 3+ Al(a) →Au(n) → Cu(s) H₂(g) Y Part A Cu Express your answer as a balanced net ionic equation including phase NOREACTION if there is no reaction. | ΑΣΦ X Submit A chemical reaction does not occur for this question . Previous Answers Request Answer 4
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 160MP
Related questions
Question
3
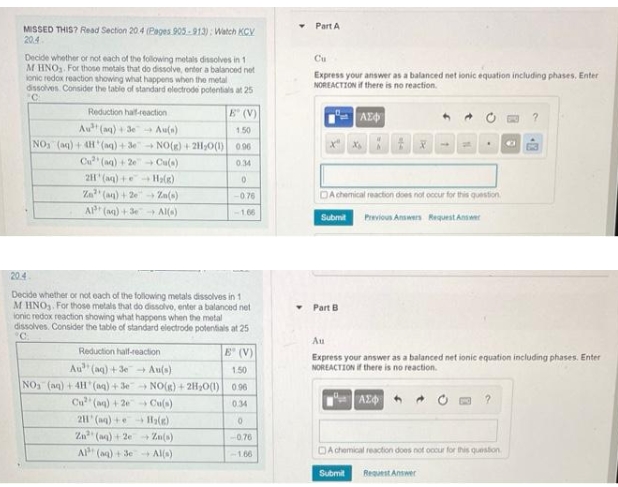
Transcribed Image Text:MISSED THIS? Read Section 20.4 (Pages 905-913): Watch KCV
20.4
Decide whether or not each of the following metals dissolves in 1
M HNO₂. For those metals that do dissolve, enter a balanced net
lonic redox reaction showing what happens when the metal
dissolves. Consider the table of standard electrode potentials at 25
C
Reduction half-reaction
Au (aq) + 3e
NO₂ (aq) + 4H(aq) + 3e
Cu (aq) +2e
2H(aq) +
E (V)
Au(s)
1.50
NO(g) + 2H₂O(1) 0.96
0.34
0
-0.76
-1.66
→ Cu(a)
H₂(g)
Za² (aq) +2e → Za(s)
AP+ (aq) +3e Al(s)
20.4
Decide whether or not each of the following metals dissolves in 1
M HNO₂. For those metals that do dissolvo, enter a balanced net
ionic redox reaction showing what happens when the metal
dissolves. Consider the table of standard electrode potentials at 25
°C.
Au (aq) +3e
NO₂ (aq) + 4H(aq) + 3e
E (V)
Au(s)
1.50
→ NO(g) + 2H₂O(1) 096
→Cu(s)
0.34
Cu (aq) +2e
2H(aq) +
H₂(e)
Zn² (aq) + 2e
→ Zn(s)
Al (aq) + 3e →Al(s)
Reduction half-reaction
0
-0.
-1.66
Y
Part A
Cu
Express your answer as a balanced net ionic equation including phases, Enter
NOREACTION if there is no reaction.
| ΑΣΦ
▾ Part B
X₂
tx
DA chemical reaction does not occur for this question
Submit
Previous Answers Request Answer
.
ΑΣΦ
4
Au
Express your answer as a balanced net ionic equation including phases. Enter
NOREACTION if there is no reaction.
A chemical reaction does not occur for this question
Submit
Request Answer
?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
