MISSED THIS? Watch KCV 8.2, IWE 8.2; Read Section 8.4. You can click on the Review link to access the section in your eText. For each of the following precipitation reactions, calculate how many grams of the first reactant are necessary to completely react with 17.9 g of the second reactant. Part A 2 KI (aq) + Pb(NO3)2 (aq) →PbI2 (s) + 2 KNO3(aq) IVE ΑΣΦ m= 54.2 Submit Previous Answers Request Answer X Incorrect; Try Again; 6 attempts remaining Part B [5] ΑΣΦ Na2CO3 (aq) + CuCl2 (aq) →CuCO3 (s) + 2 NaCl (aq) m= 41.93 ? Submit Previous Answers Request Answer g ? g
MISSED THIS? Watch KCV 8.2, IWE 8.2; Read Section 8.4. You can click on the Review link to access the section in your eText. For each of the following precipitation reactions, calculate how many grams of the first reactant are necessary to completely react with 17.9 g of the second reactant. Part A 2 KI (aq) + Pb(NO3)2 (aq) →PbI2 (s) + 2 KNO3(aq) IVE ΑΣΦ m= 54.2 Submit Previous Answers Request Answer X Incorrect; Try Again; 6 attempts remaining Part B [5] ΑΣΦ Na2CO3 (aq) + CuCl2 (aq) →CuCO3 (s) + 2 NaCl (aq) m= 41.93 ? Submit Previous Answers Request Answer g ? g
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter3: Calculations With Chemical Formulas And Equaitons
Section: Chapter Questions
Problem 3.23QP: High cost and limited availability of a reactant often dictate which reactant is limiting in a...
Related questions
Question
100%
Hi,
I need help with these three questions. please
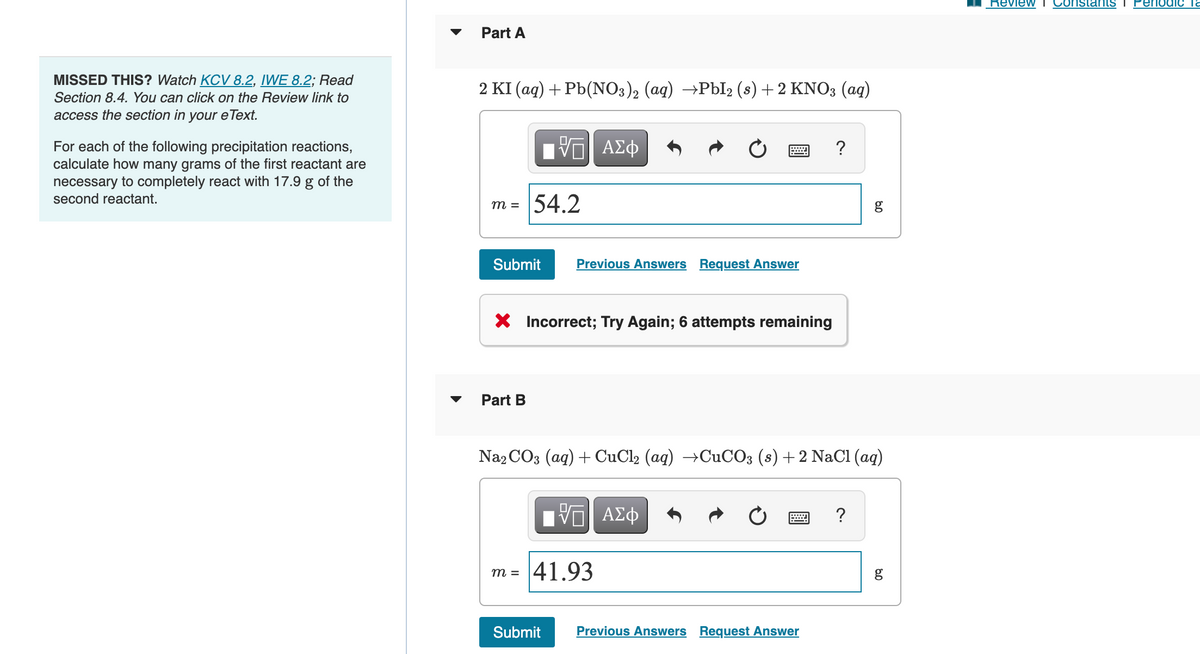
Transcribed Image Text:MISSED THIS? Watch KCV 8.2, IWE 8.2; Read
Section 8.4. You can click on the Review link to
access the section in your eText.
For each of the following precipitation reactions,
calculate how many grams of the first reactant are
necessary to completely react with 17.9 g of the
second reactant.
Part A
2 KI (aq) + Pb(NO3)2 (aq) →PbI2 (s) + 2 KNO3(aq)
m =
Submit
54.2
Part B
VE ΑΣΦ
X Incorrect; Try Again; 6 attempts remaining
Previous Answers Request Answer
Submit
m= 41.93
Na2CO3 (aq) + CuCl2 (aq) →CuCO3 (s)+2 NaCl (aq)
IVE ΑΣΦ
Previous Answers Request Answer
80
?
g
8.0
g
Review I Constants I Periodic la
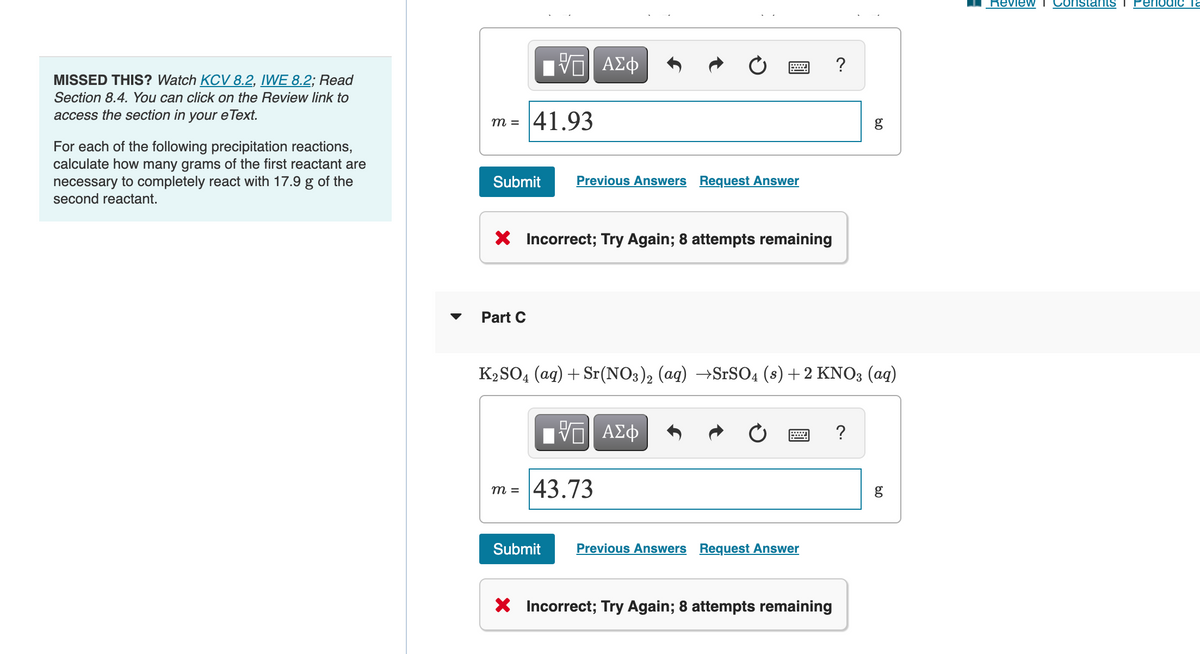
Transcribed Image Text:MISSED THIS? Watch KCV 8.2, IWE 8.2; Read
Section 8.4. You can click on the Review link to
access the section in your eText.
For each of the following precipitation reactions,
calculate how many grams of the first reactant are
necessary to completely react with 17.9 g of the
second reactant.
m =
IV—| ΑΣΦ
41.93
Submit
Part C
Previous Answers Request Answer
X Incorrect; Try Again; 8 attempts remaining
IV—| ΑΣΦ
m = 43.73
K₂SO4 (aq) + Sr(NO3)2 (aq) →SrSO4 (s) + 2 KNO3 (aq)
=
Submit Previous Answers Request Answer
?
X Incorrect; Try Again; 8 attempts remaining
8.0
?
g
8.0
g
Review I Constants I Periodic la
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning